Abstract
Despite being one of the most common gynecological issues faced by women of reproductive age, dysmenorrhea largely remains an ignored, underdiagnosed and untreated condition. It continues to be a public health issue and has a significant impact on the quality of life of the affected women in terms of inability to lead routine activities, absenteeism from academic activities or work and reduced social activities. Currently, existing evidence correlates and implicates the excessive synthesis of prostaglandins with the menstrual pain. Hence, treatment approaches that can inhibit prostaglandins' production or already formed prostaglandins can provide relief in dysmenorrhea. In this review, the impact of dysmenorrhea on the quality of life of women, the role of prostaglandins in the pathogenesis of dysmenorrhea, and how nonsteroidal anti-inflammatory drugs (NSAIDs) like mefenamic acid can be safe and effective in managing dysmenorrhea are discussed.
Keywords: Dysmenorrhea, primary dysmenorrhea, menstrual pain, NSAIDs, mefenamic acid, COX-2 inhibitors, quality of life
Dysmenorrhea is one of the most common gynecological issues faced by women of child-bearing age. It is classified as either primary dysmenorrhea, which does not involve a related organic disease, or secondary dysmenorrhea, which is related to attributable causes, such as endometriosis. The pain associated is described as lower abdominal pain of cramping nature, spreading to the thighs or lower spine, ranging from mild-moderate-severe intensity during the menstruation cycle. The pain in the lower abdomen may also be accompanied by vomiting, headache, back pain, diarrhea and fatigue.1
The global prevalence of primary dysmenorrhea ranges from 45% to 95% in women of reproductive age, while almost 2% to 29% experience severe pain.2 In a study conducted among 550 female students in six universities, the prevalence of primary dysmenorrhea was 80.9%.2 Studies from India have reported the prevalence range from 50% to 87.8%.3
The Impact of Dysmenorrhea on Quality of Life
A study recently published in 2021 has reported that dysmenorrhea leaves a significant adverse effect on almost half of the dysmenorrheic females included in the study. The study participants reported that they experience a disruption of their daily activities and studying abilities. It has been reported that primary dysmenorrhea has a considerable impact on health-related quality of life, daily activities, work productivity and academic performance. Other significant adverse effects reported among the study participants were reduced engagement in social activities, limited activities, missing classes and poor concentration.2 Primary dysmenorrhea without any obvious pathology occurs in almost 50% of menstruating females and causes significant disruption in quality of life and absenteeism from work or school.4
In earlier studies, it has already been suggested that approximately 52% of post-pubescent females suffer from dysmenorrhea; about 10% of these are incapacitated for 1 to 3 days every month. In fact, it was cited to be the largest single cause of lost working hours and school days among young women, with in excess of 140 million working hours estimated to be lost annually.5 In another study conducted in 2004 in Canada, it was reported that among the study participants, 60% experienced dysmenorrhea with moderate-to-severe pain. Fifty-one percent reported restriction of activities, while 17% absenteeism.6
A survey-based study has also shown menstrual-related presenteeism to be widespread in the general female population. In fact, the annual productivity loss due to presenteeism is almost seven times more than the annual productivity loss due to absenteeism. This burden was most pronounced in women under 21 years of age. While absenteeism refers to time away from work or school, presenteeism is defined as loss of productivity while being present at work or school.7
Additionally, the psychological aspect of pain related to dysmenorrhea influences the reactive component of pain. The anticipation of severe dysmenorrhea each month in itself is a cause for lot of stress and unproductivity in women. It is reported that dysmenorrhea is more common in working women or those who have higher stress levels.8
Dysmenorrhea: An Untreated Condition?
Despite being one of the most common and recurring gynecological conditions, dysmenorrhea is usually underdiagnosed, and undertreated.9,10 Besides, dysmenorrheic females do not get sufficient information about the condition, leaving it an inadequately addressed medical problem.2 More often than not, women do not seek medical treatment for dysmenorrhea because they assume symptoms to be normal or tolerable, prefer to self-manage, have limited access to healthcare, they think healthcare providers would not offer help, they are unaware of treatment options, wary of available treatments, feel embarrassed or afraid to seek care, or they rarely take care of any medical ailment, irrespective of dysmenorrhea.9 Sometimes, women undervalue the condition themselves, considering it part of the menstrual cycle, ignoring its implications on quality of life, and hence do not seek treatment for dysmenorrhea.11 The high prevalence of primary dysmenorrhea indicates that it is a very common public health issue and should be addressed immediately. Primary dysmenorrhea is a common, neglected and undertreated gynecological disorder characterized by menstrual pain related to severe physical and psychological symptoms that impair regular daily activities.
