Published in IJCP
April 2022
Review Article
The Journey and Excitement Following the Development of the First Peptide in a Pill: A Brief Overview of Pharmacology and Clinical Trials of Oral Semaglutide
April 15, 2022 | Banshi Saboo, Manoj Chawla, Sanjay Agarwal, Mithun Bhartia
Diabetes & Endocrinology
Abstract
Oral semaglutide is the newest discovery, the first in class peptide in a pill. Sodium N-(8-[2-hydroxybenzoyl]amino)caprylate (SNAC), a small fatty acid, has been co-formulated with semaglutide, which facilitates its absorption from the gastric mucosa. It has 94% homology with human glucagon-like peptide 1 (GLP-1). It comes in three dose forms – 3 mg, 7 mg and 14 mg. It is given as once daily dosing and is recommended in adult type 2 diabetes mellitus patients as monotherapy when metformin is contraindicated or not tolerated and in combination with other oral antidiabetic drugs (OADs). In a phase 3 trial, it has been shown to reduce glycated hemoglobin (HbA1c) up to 1.5%, with weight reduction up to 5 kg with a 14 mg dose. There was nonsignificant risk reduction of 21% in 3-point major adverse cardiovascular events (MACE) and 51% and 49% risk reduction in cardiovascular (CV) deaths and all-cause mortality, respectively. Oral semaglutide was found to be superior to empagliflozin, sitagliptin and liraglutide in both glycemic control and weight reduction. It also exhibits many pleiotropic effects – reduced energy intake, anti-inflammatory and anti-atherosclerotic effect, to name a few. Nausea was the most common side effect which was experienced by only 15% to 20% of patients. It was mild-to-moderate and transient. Overall, oral semaglutide has shown its efficacy both early and late in the management of diabetes, irrespective of renal and hepatic impairment.
Keywords: Oral semaglutide, GLP-RAs, PK-PD, PIONEER Program, safety, PIP
CHALLENGES MANAGING DIABETES IN INDIA
Need of GLP-RA and Peptide in a Pill
India ranks second in the world with over 74 million people with diabetes (PwD). This number is expected to increase to 124.9 million by 2045.1 We account for 87% of total PwD in Sout-East Asia region.1 Patients with complications incur expenses three times more than those without complications.2 Two out of three PwD have diabetes-related complications, and with direct medical cost of INR 50,000 crores.3 It is also a well-established fact that 7 out of 10 PwD fail to achieve the glycated hemoglobin (HbA1c) target of <7%.4 Uncontrolled, long-standing diabetes leads to grave complications, ultimately increasing mortality and morbidity and poor quality of life. The most common cause of death among PwD are cardiovascular diseases (CVDs), which results from macrovascular complications. There are more than 6 lakh deaths due to diabetes in India.5
Most of the time, diabetes remains a chance diagnosis, when a patient suffers from one or the other complications and visits a healthcare center for the first time. Even though diabetes is a metabolic disease, the patients visit a cardiologist directly with the complication for the first time and it has been observed that a diabetes patient with a complication visits a cardiologist more often than a diabetologist.6-10
Traditionally, we only had few options to manage diabetes, for example biguanides, sulfonylureas and primitive insulins. But over a period, there have been many developments with numerous modern and newer therapy options – Incretin-based therapy like dipeptidyl peptidase-4 (DPP-4) inhibitors and glucagon-like peptide-1 receptor agonists (GLP-1RAs) and sodium-glucose cotransporter-2 (SGLT2) inhibitors. Also, our understanding of natural history of diseases has also evolved. Now diabetes is not merely a disease where we only have imbalance of insulin and glucagon action, but rather a cardio-metabolic syndrome. It is imperative to include the therapies targeting most of the pathophysiological derangements in diabetes.11
The CAPTURE study states that 1 in 3 PwD have CVDs and of them, 85.8% are atherosclerotic in origin, and 85% of deaths among PwD with CVDs are due to atherosclerotic cardiovascular disease (ASCVD), again reiterating the importance of cardio-metabolic management approach in PwD.12 However, in spite of the alarming facts, only 2 out of 10 patients with diabetes and ASCVD are getting a glucose-lowering treatment with a proven cardiovascular (CV) benefit. There are many barriers to the management of diabetes of which nonadherence, self-medication, alternate medicine and fear of injection are few important determinants of poor outcome.13 Primary nonadherence may be particularly relevant among patients who refuse to initiate injectable hypoglycemic therapy, typically due to injection phobia, inconvenience, poor patient-physician communication and/or negative patient perceptions.14 It has been established that fear of injection is associated with poor glycemic control, increased incidence of complications and mortality among PwD.
An ideal class of modern noninsulin antidiabetic drugs (MNIADs) for PwD should have the following features: Efficient glycemic control, no to low risk of hypoglycemia, has meaningful weight reduction, established CV safety, tolerable adverse effects, acts on most of the pathophysiological pathways (DeFronzo ominous octet [Fig. 1a]15), and has a feasible mode of administration.
