Abstract
While the role of prostaglandin as a trigger in dysmenorrhea is well known, not many are aware that inflammation and nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasome are also implicated in primary dysmenorrhea (PD). Inhibition of NLRP3 inflammasome and inflammation pathways is an important approach to treating dysmenorrhea and also the symptoms of premenstrual syndrome. Mefenamic acid is an effective and selective inhibitor of the NLRP3 inflammasome, which can be considered the most important option for PD treatment owing to its action via various pathways. In this article, the authors have reviewed the role of inflammation and NLRP3 inflammasome in causing PD, how inhibitors of NLRP3 inflammasome can treat dysmenorrhea and the mechanism of action of mefenamic acid as NLRP3 inflammasome inhibitor and its role in PD.
Keywords: NLRP3 inflammasome, primary dysmenorrhea, inflammation, mefenamic acid
Primary dysmenorrhea (PD) is a common gynecological problem among young women of reproductive age group. It constitutes functional dysmenorrhea without a specific gynecological pathological origin and has a severe effect on all aspects of the lives, work and study of female patients. Currently, prostaglandin secretion is proposed to be the trigger for PD.1 It has also been reported that inflammation has a potent effect on the pathological process of PD. As a core part of inflammation, the nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasome is involved in several acute and chronic diseases including dysmenorrhea.2
In this article, the role of NLRP3 inflammasome in causing primary dysmenorrheic pain and its inhibition as a potential target for treating dysmenorrhea is explored. Mefenamic acid is known to have the NLRP3 inflammasome inhibitory action in addition to its prostaglandin inhibitory action and this may help further augment its anti-inflammatory action.
THE NLRP3 INFLAMMASOME
Excess of inflammation is known to worsen many diseases and is often linked to the aberrant activation of the NLRP3 inflammasome. NLRP3 is usually found in cells of the innate immune system such as macrophages where it responds to danger in the form of pathogen or damage-associated molecular patterns (PAMPs or DAMPs), respectively. In response to external unfavorable stimulus, NLRP3 interacts with an adaptor protein leading to an activating platform for the protease caspase-1. Caspase-1 cleaves pro-inflammatory cytokine precursors pro-interleukin (IL)-18 and pro-IL-1β into active forms secreted from the cell. It also cleaves the pore-forming protein gasdermin D which eventually forms membrane pores causing pyroptotic cell death.3
MENSTRUATION AS AN INFLAMMATORY EVENT AND NLRP3 INFLAMMASOME INVOLVEMENT
Menstruation is a self-inflammatory process leading to the shedding of the functional layer of the endometrium at the end of a nonconception cycle. Several significant changes in endometrial function occur through the entire menstrual cycle including spontaneous hormone-mediated terminal differentiation of endometrial stromal cells and decidualization. The shedding of these cells is critical for the preparation of new conception cycle. Hence, these decidualized cells play a key role in detecting hormone withdrawal at the end of a non-conception cycle, intervening local inflammation within the endometrium.4 Figure 1 shows the pathways involvig NLRP3 inflammasome leading to PD.
Figure 1. Pathways involving NLRP3 inflammasome leading to PD.
