Abstract
Background: Tetracyclines, in particular doxycycline, are recommended for the treatment of patients with acute undifferentiated febrile illness (AUFI); however, real-world studies are scarce. Methods: This retrospective, multicenter, observational study reviewed electronic medical records (April 2018 to March 2021) of adult patients (outpatient and inpatient departments [OPD and IPD]) with AUFI, treated with doxycycline monotherapy (doxycycline group) or doxycycline in combination with other antimicrobials (combination therapy group), from 7 tertiary hospitals and clinics in India. Results: Overall, 473 patients were included; 73.8% and 26.2% patients were prescribed doxycycline alone or in combination with other antimicrobials, respectively. Defervescence was achieved in 65.6% and 57.3% patients, respectively at the second (8-14 days) follow-up visit. Clinical cure rate for symptomatic resolution varied between 89.6% and 100% in OPD settings. Time taken from treatment initiation to defervescence was 3.51 ± 3.16 days for the doxycycline group and 3.46 ± 3.07 days for the combination therapy group. Both groups showed improvements in body temperature in OPD settings (84.2% and 84.5%) as well as IPD settings (97.4% and 94.1%). Adverse events in OPD patients in both groups were nausea (7.8% and 8.7%), anorexia (1.6% and 33.0%) and dyspepsia (1.6% and 67.9%). Conclusion: Doxycycline appears to be a promising candidate for treating patients with AUFI due to its demonstrated real-world effectiveness and safety profile.
Keywords: Acute undifferentiated febrile illness, doxycycline, real-world, antimicrobials
Infectious diseases are the leading causes of morbidity and mortality in the tropical Indian subcontinent.1 Acute undifferentiated febrile illness (AUFI) is common in developing countries, like India.2 It is one of the most frequently encountered presentations in clinical practice, mostly in outpatient departments (OPDs) and emergency care.3 AUFI generally lasts less than 21 days without any evidence of organ or system-specific etiology and is usually characterized by nonspecific pyrexia.4 Initially, the clinical features include fever, headache, chills, myalgia and fatigue, but later, specific organs might be involved. AUFI can vary from a mild and self-limiting illness to progressive and life-threatening for a confirmed diagnosis, investigations are utmost essential.5
The etiology of AUFI remains largely unknown; although the most common causes include malaria, dengue, Chikungunya, Zika, Yellow fever, Japanese encephalitis, enteric fever, leptospirosis and scrub typhus.6 The non-specific and overlapping clinical features pose a challenge to the treating clinicians. Diagnosis is ascertained based on the clinical presentation and specific diagnostic tests. But owing to the limited diagnostic facilities and lack of comprehensive local guideline recommendations, delay in drug therapy initiation can increase the severity, complications and mortality. Therefore, empirical treatment with antibiotics is initiated along with antipyretics and other supportive treatment.7 An empirical approach is justified in AUFI owing to the similarity in clinical presentations and the unavailability of accurate diagnostic testing, particularly during the early phase of the illness.7
Doxycycline is a cost-effective antimicrobial agent with a wide spectrum of activity, and its re-emergence can prove to be an asset for a developing country like India, notably with the current trends of resistance to several antimicrobials.8 Doxycycline presents a lower risk of Clostridium difficile-associated diarrhea than other antibiotics as doxycycline preparations are mostly fortified with lactobacillus.9 Doxycycline also reduces the incidence of multiple organ dysfunction syndrome in patients with scrub typhus.10 Doxycycline is a bacteriostatic agent with activity against both Gram-positive and Gram-negative bacteria.11 The effect of doxycycline is primarily mediated by its immunomodulatory action exerted by reducing proinflammatory cytokines and inhibiting virus replication.12,13 As per the Department of Health Research (DHR) and Indian Council of Medical Research (ICMR), doxycycline is recommended for the empirical treatment of acute undifferentiated fever presented with leptospirosis, rickettsia infections, pneumonia, cholera and scrub typhus.14,15 Thus, doxycycline appears to be a potential treatment choice for managing patients with AUFI.