Along with the lower abdominal or pelvic pain, it is also associated with other physical (menstrual migraine, lethargy, fatigue, sleepiness or sleeplessness, heaviness in the lower abdomen, nausea, vomiting, backache, pain in knee, muscle/joint/inner thighs) and psychological symptoms (anxiety, depression, irritability).2 Hence, it has a wide range of symptoms typically lasting for 9 to 72 hours, severely disrupting a woman’s quality of life. The plethora of recurring symptoms experienced by women makes dysmenorrhea a serious issue impacting the quality of life in women warranting immediate medical attention.
Pathophysiology of Dysmenorrhea
In the case of primary dysmenorrhea, pain is caused due to excessive, pathological uterine contractions and is marked by the absence of palpable lesions within the lesser pelvis.1 Dysmenorrhea is driven by a complex interplay of various pathogenetic mechanisms including behavioral and psychologic factors and thus is multifactorial.8
On the biochemical level, prostaglandins are a major factor causing the increased abnormal uterine activity. Because of the uterine muscle hypercontractility, the blood flow gets compromised leading to ischemia. Thus, the pain is thought to be due to these below 3 factors as: (i) increased abnormal uterine activity, (ii) uterine ischemia and (iii) sensitization of the nerve terminals by prostaglandins which lower the threshold of these nerves to the action of chemical and physical stimuli. Estrogen and progesterone stimulate the uterus to increase endometrial release of arachidonic acid, through lysosomal labialization and release of phospholipase A2, which in turn acts on the bilipid cell membranes to generate arachidonic acid (Fig. 1). During menstruation, prostaglandin F2α (PGF2α) and prostaglandin E2 (PGE2) induce prolonged uterine contractions that reduce blood flow resulting in uterine ischemia. Further contraction of bowel and vascular smooth muscle by PGF2α and PGE2 lead to nausea, vomiting and diarrhea.12 On the other hand, secondary dysmenorrhea usually results from specific pelvic pathology.13
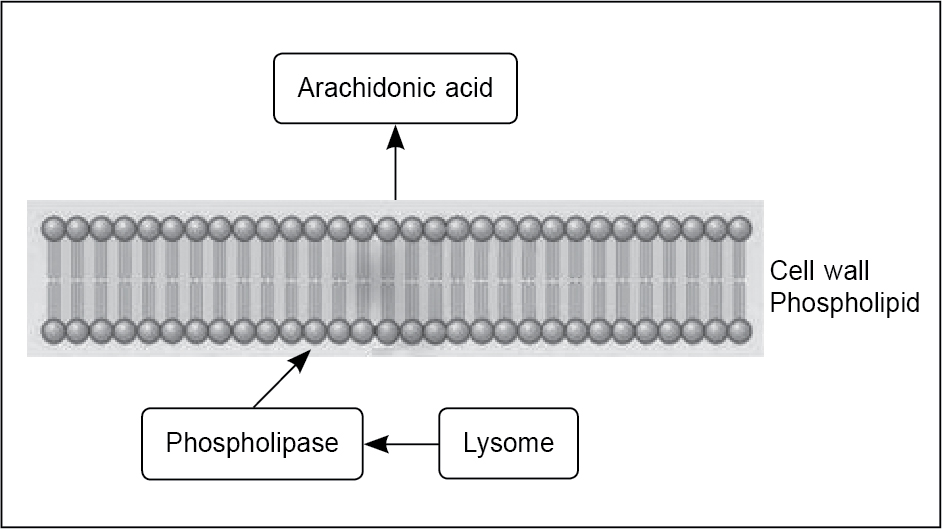
Figure 1. Release of arachidonic acid from lysosomes via phospholipids.
(Image adapted from Smith RP. The role of prostaglandins in dysmenorrhea and menorrhagia. In: Dysmenorrhea and Menorrhagia. 2018. Springer, Cham.)