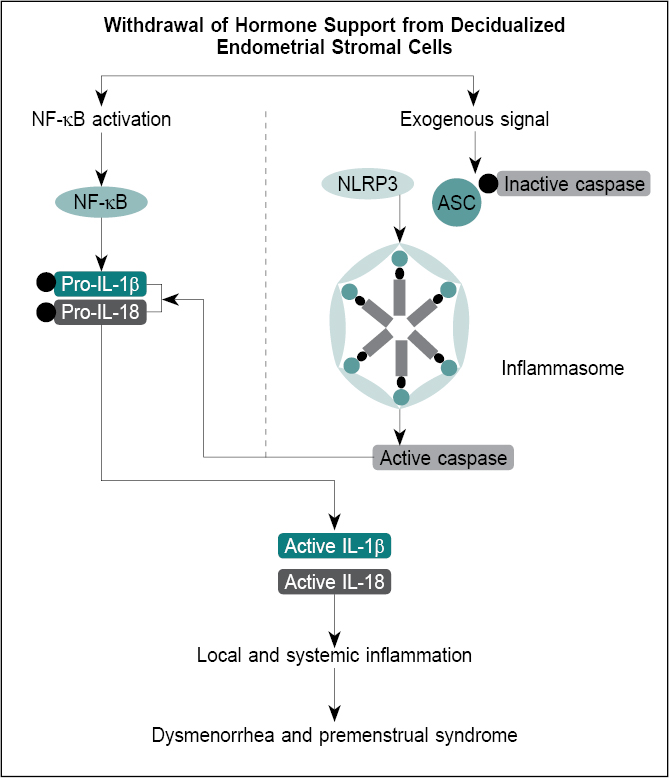
Menstruation event is related to the activation of NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells), local release of chemokines and cytokines and inflammatory leukocyte influx. Systemically, chemokines and cytokines fluctuate across the menstrual cycle.4
It has been noted that NLRP3 inflammasome components immunolocalize to decidualized endometrial stromal cells immediately before menstruation. In vitro model of menstruation has also given evidence of the activation of NLRP3 inflammasome.4
Besides, there is evidence suggesting that the NLRP3 inflammasome plays a regulatory role in the expression of cyclooxygenase-2 (COX-2) via the NF-κB pathway. Studies have shown that NLRP3 inflammasome can be activated and expressed in PD.2
In addition, serum C-reactive protein (CRP) is an important marker of inflammation. With the resolution of inflammation or tissue damage, the CRP concentration reduces. It has been seen that IL-6 acts as the primary inducer of CRP gene expression, with IL-1β further augmenting the effect.5
Prostaglandins are a major factor causing the increased abnormal uterine activity. During menstruation, prostaglandin F2α (PGF2α) and prostaglandin E2 (PGE2) stimulate prolonged uterine contractions that reduce blood flow resulting in uterine ischemia.1 Additionally, prostaglandins also modulate various steps of inflammation thereby coordinating the entire process in both pro-inflammatory and anti-inflammatory directions. PGE2 and its receptors regulate the production of inflammatory cytokines such as IL-1β, IL-6 and monocyte chemotactic protein-1.6
Prior to menstruation, endometrial tissue acquires the features of inflammation, red and edematous. Endometrial edema results from the high localized production of chemokines IL-8, pro-inflammatory cytokines such as IL-1, IL-6, tumor necrosis factor (TNF)-α and leukocyte inflow. These pro-inflammatory cytokines are responsible for upregulating inflammatory responses. These mediators also trigger the synthesis or release of prostaglandins causing excessive contraction of the uterine muscle, which leads to ischemic pain of PD. It has been reported that the level of IL-6 and TNF-α is higher in the plasma of women with dysmenorrhea compared to those without menstrual disorders.7
INHIBITOR OF THE NLRP3 INFLAMMASOME
As we have seen that inflammation underpins menstrual disorders such as dysmenorrhea; thus, targeting inflammatory pathways is a potential approach to ameliorate or reduce menstrual associated pain and general consequences of inflammation including the cluster of symptoms referred to as ‘premenstrual syndrome’.4
In animal model study, that a molecule inhibiting NLRP3 activation led to a marked alleviation of the pain and pathological damage in PD. It was seen that the molecule inhibited the activation of NLRP3 inflammasome in uterine tissues of mice with PD and also inhibited production of prostaglandin in uterine tissues of mice with PD.2
Mefenamic Acid as NRLP3 Inflammasome Inhibitors and its Role in PD
Current evidence suggests that mefenamic acid, a fenamate, is an effective and selective inhibitor of the NLRP3 inflammasome by inhibiting the volume-regulated anion channel, an action independent of the COX inhibition.8 Besides, the transient receptor potential melastatin 2 (TRPM2) is a plasma membrane cation channel through which the influx of extracellular calcium occurs. Activation of TRPM2 channel increases the activity of NLRP3 and IL secretions, aggravating the inflammation and cytokine release. Mefenamic acid inhibits the TRPM2 channels and reduces the calcium influx, which in turn blocks the NLRP3 inflammasome.9
It is known that NLRP3 mediates IL-1β secretion responsible for several inflammatory responses. The NLRP3 inhibitory action of mefenamic acid thus attenuates the pro-inflammatory cytokine (IL-1β) levels.9
The mainstay of treatment for dysmenorrhea, mefenamic acid offers complete relief of all the symptoms of dysmenorrhea in patients by exhibiting its actions via various pathways including COX-2 inhibition, bradykinin antagonism, inhibition of uterine contractility triggered by, prostaglandin-endoperoxide analogs, reduction of the high uterine resting tone and the frequency of contractions.1,10 With its additional activity in inhibiting NLRP3, mefenamic acid is an ideal treatment in conditions with dysregulated NLRP3 inflammasome such as PD.