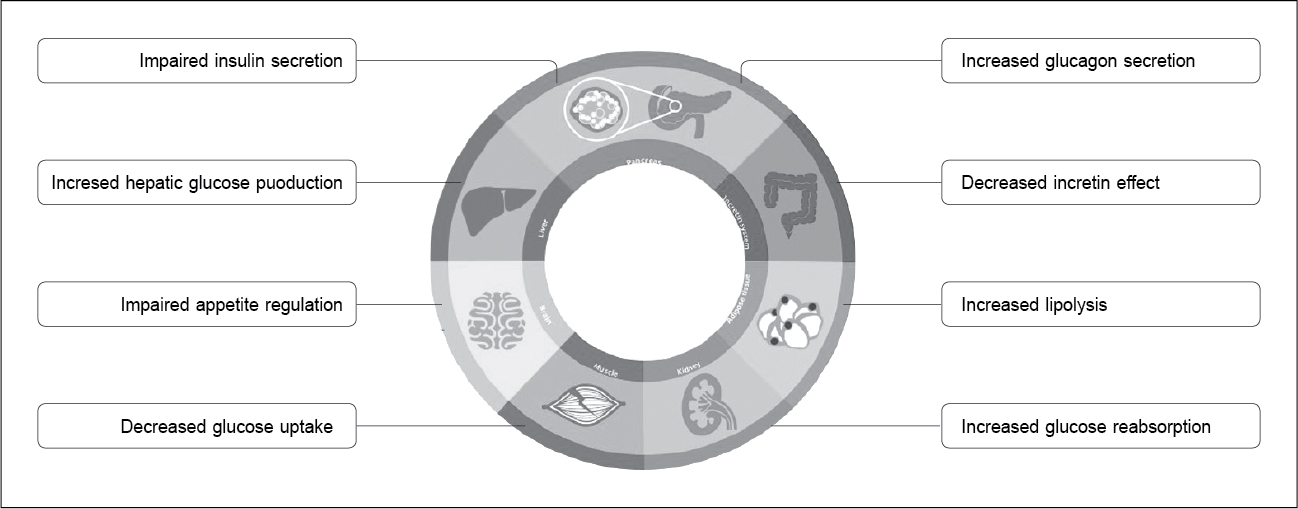
Figure 1a. DeFronzo ominous octet.
HOW THE INNOVATION OF ORAL SEMAGLUTIDE HAPPENED (STRUCTURAL CHANGES)
GLP-1 RA is the class of drugs which targets 6 out of 8 components of DeFronzo’s octet. They have proven efficacy in reducing HbA1c, body weight reduction, have low risk of hypoglycemia and have proven CV benefits and safety. Among the GLP-1RA class, the most potent molecule is semaglutide (Fig. 1b).16
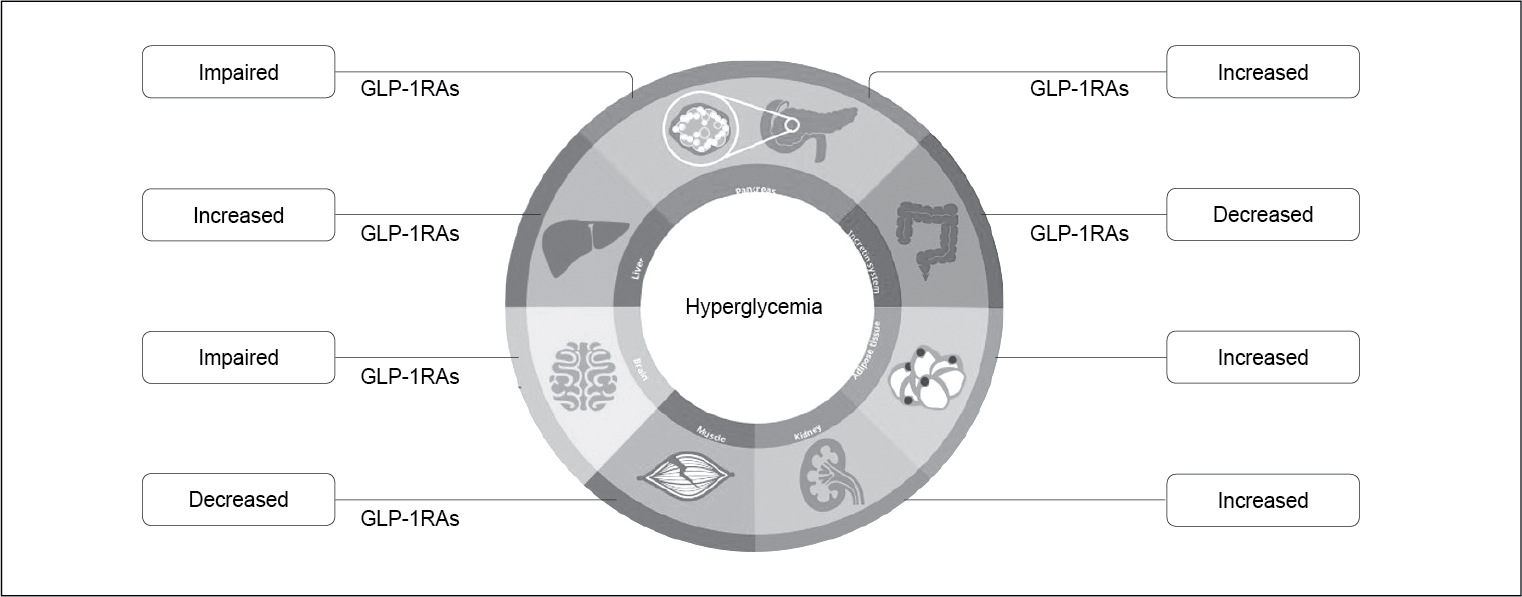
Figure 1b. Role of GLP-1RA in pathophysiology ofT2DM.
