Abstract
Introduction: Budd-Chiari syndrome (BCS) is rare in children. Underlying etiologies, natural history and management differ in children and adults. Treatment options like liver transplantation and transjugular intrahepatic portosystemic shunt have also been less well-studied in children. Presented here is the case of a male child with BCS after coronavirus disease 2019 (COVID-19) infection. Case presentation: A 3-year-old male child presented with complaints of gradually increasing abdominal distension, constipation, decreased urine output, visible veins over abdomen and swelling in lower limbs and periorbital edema since last 15 days. He was diagnosed as BCS after multiple imaging investigations, including triple phase contrast-enhanced computed tomography (CECT) abdomen. His COVID antibody tested positive. Conclusion: Practice guidelines for children with BCS should be formulated, expert group recommendations should be reviewed and a consensus statement should be issued. Underlying etiology remains obscure despite extensive work-up in most of the children. Association of COVID-19 with BCS may be incidental but should be studied further as COVID is known to cause thrombotic complications.
Keywords: Budd-Chiari syndrome, pediatric BCS, COVID-19, TIPS, DIPS, anticoagulation in children
Budd-Chiari syndrome (BCS) is characterized by hepatic vein outflow tract obstruction when there is no cardiac failure or pericardial constriction. BCS was described by George Budd in 1846 as a syndrome of hepatic vein obstruction, followed by clinical-pathological description by Hans Chiari 53 years later, and substantiated by Parker in 1959 implicating thrombophilia as an underlying condition and use of anticoagulants in the mid-1980s. It is a rare liver disorder with an incidence of 0.2 to 4.1 cases per million per year and prevalence of 2.4-7.7 per million in Asian countries and 1.4-4 per million in Western countries. The condition is still considered rare in children. Female predominance in West (52-69%) and male predominance in Asian studies (48-70%) suggests epidemiological differences.
CASE PRESENTATION
A 3-year-old male child presented with complaints of gradually increasing abdominal distension, decreased urine output, visible veins over abdomen (Fig. 1) and swelling in lower limbs and periorbital edema since last 15 days. The child was admitted for complaint of fever 1 month back for 3 days, elsewhere. He had normal development with head circumference - 49 cm, mid arm circumference - 13.7 cm, height - 92 cm (16th percentile) and weight - 12 kg (7th percentile).
Figure 1. Veins over abdomen.
On examination, pallor was present, and there was no lymphadenopathy. Liver was palpable 7 cm below right costal margin and spleen was palpable 5 cm below left costal margin with tense ascites and veins visible.
Routine investigations revealed a hemoglobin level of 10.3 g/dL; a total leukocyte count of 8,680 cells/mm3 and a differential leukocyte count of 88% neutrophils, 11% lymphocytes and 1% monocytes. Platelet count was 3.24 × 103/mm3. Liver function tests showed total serum bilirubin of 1.17 mg/dL and direct 0.47 mg/dL (higher than normal value <0.3 mg/dL/<5.1 µmol/L). Alkaline phosphatase (954 IU/L), and aspartate and alanine aminotransferases (AST/ALT-88/210 IU/L) were also mildly elevated. The total serum proteins were 5.11 g/dL, with albumin levels of 3.6 g/dL. The serum electrolytes and the renal function tests were within normal limits. Prothrombin time (PT) and partial thromboplastin time with kaolin (PTTK) were normal with international normalized ratio (INR) 1.28. The ascitic fluid was grossly yellowish in color with nil red blood cell (RBC) and less than 5 WBC (white blood cell). Biochemical examination showed a protein level of 1.74 g/dL and serum ascites albumin gradient (SAAG) was 1.1. COVID antibody and C-reactive protein (CRP) were positive. Protein C, protein S, homocysteine and antithrombin III were normal. Viral markers for hepatotropic viruses tested negative.
Doppler ultrasonogram abdomen suggested liver size 11 cm, spleen 7 cm with gross ascites with altered echotexture, with periportal cuffing with pulmonary valve (PV) Vmax 214 cm/sec, diameter 5.6 mm (intrahepatic), 6 mm at confluence, peri-gallbladder edema. Right hepatic vein was patent, middle and proximal part of middle hepatic vein was occluded and distal part was patent. Left hepatic vein had ostial stenosis and inferior vena cava (IVC) had stenosis just caudal to cavoatrial junction. Color Doppler studies showed obstruction of the hepatic veins with loss of the normal triphasic flow pattern with flow reversal. The IVC showed narrowing near the opening of the hepatic veins. A radiological diagnosis of BCS was given.