Role of Prostaglandin in Dysmenorrhea
The uterine activity induced by the excess prostaglandins in patients with primary dysmenorrhea can be very prominent. The normal resting pressure in the uterus is in the range of 5 to 15 mmHg. During normal menstruation, the uterine pressure increases up to 50 to 80 mmHg and helps in expulsion of blood and shed endometrial tissues. However, in women with dysmenorrhea, the uterine pressure peaks over 400 mmHg.14
The increased formation and secretion of uterine prostaglandins during menstruation increases abnormal uterine activity leading to uterine hypoxia and pain.2
Prostaglandins such as PGE2 and cyclic endoperoxides hypersensitize pain fibers in the pelvis and uterus to the action of pain-inducing substances or factors.8 Advances during the last three decades and recent evidence suggests that increased secretion of PGF2α and PGE2 in the endometrium are the important mediators and are involved in increasing the myometrial contractions leading to ischemia and also sensitization of pain fibers to mechanical and physical stimuli.2 PGF2α in dysmenorrhea causes uterine vasoconstriction and contraction of the myometrium, whilst PGE2 causes inflammation and contraction of the myometrium.11
The role of prostaglandin is supported by the fact that there is a similarity between symptoms of primary dysmenorrhea and prostaglandin-induced uterine contractility in labor or abortion; and an increased level of prostaglandin in the menstrual fluid of women with primary dysmenorrhea.11 Studies have also reported a significant difference in the mean levels of PGF2 between the moderate primary dysmenorrhea group and the severe primary dysmenorrhea group. It has been demonstrated that the PGF2α level was higher in the severe primary dysmenorrhea patient group than the moderate primary dysmenorrhea group with a significant relationship between the level of PGF2α and the intensity of pain in primary dysmenorrhea (p = 0.000).15
It has been observed that during normal ovulatory menstruation, both PGE2 and PGF2α have low levels in the proliferative phase, slightly increase during the early luteal phase and then increases to fourfold in the late luteal phase and sixfold during the menstrual phase.16
The current evidence suggests that women with primary dysmenorrhea have 2 to 7 times higher PGF2α than in women without primary dysmenorrhea.17 Prostaglandins are formed through arachidonic acid, which is present in cell membrane phospholipids. Its production is triggered via the cyclic adenosine monophosphate (cAMP) pathway by adrenaline, peptide and steroid hormones, mechanical stimuli and tissue trauma (Fig. 2).18 Figure 2 shows the pathway producing pain in dysmenorrhea.
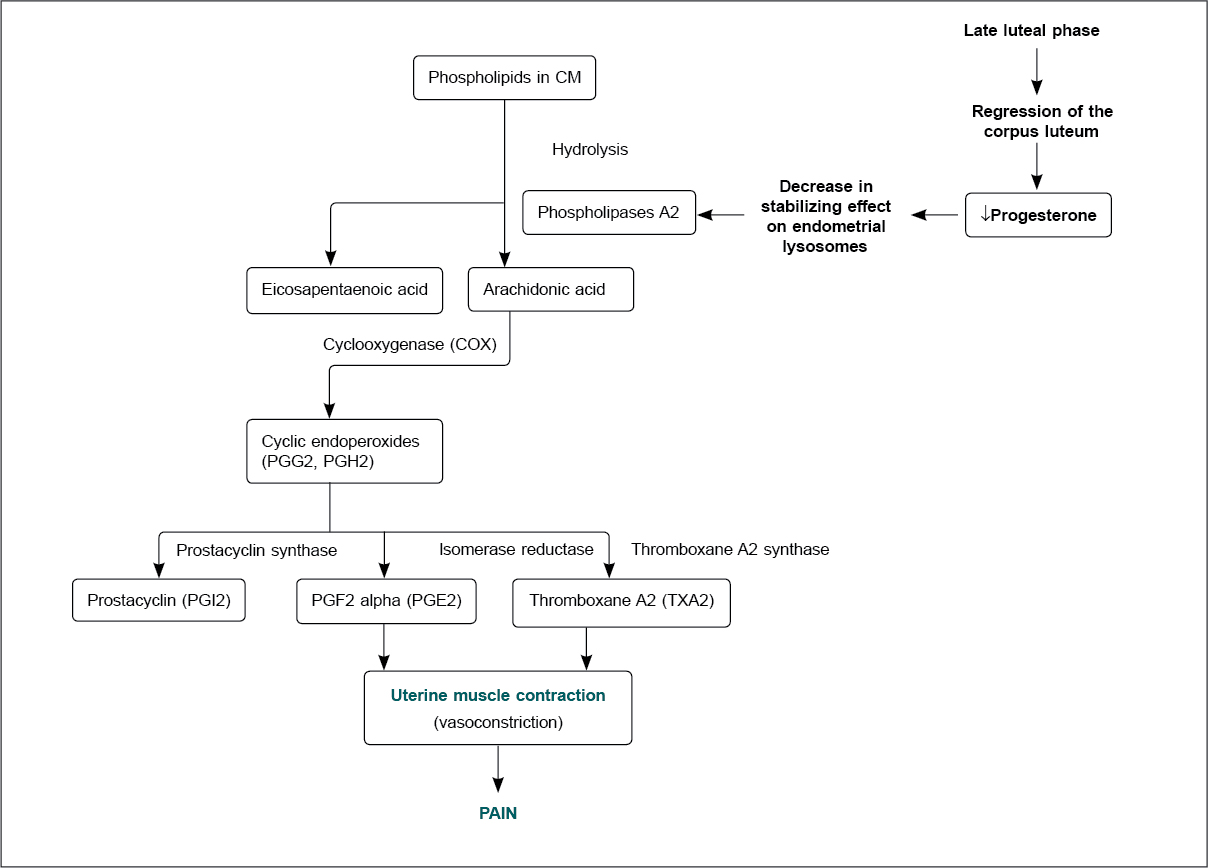
Figure 2. Prostaglandin synthesis in dysmenorrhea.