It has 94% homology with human GLP-1. It is a human GLP-based peptide with 30 amino acids (7 to 37) (Fig. 2). The following changes were made to achieve the high potency and longer half-life.
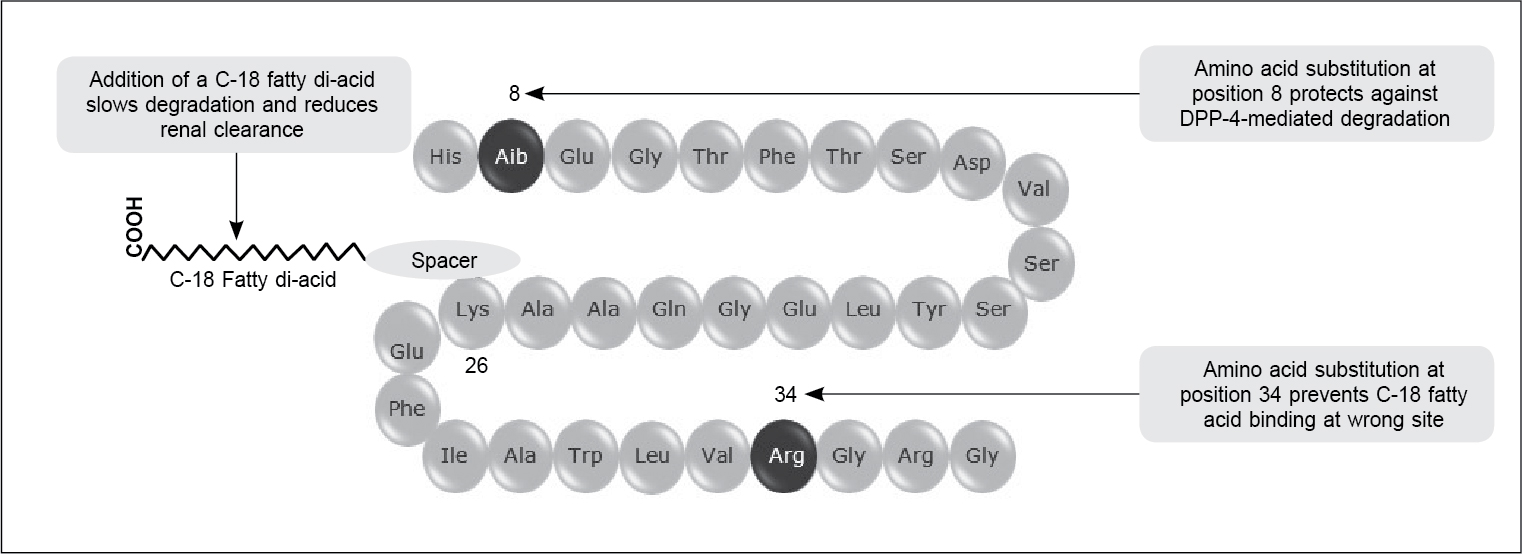
Figure 2. Structure of semaglutide.
First, at 8th position, alanine was substituted with α-aminobutyric acid to prevent degradation with DPP-4 enzyme. At 26th position, a C-18 long di-fatty acid was attached to lysine with a spacer to increase albumin binding and reduce renal clearance, and lastly at 34th position, lysine has been substituted by arginine to prevent C-18 fatty acid binding at wrong site. These changes have increased the half-life of semaglutide to 160 hours from 1.5 to 2 minutes.17
The semaglutide is a multidimensional optimized molecule, it is DPP-4–stable, it has 160 hours half-life, specific affinity towards albumin binding, small molecular weight, and is hydrophilic in nature. These features made it a suitable molecule for oral preparation.