Investigations pertaining to empirical therapy is needed due to lack of comprehensive surveillance and delay in confirmatory laboratory diagnosis in resource-constraint countries like India.4,16 Therefore, a real-world evidence-based study was conducted based on the review of electronic medical records (EMRs) to evaluate the effectiveness of doxycycline as empirical therapy for treating patients with AUFI in routine clinical practice in India.
Methods
The study was designed as a multicenter, retrospective and observational study. Routine clinical data was sourced from the EMRs of OPD and IPD patients with AUFI from 7 tertiary hospitals and clinics across 2 states (Assam and Maharashtra) of India at baseline and at the first (1-7 days) and second follow-up visits (8-14 days) from April 2018 to March 2021. The primary outcome measure was the real-world effectiveness of empirical treatment with either doxycycline monotherapy or combination therapy in patients with AUFI based on the clinical cure rate determined by the proportion of patients achieving defervescence, resolution of symptoms and improved hemodynamic parameters from baseline to follow-up visits. Improvement was considered as normalized body temperature (≤98.6°F), stabilized heart rate (≤100 bpm), systolic (≤120 mmHg) and diastolic (≤80 mmHg) blood pressure. The secondary outcomes were any change from baseline in the laboratory investigations (peripheral blood smear, complete blood count, rapid diagnostic tests, dengue, scrub typhus, enteric fever, liver function tests and renal profile). Safety was evaluated based on the adverse events (AEs) reported by the patients and captured by the clinicians.
Data was analyzed using R studio 3.5.3 and Microsoft Excel version 2110. Descriptive statistics were used to summarize the patients’ baseline characteristics. Continuous variables such as age, height, weight, body mass index (BMI), use of antipyretics, time taken for defervescence and laboratory investigations were presented as mean ± standard deviation. Inpatient department (IPD) vs. OPD and baseline vs. first and second follow-up visits comparison was conducted using the t-test or Chi-square test, wherever applicable. Statistical significance was considered at p < 0.05. The study was conducted as per protocol and principles of Declaration of Helsinki and was approved by the Royal Pune Independent Ethics Committee located at Pune, India (Ethics Approval Number: RPIEC0100521).
Results
Overall, records of 473 patients with AUFI were analyzed in this study, out of which 349 (73.8%) patients were prescribed doxycycline monotherapy (doxycycline group) and 124 (26.2%) patients were prescribed doxycycline in combination with other antimicrobials (combination therapy group) (Fig. 1).
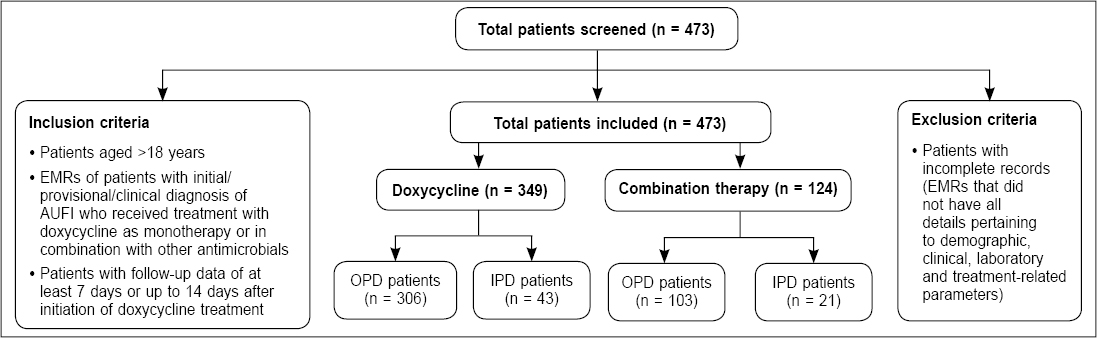
Figure 1. Flowchart of the study population.
IPD: Inpatient department; OPD: Outpatient department.