Contrast-enhanced computed tomography (CECT) scan of whole abdomen showed gross amount of free fluid in the peritoneal cavity, perihepatic and perisplenic region. Liver was enlarged with size approximately 126 mm. Portal vein appeared normal. Triple phase CECT abdomen done on Day 9 of admission showed mild hepatomegaly (128 mm) with heterogeneous contrast enhancement. Nonopacification of proximal portion of left and middle hepatic veins was seen. Moderate narrowing was seen at the origin of right hepatic vein and there was delayed faint opacification. Intrahepatic IVC appeared small in caliber. Moderate free fluid was seen in abdomen and pelvis (Fig. 2).
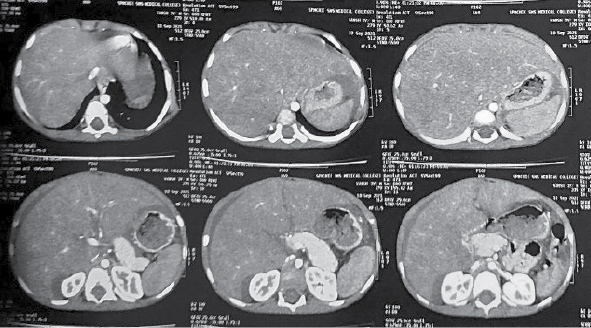
Figure 2. Triple phase CECT abdomen showing BCS.
The child was given protocol treatment by multidisciplinary team including pediatric gastroenterologist, nutritionist and pediatrician. Furosemide and spironolactone in ratio of 2.5:1 was started. Anticoagulation was started with heparin and was switched to long-term warfarin at target INR 2-3. Transjugular hepatic vein and transfemoral IVC balloon angioplasty were considered.
DISCUSSION
Pediatric BCS is a less well-described entity. Usual age of presentation is 10 (1.5-17) years, though 4 to 5 months old children have also been reported to be affected. BCS is responsible for about 3% to 7% of pediatric portal hypertension cases. Symptomatic chronic BCS is a usual presentation in children. Rapidly accumulating tense intractable ascites (83-96%), poorly controlled with diuretics is hallmark of pediatric BCS. Dilated tortuous veins (60-70%) with cephalad flow (above and below umbilicus) can be noted over abdomen and flanks. Similar collaterals over back with flow upwards characterize intrahepatic IVC obstruction. Synthetic functions are relatively preserved at presentation and the child is usually anicteric with firm hepatomegaly, near normal liver enzymes, low to normal albumin and has absence of coagulopathy. Variceal bleeding (8-25%), overt jaundice (13-24%), hepatic hydrothorax (20-36%) and growth failure (28-36%) are relatively uncommon features.
Antecedent thrombotic event resulting in hepatic venous obstruction is often unrecognized. Thrombosis, phlebitis or web in the hepatic veins or IVC are common causes of BCS in children. In our case, middle and proximal part of middle hepatic vein was occluded left ventricular hypertrophy (LHV) had ostial stenosis and IVC had stenosis just caudal to cavoatrial junction. Among the children in India, outflow obstructions include isolated IVC (2-9%), isolated hepatic vein (74-100%) and combined hepatic vein-IVC (23-25%). Secondary causes are relatively rare in children.
Thrombophilia is a vital cause seen in BCS patients. Settings that raise suspicion of thrombophilia include associated portal or mesenteric vein thrombosis, thrombotic event in past, family history, associated systemic diseases, such as inflammatory bowel disease, systemic lupus erythematosus, etc., and recurrent stent block following intervention. Prothrombotic workup even when suggesting low quantitative levels of protein C, protein S, homocysteine and antithrombin III, is considered a measure of poor synthetic functions of the liver and a cause-and-effect relationship can’t be established. Genetic mutation studies of the particular thrombophilia in a child and parents are confirmatory. A thrombophilic state in children requires lifelong anticoagulation therapy.
In our case, the child probably had chronic asymptomatic BCS which later became symptomatic during the pandemic in its natural history; process of chronicity in BCS requires time. Hence, COVID-19 may be an incidental exposure but may not be the causative phenomenon or precipitating factor of decompensation in our case. This should be studied further as COVID-19 is known to cause thrombotic complications.
Though invasive venography ± cavography is the gold standard for diagnosis of BCS, this procedure is deferred in pediatric cases until endovascular intervention is required. Doppler-ultrasonography (DUS), being radiation-free, is the procedure of choice for diagnosis of pediatric BCS (60-96%). Establishing radiological diagnosis requires skills and expertise in pediatric cases. We had to review the radiological investigations repeatedly in this case as imaging is of utmost importance in patients with BCS.