Sodium N-(8-[2-hydroxybenzoyl]amino)caprylate (SNAC) (Fig. 3) is a small fatty acid which was first used in the USA to increase the absorption of vitamin B12. It is a food grade FDA-approved molecule developed by Emisphere Technologies, Inc.
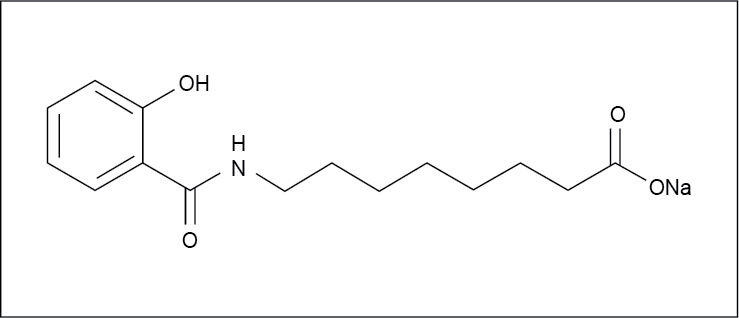
Figure 3. Structure of SNAC.
SNAC acts as an absorption enhancer which has been co-formulated with semaglutide for oral formulation. SNAC enhances semaglutide absorption in the gastric epithelium by increasing the local pH, and increasing cell membrane permeability. This facilitates local transcellular absorption of the drug in stomach. This has increased the bioavailability of semaglutide from 0.01% to 1%.17-20 SNAC is metabolized in the liver by beta glucuronidation and eliminated in kidney. This co-formulation is the world’s first peptide in a pill (Fig. 4).
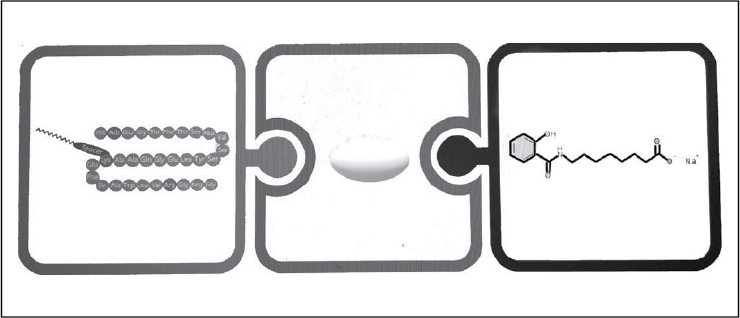
Figure 4. Co-formulation of semaglutide with SNAC.
PHARMACOLOGY OF ORAL SEMAGLUTIDE21-26
Pharmacodynamics
- GLP-1RAs are incretin-based therapies. Oral semaglutide exerts its action via GLP-1 receptors. It induces glucose-dependant insulin release and glucagon suppression after food intake. It also prevents beta-cell apoptosis, and increases primary and secondary release of insulin. It also delays gastric emptying. Since the release of insulin is glucose-dependent, the risk of hypoglycemia is minimal.
- It regulates blood sugar levels and reduces HbA1c. Oral semaglutide also exhibits many pleiotropic effects – weight reduction, reduced energy intake, reduce hunger and increasing satiety, lipid-lowering action, anti-atherosclerotic action and anti-inflammatory action.
Pharmacokinetics
- Oral semaglutide is absorbed in stomach transcellularly with the help of SNAC; it is degraded by DPP-4 enzyme and neutral endopeptidase; it is eliminated in feces and urine. SNAC is metabolized in liver by beta-glucuronidation and eliminated in kidney.
Drug interaction
- Co-administration of placebo with oral semaglutide reduced its area under the curve (AUC) by 34% and maximum concentration (Cmax) by 32%. There was an increase in AUC by 33% when levothyroxine was co-administered with oral semaglutide at steady state, however, the Cmax did not change.
Dosing
- Oral semaglutide is available in three dose forms – 3 mg, 7 mg and 14 mg. It should be started with 3 mg once daily dose after overnight fasting with up to 120 mL (half a glass) of water, followed by post administration fasting for 30 minutes.
- The escalation from 3 to 7 mg should be done after 4 weeks and if further HbA1c control is required, the dose can be further escalated to 14 mg after 4 weeks of initiating 7 mg dose.
WHAT TRIALS REFLECT26-35
The phase 3 trial for oral semaglutide is called PIONEER Program (Peptide InnOvatioN for Early diabEtes tReatment) (Fig. 5). There were 10 trials in this program wherein semaglutide has been compared with placebo as monotherapy (PIONEER 1), with other oral antidiabetic drugs (OADs) (PIONEER 2, 3, 4 and 7), in special population like CVOT trials (PIONEER 6), renal impairment patients (PIONEER 5), as an add-on therapy to insulin (PIONEER 8) and two trials in Japanese population (PIONEER 9 and 10).