Treatment
Antimicrobial drugs
In doxycycline group, all the patients in OPD settings, were prescribed oral doxycycline 100 mg (1 patient received 200 mg) for 5 to 14 days (average). IPD patients were prescribed doxycycline 100 mg (oral; n = 28) and 200 mg intravenous (IV; n = 15) for 7 to 10 days. In the combination therapy group, doxycycline was co-prescribed with other antimicrobials, including cephalosporins (cefixime, cefuroxime, cefpodoxime, ceftriaxone), fluoroquinolones (ciprofloxacin and ofloxacin), nitrofurantoin, macrolides (azithromycin and erythromycin), penicillin-like antibiotics (amoxicillin-clavulanic acid), carbapenems (faropenem) and oxazolidinones (linezolid). The oral formulations were administered for 5 to 14 days in OPD settings, while in IPD settings, both oral (12) and IV (9) formulations were prescribed for 7 to 14 days. None of the patients in OPD or IPD settings were switched to another antimicrobial at first and second follow-up visits. No other antimicrobials were added to the treatment regimen, except in one outpatient at the first follow-up visit who was prescribed an additional injection of artesunate 60 mg.
Antipyretics usage
Among patients treated with doxycycline monotherapy, paracetamol was the most common antipyretic prescribed to 300 (86.0%) patients in both settings. Nimesulide was prescribed to only 49 (14.0%) OPD patients. In the combination therapy group, paracetamol was prescribed to 112 (90.3%) patients, followed by nimesulide to 12 (9.7%) patients in OPD.
Concomitant medication
The most commonly prescribed concomitant medications in the doxycycline group were antipyretics, corticosteroid and proton pump inhibitor (PPI) + antiemetic in both OPD and IPD settings. In the combination therapy group, antipyretics, corticosteroid and PPI + prokinetic were more commonly prescribed in OPD settings, while antipyretics, corticosteroid and PPI + antiemetic were prescribed in IPD settings.
Effectiveness Outcomes
Occurrence of defervescence and symptomatic relief
In both doxycycline and combination therapy groups, 65.6% (229/349) and 57.3% (71/124) patients, respectively, achieved defervescence at the second follow-up visits. For OPD patients (n = 409), the proportion of patients showing defervescence in doxycycline group increased from the first follow-up (32.3%) to the second follow-up visits (56.0%), while in the combination therapy group, defervescence occurred in only 13.2% at the first follow-up and 17.4% at the second follow-up visits. In OPD settings, the clinical cure rate in the doxycycline group varied from 89.6% to 100% and 90.3% to 100.0% in the combination therapy group. In both groups, most patients achieved resolution of symptoms at the second follow-up visit.
Time taken for defervescence after treatment initiation
Reduction in body temperature was observed in both doxycycline (100.53 ± 1.44°F to 98.28 ± 0.19°F) and combination therapy groups (100.77 ± 1.59°F to 98.28 ± 0.20°F) from first to the second follow-up visits. Time taken from fever onset to defervescence was 6.69 ± 3.50 days for the doxycycline group and 6.57 ± 3.40 days for the combination therapy group. Time taken from the start of doxycycline treatment to defervescence was 3.51 ± 3.16 days for the doxycycline group and 3.46 ± 3.07 days for the combination therapy group.
Clinical cure rate
In OPD settings, the improvement was 84.2% for the doxycycline group and 84.5% for the combination therapy group. In IPD settings, improvement in the doxycycline and combination therapy groups were 97.4% and 94.1%, respectively (Fig. 2). Both doxycycline and combination therapy groups stabilized the heart rate (≤100 bpm) in OPD (48.2% and 39.5%) and IPD (16.2% and 0.0%) settings. For systolic BP (≤120 mmHg), minor improvements (0.8%) in OPD patients in the doxycycline group and 23.5% improvement in IPD patients in the combination therapy groups were observed. Diastolic BP (≤80 mmHg) was normal in all the patients in both OPD and IPD settings.