Mefenamic acid provides significant protection against increased levels of TNF-α and IL-1β. The use of mefenamic acid has previously been shown to reduce the CRP levels reflecting its significant anti-inflammatory activity.5
CONCLUSION
It is a well-established fact that menstruation is a self-inflammatory process. Apart from prostaglandins and other mediators like bradykinin, histamine and acetylcholine, etc. NLRP3 activation leads to expression of pro-inflammatory cytokines like IL-1β, which play prominent role in the underlying inflammatory process in dysmenorrhea. The inhibition of NLRP3 will help in easing the excess inflammation in PD and associated premenstrual symptoms. Mefenamic acid is an effective and selective inhibitor of NLRP3 inflammasome that acts by blocking the volume-regulated anion channel and TRPM2, a pathway acting independently of COX inhibition. By inhibiting NLRP3 inflammasome, mefenamic acid minimizes the formation of pro-inflammatory cytokine, IL-1β levels. Well-established safety and efficacy, and its action through multiple pathways including COX inhibition, bradykinin antagonism, inhibition of uterine contractility, reduction of CRP levels and NLRP3 inhibition makes mefenamic acid an ideal option to treat dysmenorrhea.
REFERENCES
- Rao KA, Chandra S, Kasture PN. Role of prostaglandins in pathogenesis of dysmenorrhea and place of mefenamic acid and dicyclomine in its management. IJCP. 2022;32(10):27-34.
- Tang B, Liu D, Chen L, Liu Y. NLRP3 inflammasome inhibitor MCC950 attenuates primary dysmenorrhea in mice via the NF-κB/COX-2/PG pathway. J Inflamm (Lond). 2020;17:22.
- Swanton T, Beswick JA, Hammadi H, Morris L, Williams D, de Cesco S, et al. Selective inhibition of the K+efflux sensitive NLRP3 pathway by Cl- channel modulation. Chem Sci. 2020;11(43):11720-8.
- Azlan A, Salamonsen LA, Hutchison J, Evans J. Endometrial inflammasome activation accompanies menstruation and may have implications for systemic inflammatory events of the menstrual cycle. Hum Reprod. 2020;35(6):1363-76.
- Aggarwal KK. Mefenamic acid as steroid-sparing anti-inflammatory drug during viral phase of COVID-19: 5 case reports. IJCP. 2021;31(8):759-63.
- Ricciotti E, FitzGerald GA. Prostaglandins and inflammation. Arterioscler Thromb Vasc Biol. 2011;31(5):986-1000.
- Barcikowska Z, Rajkowska-Labon E, Grzybowska ME, Hansdorfer-Korzon R, Zorena K. Inflammatory markers in dysmenorrhea and therapeutic options. Int J Environ Res Public Health. 2020;17(4):1191.
- Daniels MJ, Rivers-Auty J, Schilling T, Spencer NG, Watremez W, Fasolino V, et al. Fenamate NSAIDs inhibit the NLRP3 inflammasome and protect against Alzheimer’s disease in rodent models. Nat Commun. 2016;7:12504.
- Kasture P, Mehta K, Gowda A. Inflammasome, inflammation, infection and mefenamic acid. J Assoc Physicians India. 2022;70(3):11-2.
- Kintis GA, Coutifaris B. Treatment of primary dysmenorrhea with mefenamic acid. Int J Gynaecol Obstet. 1980;18(3):172-5.