It has been established that the increased formation of arachidonic acid, intracellular destruction and tissue trauma during menstruation is responsible for enhancing prostaglandin production.11,18 With an increasing prostaglandin concentration, the severity of menstrual pain and associated symptoms also increases.18
Other Mediators in Dysmenorrhea
We have seen dysmenorrhea is considered to be an inflammatory event primarily driven by prostaglandin, however, there are several other concerning factors involved in dysmenorrhea.
While arguable, vasopressin may be involved in producing dysrhythmic uterine contractions, causing reduced uterine flow, hypoxia and eventually pain. A study has also linked nitric oxide to the uterine quiescence during pregnancy.18,19
Experimental studies have revealed high uterine histamine levels, mediated by estradiol, and greater uterine contractility and this may be an indication of myometrial histamine receptors (uterine H1 receptors).20 Besides, in some women, a reduction of diamine oxidase levels is also reported and a resultant severe effect of histamine at the beginning of the menstruation, further corroborating the hypothesis that histamine is one of the significant factors causing dysmenorrhea.20
A study conducted among dysmenorrheic patients revealed a significant association between dysmenorrhea and increased mast cells in the uterine cervix. The release of mast cell mediators such as histamine, prostaglandin and serotonin in the uterine myometrium evokes strong contractions.21
Preclinical studies have also shown that bradykinin induces uterine smooth muscle contractions via activating phospholipase C.22,23 The pain in the pelvis results from an increased uterine activity, uterine ischemia and hypersensitization of pain fibers by the action of bradykinin and other physical stimulus.24 It has also been shown that the response to bradykinin is dependent on the presence of prostaglandins, especially PGE2. It has been suggested that bradykinin has a self-sensitizing action and stimulates the release of PGE2.25 In addition, we know that cholinergic nerves form moderately dense plexuses in the myometrium, cervical smooth muscles and microarterial system of the uterine horns and cervix. Acetylcholine also stimulates contraction of the myometrium26 suggesting its role in the pathogenesis of dysmenorrhea by inducing contractions in the uterus.27 Inflammatory cytokines also undergo changes in concentration during menstruation including tumor necrosis factor (TNF)-α, interleukin (IL)-6 which are higher in dysmenorrheic women compared to healthy women.1
Treatment of Dysmenorrhea
Approach to Management
The approach to managing dysmenorrhea includes nonpharmacological, pharmacological and surgical options. Nonpharmacological approaches to treat dysmenorrhea include lifestyle changes such as proper diet, exercise, smoking cessation, low alcohol consumption and use of topical heat or transcutaneous electrical nerve stimulation (TENS) to provide relief from pain and discomfort. Nonsteroidal anti-inflammatory drugs (NSAIDs) are the best-established initial therapy for dysmenorrhea,28 and they have a direct analgesic effect through inhibition of prostaglandin synthesis, and they decrease the volume of menstrual flow.