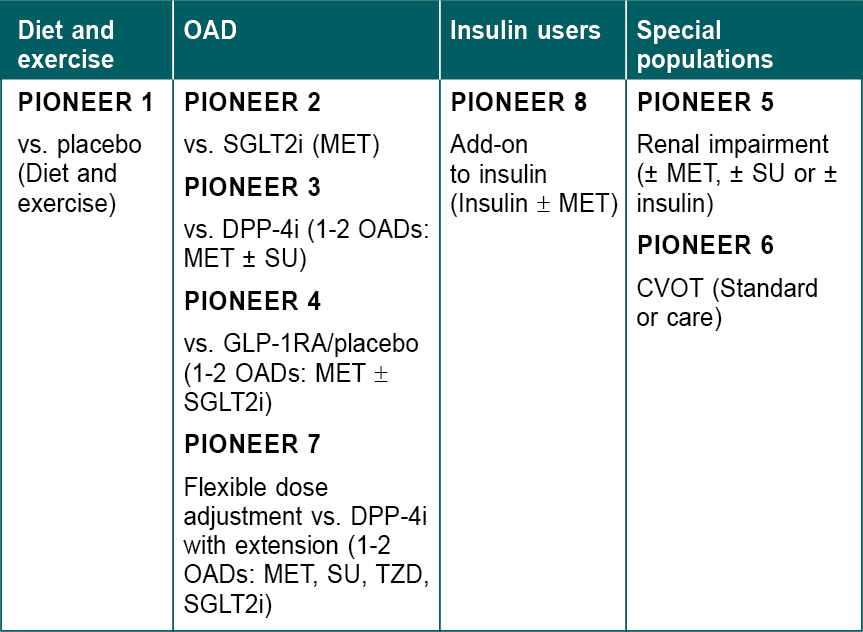
Figure 5. Overview of PIONEER Program.
SGLT2i = Sodium-glucose cotransporter-2 inhibitor; MET = Metformin; DPP-4i = Dipeptidyl peptidase 4 inhibitor; OAD = Oral antidiabetic drug; SU = Sulfonylurea; GLP-1RA = Glucagon-like peptide-1 receptor agonist; TZD = Thiazolidinedione; CVOT = Cardiovascular outcome trial.
The population included were people aged ≥18 years, subjects diagnosed with type 2 diabetes at least 90 days prior to screening, HbA1c 7% to 10.5%. Stable treatment with background medication was allowed. Patients with proliferative retinopathy with acute treatment, multiple endocrine neoplastic syndrome, medullary thyroid carcinoma, history of acute and chronic pancreatitis, history of major stomach surgery, major CV event within 180 days, history of malignant neoplasm in last 5 years, were excluded from the study. The primary endpoint was change in HbA1c from baseline (PIONEER 1-5 and 8), time to first 3P major adverse cardiovascular event or MACE (PIONEER 6), and proportion of patients achieving HbA1c <7.0% (PIONEER 7). Secondary confirmatory endpoints were change in body weight from baseline (PIONEER 1-5, 7-8) and time of first expanded composite CV endpoint (PIONEER 6). Summary of PIONEER Program has been mentioned in Table 1.
|
Table 1. The Pioneer Program
|
|
Trial (N randomized)
|
Background regimen/trial duration/time of primary endpoint
|
Mean baseline characteristics
|
Treatment arms
|
Reduction in HbA1c (%)
|
Change in body weight (Kg)
|
HbA1c <7.0% (%)
|
Body weight loss ≥5% (%)
|
Nausea
|
|
PIONEER 1 (N = 703)
|
Diet and exercise/26 weeks/week 26
|
Age: 55 years
HbA1c: 8.0%
Diabetes duration: 3.5 years
Body weight: 88.1 kg
BMI: 31.8 kg/m2
eGFR: 98 mL/min/1.73 m2
|
Oral semaglutide 3 mg (n = 175)
Oral semaglutide 7 mg (n = 175)
Oral semaglutide 14 mg (n = 175)
Placebo (n = 178)
|
0.8
1.3
1.5
0.1
|
1.70
2.50
4.10
1.50
|
59
72
80
34
|
42
16
|
8
5
16
6
|
|
PIONEER 2 (N = 822)
|
MET/52 weeks/week 26
|
Age: 58 years
HbA1c: 8.1%
Diabetes duration: 7.4 years
Body weight: 91.6 kg
BMI: 32.8 kg/m2
eGFR: 95 mL/min/1.73 m2
|
Oral semaglutide 14 mg (n = 411)
Empagliflozin 25 mg (n = 410)
|
1.3
0.8
|
4.70
3.80
|
72
47
|
47
42
|
20
3
|
|
PIONEER 3 (N = 1,864)
|
MET ± SU/78 weeks/week 26
|
Age: 58 years
HbA1c: 8.3%
Diabetes duration: 8.6 years
Body weight: 91.2 kg
BMI: 32.5 kg/m2
eGFR: 95-96 mL/min/1.73 m2
|
Oral semaglutide 3 mg (n = 466)
Oral semaglutide 7 mg (n = 465)
Oral semaglutide 14 mg (n = 465)
Sitagliptin 100 mg (n = 467)
|
0.3
0.7
1.1
0.4
|
1.90
2.70
3.50
1.10
|
33
50
52
39
|
35
14
|
7
13
15
7
|
|
PIONEER 4 (N = 711)
|
Stable dose of metformin ± SGLT2i for ≥90 days
|
Age: 56 years
HbA1c: 8.0%
Diabetes duration: 7.6 years
Body weight: 94 kg
BMI: 33 kg/m2
eGFR: 95-96 mL/min/1.73 m2
|
Oral semaglutide 14 mg (n = 285)