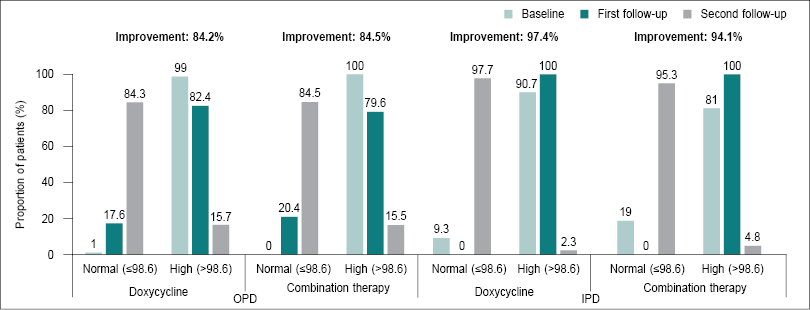
Figure 2. Clinical cure rate of body temperature (n = 473).
Laboratory Investigations of Patients with AUFI
Both doxycycline and combination therapy groups showed significant improvements (p = 0.000) in all the laboratory parameters except aspartate transaminase, direct bilirubin, gamma-glutamyl transferase, serum uric acid, serum albumin and serum protein at the second follow-up visits. In the doxycycline group, 1 (0.2%), 9 (2.6%), 31 (8.9%) and 2 (0.6%) patients tested positive for malaria, dengue, scrub typhus and typhoid, respectively. In the combination therapy group, 4 (3.2%), 7 (5.6%) and 29 (23.4%) patients tested positive for dengue, scrub typhus and typhoid.
Safety Outcomes
Patients in OPD setting, in both groups, reported nausea (7.8% vs. 8.7%), anorexia (1.6% vs. 33.0%) and dyspepsia (1.6% vs. 67.9%). In IPD settings, data regarding adverse events was not available.
Discussion
In a prospective study conducted in Mumbai, most patients were male and below the age of 35 years.17 Our study also had a male predominance and mean age ~45 years. The five major comorbidities in both groups included endocrinological (diabetes mellitus), cardiovascular (hypertension and ischemic heart disease [IHD]), urogenital (benign prostatic hyperplasia [BPH]) and thyroid disorders, similar to a study conducted in South India, which reported diabetes mellitus, essential hypertension, IHD and chronic liver disease as the most common comorbidities in AUFI patients.7
In the present study, the major complaints were fever, malaise, fatigue, headache, cough and myalgia. This was in accordance with a study conducted in the rural areas and suburbs of Maharashtra, wherein all the patients with AUFI complained of fever, in addition to nonspecific (such as headache, myalgia, generalized weakness), respiratory (cough with breathlessness, sore throat), neurological (altered sensorium, seizures, meningitis) and gastrointestinal (vomiting, diarrhea) symptoms.18 Another study in Eastern India reported fever, headache, nausea, vomiting, abdominal pain, cough, myalgia and rash in scrub typhus patients presenting with AUFI.19 With the emergence and re-emergence of pathogens, doxycycline seems to be a potential candidate for the empirical antibiotic of choice in AUFI patients.3,20
Oral doxycycline 100 mg BD was primarily prescribed in patients with AUFI in both OPD and IPD settings, which is in accordance with the 2019 ICMR “Treatment Guidelines for Antimicrobial Use in Common Syndromes”, which recommends empirical treatment with a standard dose of doxycycline 100 mg BD (oral or IV) for patients with acute undifferentiated fever.14 A study of hospitalized patients with febrile illness in South India also used doxycycline 100 mg twice daily for their management.21 Aspirin, paracetamol, nimesulide, ibuprofen, mefenamic acid, etc., are also reportedly used for managing fever in patients with AUFI.22,23
In a retrospective observational study conducted at a tertiary care hospital in Hyderabad, defervescence was achieved in all the scrub typhus patients within 2 days of treatment initiation with doxycycline.24 Overall, the clinical cure rate of defervescence, fatigue, malaise, diarrhea, abdominal pain, vomiting and rashes was found to be higher in both treatment groups in OPD and IPD settings in the present study.