Combined oral contraceptive is the second line of treatment, and works by decreasing menstrual volume and prostaglandin secretion, with a resultant decrease in intrauterine pressure and uterine contractility.11 In rare cases, a surgical approach may be needed for women with severe and refractory dysmenorrhea.29
NSAIDs in the Treatment of Dysmenorrhea
An effective treatment of primary dysmenorrhea targets all the components of the pain causing pathways, along with focusing on suppression of uterine contraction.24 Pharmacological agents that inhibit cyclooxygenase (COX) lead to reduced prostaglandin synthesis, and its concentration in menstrual fluid decreases uterine contractility and menstrual volume.4
The cramps experienced during menstruation and the intensity of pain are correlated to the high concentration of PGF2α and PGE2 in the endometrium, making NSAIDs the cornerstone in the management of dysmenorrhea.2 The understanding of the pathogenesis of pain in primary dysmenorrhea (as explained in the previous section) provides the rationale for the use of NSAIDs for the relief of primary dysmenorrhea instead of a pharmacotherapeutic agent which only acts to block uterine contractions.8
NSAIDs are used as the first-line therapy in the treatment of dysmenorrhea.30 They act by inhibiting prostaglandin production through the blockade of COX activity. The common painkiller NSAIDs used in dysmenorrhea include aspirin, naproxen, ibuprofen and mefenamic acid.31 Clinical evidence has shown that most NSAIDs are more potent than paracetamol in managing primary dysmenorrhea. It has also been shown that NSAIDS are two times more effective than paracetamol for pain relief.2 Out of these agents, only mefenamic acid has dual action of inhibiting prostaglandin synthesis and blocking of EP receptors, which makes it unique and more effective for treating patients with dysmenorrhea.31 The ability of mefenamic acid to inhibit pre-existing prostaglandin causes a faster onset of uterine relaxation providing better pain relief.16
Mefenamic acid in Dysmenorrhea
Mefenamic acid is commonly used NSAID for the treatment of dysmenorrhea and is widely available across several regulated markets viz. European countries, USA, Canada, Australia, New Zealand, Japan, Hong Kong, India, Malaysia, Thailand and Singapore. Given in a usual oral dose of 500 mg thrice daily, it is one of the most highly prescribed medicines across the globe.32
Several studies provide the evidence of mefenamic acid providing adequate pain relief and also reducing the volume of blood loss in dysmenorrheic women associated with menorrhagia.29,31
Several studies have shown superior efficacy of mefenamic acid in reliving the pain in dysmenorrhea over placebo and other NSAIDs like ibuprofen, naproxen, indomethacin, combination of dextropropoxyphene and paracetamol.33-35
Mefenamic acid also acts as a bradykinin antagonist, and hence may have an extended pathway of pain relief.33 It can also inhibit the increased uterine contractility stimulated by prostaglandin-endoperoxide analogs.34 Mefenamic acid also reduces the high uterine resting pressure or tone, and the frequency of contractions.35 Furthermore, it has been seen that mefenamic acid reduces the mean menstrual blood loss by 40%. It acts via an improvement of platelet aggregation and degranulation and through increased vasoconstriction.36 This is a desirable benefit of mefenamic acid.37 This reduction in menstrual blood loss was also seen to sustain at 6 to 9 months and at 12 to 15 months in women with menorrhagia treated with mefenamic acid during all menstrual periods for over a year. Significant sustained decrease in blood loss were observed in women with menorrhagia due to ovulatory dysfunctional bleeding and in those who had undergone tubal sterilization.38
In patients with dysmenorrhea, paracetamol does not work effectively as it cannot eliminate the cause of pain in dysmenorrhea, particularly, the presence of excess prostaglandins in the endometrium. A study has revealed that paracetamol was not better than a placebo in relieving primary dysmenorrhea. Usually, the paracetamol dose available for pain relief is 500 mg which is not effective as an analgesic for acute, mild-to-moderate pain. However, high-dose immediate-release paracetamol (1,000 mg), has analgesic action than lower doses for adults with mild-to-moderate acute pain. However, those suffering from pain want to achieve either a substantial reduction in their pain intensity or to achieve a low pain state, which may not be possible with suboptimal doses of paracetamol available in most marketed paracetamol preparations for pain relief.39
Combination Treatment
Women with primary dysmenorrhea experience four different uterine contraction-related abnormalities including increased uterine resting tone, increased active pressure, increased number of contractions and dysrhythmic uterine activity.8
Studies have also shown that the combined administration of NSAIDs and antispasmodics is beneficial in treatment of dysmenorrhea. Given the mechanism of prostaglandin-mediated pain, NSAIDs that inhibit COX pathway may be the ideal therapeutic path. However, owing to the more complex physiology of dysmenorrhea, use of spasmolytic agents acting on the uterine muscle contraction becomes imperative.40
Studies have demonstrated the synergistic activity of mefenamic acid with dicyclomine in dysmenorrhea patients.41 The combination emerged as a highly effective and well-tolerated option for treating spasmodic dysmenorrhea and also for additional concomitant benefits in menorrhagia. Mefenamic acid relieves primary dysmenorrhea mainly by inhibiting endometrial prostaglandin formation, restoring normal uterine activity.42 An observational study reported that a fixed-dose combination of mefenamic acid and dicyclomine was effective in restoring the functional ability of the patients with dysmenorrhea.43 Dicyclomine has a dual action of inhibiting muscarinic action of acetylcholine of postganglionic parasympathetic effector regions and antispasmodic action via its direct action on the uterine smooth muscles.44,45 Their synergistic activity is also proven by the fact that dicyclomine given alone was less efficacious in moderate and severe dysmenorrhea.46 A comparative study conducted among healthcare practitioners revealed that mefenamic acid and dicyclomine was used by 77% of the study participants for pain relief, of which 35% used it for dysmenorrhea.47 The anticholinergic side effects of dicyclomine are not prominent in the combination owing to the lower drug dose used.