Liraglutide 1.8 mg s/c (n = 284)
Placebo (n = 142)
|
1.3
1.1
0.1
|
5.00
3.10
1.10
|
69
63
18
|
50
26
12
|
20
18
7
|
|
PIONEER 5 (N = 324)
|
Met ± SU, SU alone or insulin ± met ≥90 days
|
Age: 70 years
HbA1c: 8.0%
Diabetes duration: 14 years
Body weight: 91 kg
BMI: 33 kg/m2
eGFR: 30-59 mL/min/1.73 m2
|
Oral semaglutide 14 mg (n = 163)
Placebo (n = 161)
|
1.1
0.1
|
3.70
1.10
|
64
21
|
40
10
|
19
7.5
|
|
PIONEER 7 (N = 504)
|
1-2 of: MET, SU, TZD, SGLT2i/52 weeks/week 52
|
Age: 57 years
HbA1c: 8.3%
Diabetes duration: 8.8 years
Body weight: 88.6 kg
BMI: 31.5 kg/m2
eGFR: 95-97 mL/min/1.73 m2
|
Oral semaglutide (flexible 3, 7, or 14 mg) (n = 253)
Sitagliptin 100 mg (n = 251)
|
1.4
0.7
|
2.90
0.80
|
63
28
|
|
20.9
2.4
|
|
PIONEER 8 (N = 731)
|
Metformin or no metformin; basal, basal-bolus or premixed insulin/52 weeks/26 weeks
|
Age: 61 years
HbA1c: 8.2%
Diabetes duration: 15 years
Body weight: 85.9 kg
BMI: 31 kg/m2
eGFR: 92 mL/min per 1.73 m2
|
Oral semaglutide 3 mg (n = 184)
Oral semaglutide 7 mg (n = 182)
Oral semaglutide 14 mg (n = 181)
Placebo (n = 184)
|
0.6
1
1.4
0
|
1.30
3.00
4.10
0.6 (gained)
|
36
47
64
10
|
12
31
41
2.5
|
11
17
23
7
|
HbA1c = Glycated hemoglobin; BMI = Body mass index; eGFR: Estimated glomerular filtration rate; MET = Metformin; SU = Sulfonylurea; TZD = Thiazolidinedione; SGLT2i =
Sodium-glucose cotransporter-2 inhibitor.
In the CVOT trial (PIONEER 6), the population included was at least 50 years of age with one CV event in the past or at least 60 years with one established CV risk factor. There were two arms – one placebo and other 14 mg oral semaglutide. There were a total of 3,183 patients, randomized 1:1 into the two arms. Primary endpoint was time to first occurrence of 3P MACE. It was an event-based study to prove noninferiority of oral semaglutide. There was 21% relative risk reduction (RRR) in 3P MACE, 51% RRR in CV mortality and 49% RRR in all-cause mortality.
Safety
Across the trial, up to 80% to 85% of subjects tolerated the treatment with the highest dose of oral semaglutide, i.e., 14 mg. The most common system involved was the gastrointestinal tract, and the most common side effect was mild-to-moderate nausea, which was transient in nature.
To tackle nausea, an apt counseling from the healthcare professionals will help. Inform the patients that the nausea can be transient, advise them to have small frequent meals, avoid fried food, take large quantity fibrous food in a single meal, avoid alcohol and cigarettes and drink cold water and to stop eating at first sign of fullness. In case of severe symptoms, they must report to the treating physician.
Patient management strategy for nausea is pause-play-escalate. Stop the drug if a patient in having severe nausea; once it settles, restart the treatment. If the gap is more than 21 days, start with 3 mg dose.