In a multicenter trial of 296 adult patients with AUFI in Thailand, the median time to defervescence with doxycycline was 45 hours and 48 hours for patients with laboratory-confirmed leptospirosis and scrub typhus, respectively.25 A study conducted at a tertiary care hospital in Southern India (n = 645) reported defervescence in all the scrub typhus patients with fever of 1 to 2 weeks (58.6%) within 2 days of initiating doxycycline treatment.24 AUFI may have a similar clinical presentation, but their etiology can vary, due to which it often remains undiagnosed. A cross-sectional, prospective study conducted in Central India reported 18 (6.6%) undiagnosed patients.26 In Asia, the frequencies of undiagnosed cases ranged from 8% to 80%.27 In a study of 1,564 patients conducted in six states of India, the diagnoses were malaria (17%), dengue (16%), scrub typhus (10%), bacteremia (8%), leptospirosis (7%) and chikungunya (6%).28 Other Indian studies too have reported scrub typhus, dengue, malaria, enteric fever, leptospirosis, typhoid as the different etiologies of AUFI.1,2,7
Doxycycline is the treatment of choice in clinically stable patients in co-endemic regions for AUFI, especially in South-East Asia, including India.5 But for severely ill patients, a combination of third-generation cephalosporins and doxycycline is preferred.29 Comorbidities, pregnancy, geriatrics and immunocompromised patients are other groups that benefit from combination therapy.6 Combination therapy is effective in patients with AUFI with complications such as pneumonia or acute respiratory distress syndrome, encephalopathy, malaria and liver involvement.29 In the current study, patients with multiple comorbidities were effectively treated by a combination of doxycycline with other antimicrobials. However, combination therapy facilitates development of multidrug resistance (MDR);30 this can be curtailed if pathogen-specific diagnostics tests are conducted. A study conducted in Pune (India) showed that due to diagnostic uncertainty, all the hospitalized patients received antibiotics, which the physicians readily discontinued once they received the results of positive malaria or dengue tests.31 Thus, doxycycline monotherapy can be considered over the combination therapy to ensure the judicious use of antibiotics, consequently avoiding the development of MDR.
Fewer patients on doxycycline monotherapy in our study reported anorexia and dyspepsia. A study evaluating the effectiveness of doxycycline for the treatment of scrub typhus and leptospirosis also reported nausea and vomiting.25 Increased antibiotic use may cause gut microbiota dysbiosis.32 Therefore, doxycycline monotherapy may avert unwanted gastrointestinal effects in patients with AUFI.
Hitherto, to the best of our knowledge, no studies have examined the effectiveness of combination of doxycycline with other antimicrobials. Our study included adult patients with a broad age range receiving various levels of care and visiting different settings providing greater external validity. However, our study has certain limitations, such as bias associated with a retrospective study design and missing information of some patients at the second follow-up visit. Information regarding many potential pathogens (spotted fever, hantavirus and chikungunya virus) and serological evidence (using indirect immunofluorescence and polymerase chain reaction) was also unavailable.
Conclusion
The management of AUFI warrants accurate diagnosis, for which it is imperative to identify the etiology. In addition to the indispensable need for a broader diagnostic approach, re-purposing of existing drugs is the need of the hour. Based on the findings of the current study, it can be concluded that doxycycline could be an effective approach for the treatment of patients with AUFI having different etiologies owing to its demonstrated real-world effectiveness safety profile, with both oral and injectable dosage forms. Further large-scale prospective studies are warranted to confirm these results.
Acknowledgments: The authors thank Saleha Rehman (Medical Writer), Rupali Jangid (Senior Medical Writer) and Dr Venugopal Madhusudhana (Vice President-Research) from Medical affairs, THB c/o Sekhmet Technologies Pvt Ltd. (Gurugram, Haryana, India) for their support in the manuscript preparation.
Conflicts of Interest: The authors Anup Uttam Petare, Krishna Chaitanya Veligandla, Rahul Rathod, Seema Bhagat and Amey Mane declare that they are an employee at Dr. Reddy’s Laboratories Ltd., Hyderabad, Telangana, India. Sundaram Arulrhaj and Gifty Immanuel declare no conflict of interest for this publication, however, they have received speaker honorarium from Dr. Reddy’s laboratories in the past. Rajiv Saikia declares no conflict of interest.