Table 1 shows different agents used along with mefenamic acid in dysmenorrhea.40,48
|
Table 1. Different Spasmolytic Agents Used in Dysmenorrhea
|
|
Drug
|
Mechanism of action
|
Combination with mefenamic acid
|
|
Dicyclomine
|
Relieves smooth muscle spasm via dual mechanism. A specific anticholinergic effect at the acetylcholine receptor site and a direct smooth muscle antagonism of bradykinin- and histamine-induced spasms.
|
Symptomatic relief from menstrual pain and cramps. It is also used to treat abdominal pain by relieving spasms of the muscles in the stomach and intestines.40
|
|
Drotaverine
|
Direct spasmolytic action, selective inhibitor of phosphodiesterase-4 (PDE-4).
Does not have anticholinergic effects.
|
Antispasmodic agent with noncholinergic action.40
|
|
Camylofin
|
Both anticholinergic action as well as direct smooth muscle action; PDE-4 inhibitor.
|
Pain relief, but results were not statistically significant compared with dicyclomine + mefenamic acid.48
|
Safety and Efficacy of Mefenamic Acid
Mefenamic acid is generally well-tolerated, but side effects can include headache, dizziness, somnolence, nausea, diarrhea, abdominal discomfort, heartburn, peripheral edema and hypersensitivity reactions.49 The combination of mefenamic acid and dicyclomine is better tolerated as due to its antimuscarinic and spasmolytic action, dicyclomine reduces the gastrointestinal side effects that may be associated with mefenamic acid.46
There are certain concerns about the hepatotoxicity of mefenamic acid; however, in prospective studies, less than 5% of patients taking mefenamic acid experienced transient rise in serum aminotransferase levels. The abnormalities usually reduced even while the drug was continued and without dosage adjustment. There may have been rare cases of mefenamic acid-induced liver injury but mefenamic acid is not a causative agent in any large case series on drug-induced liver injury.49
Patients with dysmenorrhea need to take mefenamic acid irregularly, hence it has lesser chances of interfering with the COX system and presents a lower cardiovascular risk compared with other drugs such as celecoxib.50
Conclusion
Dysmenorrhea affects a significant population of women across the globe, but it continues to be a neglected health issue, with considerable underuse of medications. Even though it is not a life-threatening condition, it leads to a significant burden on the quality of life of female adolescents or women interfering with daily activities. Besides, another challenge affecting dysmenorrhea management is insufficient information or lack of awareness about primary dysmenorrhea among the younger population. NSAIDs are the first line of treatment in the treatment of dysmenorrhea and amongst the NSAIDs, mefenamic acid is a preferential COX-2 inhibitor that can effectively reduce menstrual pain with fewer side effects. Mefenamic acid is one of the most thoroughly investigated agents for the treatment of dysmenorrhea, and the results have been consistently effective in reducing subjective discomfort. One of the first approved drugs for dysmenorrhea in the USA, mefenamic acid is very effective in improving symptoms and changing the underlying pathophysiology of the disease. Mefenamic acid has a multifactorial action in offering relief from dysmenorrhea including inhibition of prostaglandin, decrease in uterine tonicity, and bradykinin-antagonistic activity. Considering the complex pathophysiology of dysmenorrhea involving multiple pathways, combination therapies using NSAID, and other antispasmodic agents becomes imperative to treat the condition. For instance, the synergistic combination of dicyclomine and mefenamic acid is effective and safe in treating pain and discomfort related to dysmenorrhea.
In an era of women empowerment, every woman needs to be in the optimum physiological and psychological condition. Hence, it becomes all the more important to address the menstrual burden and provide effective treatment. Healthcare professionals can enhance awareness in their women patients about the problems associated with dysmenorrhea. They can provide educational literature, posters or websites such as www.painfulperiods.in where patients can get ready access to authentic information curated by the doctors.
References
- Barcikowska Z, Rajkowska-Labon E, Grzybowska ME, Hansdorfer-Korzon R, Zorena K. Inflammatory markers in dysmenorrhea and therapeutic options. Int J Environ Res Public Health. 2020;17(4):1191.
- Karout S, Soubra L, Rahme D, Karout L, Khojah HM, Itani R. Prevalence, risk factors, and management practices of primary dysmenorrhea among young females. BMC Womens Health. 2021;21(1):392.
- Omidvar S, Bakouei F, Amiri FN, Begum K. Primary dysmenorrhea and menstrual symptoms in Indian female students: prevalence, impact and management. Glob J Health Sci. 2015;8(8):53632.
- Dawood MY. Primary dysmenorrhea: advances in pathogenesis and management. Obstet Gynecol. 2006;108(2):428-41.
- Ylikorkala O, Dawood MY. New concepts in dysmenorrhea. Am J Obstet Gynecol. 1978;130(7):833-47.