USE OF PEPTIDE IN A PILL – INDIAN PERSPECTIVE
Where can We Place Oral Semaglutide – First-line, Second-line and Third-line
According to the European Society of Cardiology guidelines for diabetes management, GLP-1RA should be started as monotherapy among those drug naïve patients who have established CV risk factor or CV event in the past.36
According to the American Diabetes Association (ADA) guideline, in drug naïve patients after lifestyle modification and diet management, GLP-RA should be added to metformin in the presence of CV risk factors or established ASCVD.
GLP-1RAs are also recommended if metformin treatment fails as second-line drug; it can also be added as an add-on therapy to insulin.11
Oral semaglutide was found to be superior to empaglifozin, sitagliptin and liraglutide in reducing HbA1c and weight at the end of the trial. It is cardio-safe and well-tolerated. Its efficacy is established early and late in the treatment journey, irrespective of renal and hepatic impairment and no dose adjustment is needed in the elderly.
India is the sixth country to get approval for its commercial use. It is indicated for the management of type 2 diabetes mellitus as monotherapy when metformin is contraindicated or not tolerated, and as a combination therapy with other OADs, in special populations like renal and hepatic impairment patients.
CONCLUSION
Oral semaglutide, the first peptide in a pill is the newest innovation in the managemet of diabetes. GLP-1RA as a class are now recommended to be used as first-line management in drug naïve patients with established CV risk factors, as second-line therapy when metformin fails or is not tolerated and also as third-line when second-line management fails. Oral semaglutide has shown promising data that can help occupy a prominent position in future guidelines.
REFERENCES
- Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119.
- Yesudian CA, Grepstad M, Visintin E, Ferrario A. The economic burden of diabetes in India: a review of the literature. Global Health. 2014;10:80.
- Oberoi S, Kansra P. Economic menace of diabetes in India: a systematic review. Int J Diabetes Dev Ctries. 2020;40(4):464-75.
- Unnikrishnan R, Anjana RM, Deepa M, Pradeepa R, Joshi SR, Bhansali A, et al; ICMR–INDIAB Collaborative Study Group. Glycemic control among individuals with self-reported diabetes in India—The ICMR–INDIAB Study. Diabetes Technol Ther. 2014;16(9):596-603.
- India diabetes report 2000 — 2045. [cited 2021 Dec 30]. Available from: https://www.diabetesatlas.org/data/en/country/93/in.html
- Poudel P, Griffiths R, Wong VW, Arora A, Flack JR, Khoo CL, et al. Oral health knowledge, attitudes and care practices of people with diabetes: a systematic review. BMC Public Health. 2018;18(1):577.
- Srinivas G, Suresh E, Jagadeesan M, Amalraj E, Datta M. Treatment-seeking behavior and compliance of diabetic patients in a rural area of south India. Ann N Y Acad Sci. 2002;958:420-4.
- Kakumani KV, Waingankar P. Assessment of compliance to treatment of diabetes and hypertension amongst previously diagnosed patients from rural community of Raigad district of Maharashtra. J Assoc Physicians India. 2016;64(12):36-40.
- Nimesh VV, Halder A, Mitra A, Kumar S, Joshi A, Joshi R, et al. Patterns of healthcare seeking behavior among persons with diabetes in Central India: a mixed method study. J Fam Med Prim Care. 2019;8(2):677-83.
- Mentock SM, Ng VY, Narayana R, Ullal H, Kumari S, Badiger S, et al. Treatment-seeking behavior and obstacles to treatment compliance in diabetic patients in Mangaluru, India. Diabetes Metab Syndr Clin Res Rev. 2017;11:S617-22.
- American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes—2020. Diabetes Care. 2020;43(Suppl 1):S98-110.
- Mosenzon O, Alguwaihes A, Leon JLA, Bayram F, Darmon P, Davis TME, et al. CAPTURE: a multinational, cross-sectional study of cardiovascular disease prevalence in adults with type 2 diabetes across 13 countries. Cardiovasc Diabetol. 2021;20(1):154.
- Sohal T, Sohal P, King-Shier KM, Khan NA. Barriers and facilitators for type-2 diabetes management in South Asians: a systematic review. PLoS One. 2015;10(9):e0136202.
- Polonsky WH, Henry RR. Poor medication adherence in type 2 diabetes: recognizing the scope of the problem and its key contributors. Patient Prefer Adherence. 2016;10:1299-307.
- Defronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes. 2009;58(4):773-95.
- Kalra S, Sahay R. A review on semaglutide: an oral glucagon-like peptide 1 receptor agonist in management of type 2 diabetes mellitus. Diabetes Ther. 2020;11(9):1965-82.
- Knudsen LB, Lau J. The discovery and development of liraglutide and semaglutide. Front Endocrinol (Lausanne). 2019;10:155.
- Hughes S, Neumiller JJ. Oral semaglutide. Clin Diabetes. 2020;38(1):109-11.
- Neumiller JJ. Incretin-based therapies. Med Clin North Am. 2015;99(1):107-29.