Management guidelines of pediatric BCS are lacking. Duration of disease, site of block, prothrombotic conditions, secondary causes and state of the liver are intervened individually. Balloon angioplasty is preferred in infants and younger children. Due to longer life span as compared to adults, most preferred method is balloon angioplasty with stenting, but poor availability of age-appropriate stent sizes is a limitation. Transjugular intrahepatic portosystemic shunt (TIPS), modified TIPS or direct intrahepatic portocaval shunt (DIPS) are the shunts performed. Anticoagulation starting with heparin at radiological procedure and switching and continuation of long-term warfarin at target INR 2-3 is recommended. Life-long risk of anticoagulation after successful endovascular intervention is a major issue. More data is required on the underlying etiologies and thrombophilia in children. Liver transplantation is considered in end-stage liver disease, failure of radiological management and acute BCS presenting as acute liver failure.
CONCLUSION
Practice guidelines in children with BCS should be formulated and expert group recommendations should be reviewed. The diagnosis of BCS is established through imaging, primarily magnetic resonance imaging (MRI) and ultrasound. Direct features include occlusion of the hepatic veins or the IVC, and hepatic venous collaterals. Indirect features involve the morphological changes that develop secondary to the vascular lesions. Underlying etiology remains obscure despite extensive work-up in most of the children. The relatively enlarged liver in BCS entails choosing a large size stent, raising a query - whether the stent lengthens once the liver has sufficiently decongested or falls short as the child grows? Self-expandable metallic stent use and effects, long-term patency rates, technical and ethical issues of repeated stenting with exposure to repeated radiation during procedures in children over lifetime - these are all issues highlighting the need of prospective studies.
SUGGESTED READING
- Yachha SK, Sarma MS, Srivastava A. Budd-Chiari syndrome in children. In: Qi X (Ed.). Budd-Chiari Syndrome. Springer: Singapore; 2020.
- Riva N, Ageno W. Epidemiology of Budd-Chiari syndrome. In: Budd-Chiari Syndrome. 2020. pp. 15-25.
- Shukla A, Bhatt P, Gupta DK, Modi T, Patel J, Gupte A, et al. Budd-Chiari syndrome has different presentations and disease severity during adolescence. Hepatol Int. 2018;12(6):560-6.
- Nagral A, Hasija RP, Marar S, Nabi F. Budd-Chiari syndrome in children: experience with therapeutic radiological intervention. J Pediatr Gastroenterol Nutr. 2010;50(1):74-8.
- Nobre S, Khanna R, Bab N, Kyrana E, Height S, Karani J, et al. Primary Budd-Chiari syndrome in children: King’s College Hospital experience. J Pediatr Gastroenterol Nutr. 2017;65(1):93-6.
- Squires RH Jr, Shneider BL, Bucuvalas J, Alonso E, Sokol RJ, Narkewicz MR, et al. Acute liver failure in children: the first 348 patients in the pediatric acute liver failure study group. J Pediatr. 2006;148(5):652-8.
- Amarapurkar DN, Punamiya SJ, Patel ND. Changing spectrum of Budd-Chiari syndrome in India with special reference to non-surgical treatment. World J Gastroenterol. 2008;14(2):278-85.
- Singh SK, Sen Sarma M, Yadav R, Kumar S, Prasad R, Yachha SK, et al. Prognostic scoring systems and outcome of endovascular radiological intervention of chronic Budd-Chiari syndrome in children. Liver Int. 2018;38(7):1308-15.
- Valla DC. Budd-Chiari syndrome/hepatic venous outflow tract obstruction. Hepatol Int. 2018;12(Suppl 1):168-80.
- Meyers RL, Maibach R, Hiyama E, Häberle B, Krailo M, Rangaswami A, et al. Risk-stratified staging in paediatric hepatoblastoma: a unified analysis from the Children’s Hepatic tumors International Collaboration. Lancet Oncol. 2017;18(1):122-31.
- Sharma VK, Ranade PR, Marar S, Nabi F, Nagral A. Long-term clinical outcome of Budd- Chiari syndrome in children after radiological intervention. Eur J Gastroenterol Hepatol. 2016;28(5):567-75.
- Mukund A, Gamanagatti S, Acharya SK. Radiological interventions in HVOTO - practical tips. Trop Gastroenterol. 2011;32(1):4-14.
- Qi X, Han G, Guo X, De Stefano V, Xu K, Lu Z, et al. Review article: the aetiology of primary Budd-Chiari syndrome - differences between the West and China. Aliment Pharmacol Ther. 2016;44(11-12):1152-67.