- Burnett MA, Antao V, Black A, Feldman K, Grenville A, Lea R, et al. Prevalence of primary dysmenorrhea in Canada. J Obstet Gynaecol Canada. 2005;27(8):765-70.
- Schoep ME, Adang EMM, Maas JWM, De Bie B, Aarts JWM, Nieboer TE. Productivity loss due to menstruation-related symptoms: a nationwide cross-sectional survey among 32 748 women. BMJ Open. 2019;9(6):1-10.
- Dawood MM. Dysmenorrhea. Glob Libr Women’s Med. 2009. Available from: http://www.glowm.com/section-view/heading/Dysmenorrhea/item/9. Accessed Jan 17, 2022.
- Chen CX, Shieh C, Draucker CB, Carpenter JS. Reasons women do not seek health care for dysmenorrhea. J Clin Nurs. 2018;27(1-2):e301-8.
- Ramos-Pichardo JD, Ortega-Galán ÁM, Iglesias-López MT, Abreu-Sánchez A, Fernández-Martínez E. Why do some Spanish nursing students with menstrual pain fail to consult healthcare professionals? Int J Environ Res Public Health. 2020;17(21):8173.
- Guimarães I, Póvoa AM. Primary dysmenorrhea: assessment and treatment. Rev Bras Ginecol Obstet. 2020;42(8):501-7.
- Rainone F. Dysmenorrhea (Chap 56). In: Integrative Medicine. 2nd Edition, Elsevier; 2007. pp. 613-7. Available from: https://www.sciencedirect.com/science/article/pii/B9781416029540500600. Accessed Jan 31, 2022.
- Osayande AS, Mehulic S. Diagnosis and initial management of dysmenorrhea. Am Fam Physician. 2014;89(5):341-6.
- Smith RP, Powell JR. Simultaneous objective and subjective evaluation of meclofenamate sodium in the treatment of primary dysmenorrhea. Am J Obstet Gynecol. 1987;157(3):611-8.
- Fajrin I, Alam G, Usman AN. Prostaglandin level of primary dysmenorrhea pain sufferers. Enfermería Clínica. 2020;30(2):5-9.
- Downie J, Poyser NL, Wunderlich M. Levels of prostaglandins in human endometrium during the normal menstrual cycle. J Physiol. 1974;236(2):465-72.
- Smith RP, Ellis J. NSAIDs: is newer better for dysmenorrhea? OBG Manag. 2002;14(7):71-81.
- Iacovides S, Avidon I, Baker FC. What we know about primary dysmenorrhea today: a critical review. Hum Reprod Update. 2015;21(6):762-78.
- Buhimschi I, Yallampalli C, Dong YL, Garfield RE. Involvement of a nitric oxide-cyclic guanosine monophosphate pathway in control of human uterine contractility during pregnancy. Am J Obstet Gynecol. 1995;172(5):1577-84.
- Jarisch R. Histamine intolerance in women. In: Jarisch R (Eds.) Histamine Intolerance: Histamine and Sea sickness. Springer, Berlin, Heidelberg; 2015. pp. 109-15. Available from: https://link.springer.com/chapter/10.1007/978-3-642-55447-6_6
- Tsuji S, Tsuji K, Otsuka H, Murakami T. Increased mast cells in endocervical smears of women with dysmenorrhea. Cytojournal. 2018;15(1):27.
- Hahn DW, Carraher R, McGuire JL. Effects of suprofen and other prostaglandin synthetase inhibitors in a new animal model for myometrial hyperactivity. Prostaglandins. 1982;23(1):1-16.
- Wassdal I, Nicolaysen G, Iversen JG. Bradykinin causes contraction in rat uterus through the same signal pathway as oxytocin. Acta Physiol Scand. 1998;164(1):47-52.
- Dawood MY. Current concepts in the etiology and treatment of primary dysmenorrhea. Acta Obstet Gynecol Scand Suppl. 1986;138:7-10.
- Maubach KA, Grundy D. The role of prostaglandins in the bradykinin-induced activation of serosal afferents of the rat jejunum in vitro. J Physiol. 1999;515(Pt 1):277.
- Papka RE, Traurig HH, Schemann M, Collins J, Copelin T, Wilson K. Cholinergic neurons of the pelvic autonomic ganglia and uterus of the female rat: distribution of axons and presence of muscarinic receptors. Cell Tissue Res. 1999;296(2):293-305.
- Sharghi M, Mansurkhani SM, Ashtary-Larky D, Kooti W, Niksefat M, Firoozbakht M, et al. An update and systematic review on the treatment of primary dysmenorrhea. JBRA Assist Reprod. 2019;23(1):51-7.