- Buckley ST, Bækdal TA, Vegge A, Maarbjerg SJ, Pyke C, Ahnfelt-Rønne J, et al. Transcellular stomach absorption of a derivatized glucagon-like peptide-1 receptor agonist. Sci Transl Med. 2018;10(467):eaar7047.
- Mosenzon O, Miller EM, Warren ML. Oral semaglutide in patients with type 2 diabetes and cardiovascular disease, renal impairment, or other comorbidities, and in older patients. 2020;132(sup2):37-47.
- Sofogianni A, Tziomalos K. Oral semaglutide, a new option in the management of type 2 diabetes mellitus: a narrative review. Adv Ther. 2020;37(10):4165-74.
- Pearson S, Kietsiriroje N, Ajjan RA. Oral semaglutide in the management of type 2 diabetes: a report on the evidence to date. Diabetes Metab Syndr Obes. 2019;12:2515-29.
- Anderson SL, Beutel TR, Trujillo JM. Oral semaglutide in type 2 diabetes. J Diabetes Complications. 2020;34(4):107520.
- Avgerinos I, Michailidis T, Liakos A, Karagiannis T, Matthews DR, Tsapas A, et al. Oral semaglutide for type 2 diabetes: a systematic review and meta-analysis. Diabetes Obes Metab. 2020;22(3):335-45.
- Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, Franco DR, et al; PIONEER 6 Investigators. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019;381(9):841-51.
- Aroda VR, Rosenstock J, Terauchi Y, Altuntas Y, Lalic NM, Villegas EC, et al; PIONEER 1 Investigators. PIONEER 1: randomized clinical trial of the efficacy and safety of oral semaglutide monotherapy in comparison with placebo in patients with type 2 diabetes. Diabetes Care. 2019;42(9):1724-32.
- Rodbard HW, Rosenstock J, Canani LH, Deerochanawong C, Gumprecht J, Lindberg SØ, et al; PIONEER 2 Investigators. Oral semaglutide versus empagliflozin in patients with type 2 diabetes uncontrolled on metformin: the PIONEER 2 trial. Diabetes Care. 2019;42(12):2272-81.
- Rosenstock J, Allison D, Birkenfeld AL, Blicher TM, Deenadayalan S, Jacobsen JB, et al; PIONEER 3 Investigators. Effect of additional oral semaglutide vs sitagliptin on glycated hemoglobin in adults with type 2 diabetes uncontrolled with metformin alone or with sulfonylurea: the PIONEER 3 randomized clinical trial. JAMA. 2019;321(15):1466-80.
- Pratley R, Amod A, Hoff ST, Kadowaki T, Lingvay I, Nauck M, et al; PIONEER 4 Investigators. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomised, double-blind, phase 3a trial. Lancet. 2019;394(10192):39-50.
- Mosenzon O, Blicher TM, Rosenlund S, Eriksson JW, Heller S, Hels OH, et al; PIONEER 5 Investigators. Efficacy and safety of oral semaglutide in patients with type 2 diabetes and moderate renal impairment (PIONEER 5): a placebo-controlled, randomised, phase 3a trial. Lancet Diabetes Endocrinol. 2019;7(7):515-27.
- Yamada Y, Katagiri H, Hamamoto Y, Deenadayalan S, Navarria A, Nishijima K, et al; PIONEER 9 Investigators. Dose-response, efficacy, and safety of oral semaglutide monotherapy in Japanese patients with type 2 diabetes (PIONEER 9): a 52-week, phase 2/3a, randomised, controlled trial. Lancet Diabetes Endocrinol. 2020;8(5):377-91.
- Yabe D, Nakamura J, Kaneto H, Deenadayalan S, Navarria A, Gislum M, et al; PIONEER 10 Investigators. Safety and efficacy of oral semaglutide versus dulaglutide in Japanese patients with type 2 diabetes (PIONEER 10): an open-label, randomised, active-controlled, phase 3a trial. Lancet Diabetes Endocrinol. 2020;8(5):392-406.
- Pieber TR, Bode B, Mertens A, Cho YM, Christiansen E, Hertz CL, et al. Efficacy and safety of oral semaglutide with flexible dose adjustment versus sitagliptin in type 2 diabetes (PIONEER 7): a multicentre, open-label, randomised, phase 3a trial. Lancet Diabetes Endocrinol. 2019;7(7):528-39.
- Zinman B, Aroda VR, Buse JB, Cariou B, Harris SB, Hoff ST, et al; PIONEER 8 Investigators. Efficacy, safety, and tolerability of oral semaglutide versus placebo added to insulin with or without metformin in patients with type 2 diabetes: the PIONEER 8 Trial. Diabetes Care. 2019;42(12):2262-71.
- Cosentino F, Grant PJ, Aboyans V, Bailey CJ, Ceriello A, Delgado V, et al; ESC Scientific Document Group. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2020;41(2):255-323.
|