- Zahradnik HP, Hanjalic-Beck A, Groth K. Nonsteroidal anti-inflammatory drugs and hormonal contraceptives for pain relief from dysmenorrhea: a review. Contraception. 2010;81(3):185-96.
- Speer L. Dysmenorrhea. Am Fam Physician. 2005;71(2):285-91.
- Navvabi Rigi S, Kermansaravi F, Navidian A, Safabakhsh L, Safarzadeh A, Khazaeian S, et al. Comparing the analgesic effect of heat patch containing iron chip and ibuprofen for primary dysmenorrhea: a randomized controlled trial. BMC Womens Health. 2012;12:25.
- Marjoribanks J, Ayeleke RO, Farquhar C, Proctor M. Nonsteroidal anti-inflammatory drugs for dysmenorrhoea. Cochrane Database Syst Rev. 2015;2015(7):CD001751.
- Moll R, Derry S, Moore RA, McQuay HJ. Single dose oral mefenamic acid for acute postoperative pain in adults. Cochrane Database Syst Rev. 2011;2011(3):CD007553.
- Collier HOJ, Shorley PG. Antagonism by mefenamic and flufenamic acids of the bronchoconstrictor action of kinins in the guinea-pig. Br J Pharmacol Chemother. 1963;20(2):345-51.
- Sanger GJ, Bennett A. Fenamates may antagonize the actions of prostaglandin endoperoxides in human myometrium. Br J Clin Pharmacol. 1979;8(5):479-82.
- Pulkkinen MO. Suppression of uterine activity by prostaglandin synthetase inhibitors. Acta Obstet Gynecol Scand Suppl. 1979;87:39-43.
- van Eijkeren MA, Christiaens GCML, Geuze HJ, Haspels AA, Sixma JJ. Effects of mefenamic acid on menstrual hemostasis in essential menorrhagia. Am J Obstet Gynecol. 1992;166(5):1419-28.
- Kintis GA, Coutifaris B. Treatment of primary dysmenorrhea with mefenamic acid. Int J Gynaecol Obstet. 1980;18(3):172-5.
- Fraser IS, McCarrom G, Markham R, Robinson M, Smyth E. Long-term treatment of menorrhagia with mefenamic acid. Obstet Gynecol. 1983;61(1):109-12.
- Gaul C, Eschalier A. Dose can help to achieve effective pain relief for acute mild to moderate pain with over-the-counter paracetamol. Open Pain J. 2018;11(1):12-20.
- Janczura M, Kobus-Moryson M, Sip S, Zarowski M, Warenczak A, Cielecka-Piontek J. Fixed-dose combination of NSAIDs and spasmolytic agents in the treatment of different types of pain—A practical review. J Clin Med. 2021;10(14):3118.
- Prajapati D, Raj H. Simultaenous estimation of mefenamic acid and dicyclomine hydrochloride by spectroscopic methods. Int J Pharm Sci Res. 2012;3(10):3766-75.
- KM D. Mefenamic acid with dicyclomine for dysmenorrhoea. Indian Pract. 1999;52(11):767-72.
- Dysmenorrhoea Expert Study Group. Mefenamic acid & dicyclomine: a fixed dose combination in dysmenorrhoea. Indian Pract. 2005;58(1):21-4.
- Kulkarni SG, Mehendale HM. Anticholinergics. Encycl Toxicol. 2005;146-8.
- Vardanyan RS, Hruby VJ. Anticholinergic drugs (Chap 14). In: Vardanyan RS, Hruby VJ (Eds.). Synthesis of Essential Drugs. Elsevier; 2006. pp. 195-208. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780444521668500145
- Sugumar R, Krishnaiah V, Channaveera GS, Mruthyunjaya S. Comparison of the pattern, efficacy, and tolerability of self-medicated drugs in primary dysmenorrhea: a questionnaire based survey. Indian J Pharmacol. 2013;45(2):180-3.
- Gabhane M, Braganza L. Preference trends for antispasmodics among Indian healthcare professionals: results of a cross sectional survey. Indian Pract. 2015;68(5):32-7.
- Dhandapani S, Das S, Gangadhar A. A Randomised, comparative study to evaluate the efficacy and tolerability of two fixed dose combinations of camylofin & mefenamic acid and dicyclomine & mefenamic acid in the management of primary dysmenorrhea. Indian Pract. 2017;70(2):16-20.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Mefenamic Acid. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548029/
- Dong YH, Chang CH, Wu LC, Hwang JS, Toh S. Comparative cardiovascular safety of nonsteroidal anti-inflammatory drugs in patients with hypertension: a population-based cohort study. Br J Clin Pharmacol. 2018;84(5):1045-56.