References
- Mittal G, Ahmad S, Agarwal RK, Dhar M, Mittal M, Sharma S. Aetiologies of acute undifferentiated febrile illness in adult patients - an experience from a tertiary care hospital in Northern India. J Clin Diagn Res. 2015;9(12):DC22-4.
- Prasad D, Kumar S, Deepak RK, Kumar M. Evaluating the etiology and disease specific clinical profiles of acute undifferentiated febrile illness. Int J Sci Res. 2020;9(11):73-5.
- Aggarwal KK. Management of acute febrile illness: doxycycline, the empirical antibiotic of choice. Ind J Clin Pract. 2018;29(6):505-6.
- Bhaskaran D, Chadha SS, Sarin S, Sen R, Arafah S, Dittrich S. Diagnostic tools used in the evaluation of acute febrile illness in South India: a scoping review. BMC Infect Dis. 2019;19(1):970.
- Bhargava A, Ralph R, Chatterjee B, Bottieau E. Assessment and initial management of acute undifferentiated fever in tropical and subtropical regions. BMJ. 2018;363:k4766.
- Joshi S, Immanuel G, Arulrhaj S, Tiwaskar M, Vora A, Samavedam S. Roadmap for the management of acute undifferentiated febrile illness: an expert discussion and review of available guidelines. J Assoc Physicians India. 2021;69(9):11-2.
- Abhilash KPP, Jeevan JA, Mitra S, Paul N, Murugan TP, Rangaraj A, et al. Acute undifferentiated febrile illness in patients presenting to a tertiary care hospital in south india: clinical spectrum and outcome. J Glob Infect Dis. 2016;8(4):147-54.
- Grossman TH. Tetracycline antibiotics and resistance. Cold Spring Harb Perspect Med. 2016;6(4):a025387.
- Doernberg SB, Winston LG, Deck DH, Chambers HF. Does doxycycline protect against development of Clostridium difficile infection? Clin Infect Dis. 2012;55(5):615-20.
- Venkategowda PM, Rao SM, Mutkule DP, Rao MV, Taggu AN. Scrub typhus: clinical spectrum and outcome. Indian J Crit Care Med. 2015;19(4):208-13.
- Ziegler T, Winkler C, Wege K, Schmechel H. Doxycycline–the forgotten antibiotic. Med Klin (Munich). 2000;95(11):629-31.
- Garg P. Role of doxycycline in the management of dengue fever. Ind J Clin Pract. 2018;29(2):132-5.
- Fredeking RM, Zavala-Castro JE, González-Martínez P, Moguel-Rodríguez W, Sanchez EC, Foster MJ, et al. Dengue patients treated with doxycycline showed lower mortality associated to a reduction in IL-6 and TNF levels. Recent Pat Antiinfect Drug Discov. 2015;10(1):51-8.
- Aggarwal S, Walia K, Madhumati J, Gopalkrishnan R, Ohri VC, Gangakhedkar R. Treatment Guidelines for Antimicrobial Use in Common Syndromes. 2nd Edition, ICMR: New Delhi, India; 2019.
- Rahi M, Gupte MD, Bhargava A, Varghese GM, Arora R. DHR-ICMR Guidelines for diagnosis & management of Rickettsial diseases in India. Indian J Med Res. 2015;141(4):417-22.
- Fernandes JP, Hazra D, David SN, Paul K, Abhilash P. Acute undifferentiated febrile illness in the elderly: a clinical profile. Curr Med Issues. 2019;17(4):103-7.
- Salagre KD, Sahay RN, Pazare AR, Dubey A, Marathe KK. A study of clinical profile of patients presenting with complications of acute febrile illnesses during monsoon. J Assoc Physicians India. 2017;65(9):37-42.
- Bhise SM, Taiwade PD, Zodpey SS, Agarwal VA. Scrub typhus in rural areas & suburbs of Vidarbha region of Maharashtra. Indian J Med Res. 2020;151(1):104-6.
- Behera B, Biswal M, Das RR, Dey A, Jena J, Dhal S, et al. Clinico-epidemiological analysis of scrub typhus in hospitalised patients presenting with acute undifferentiated febrile illness: a hospital-based study from Eastern India. Indian J Med Microbiol. 2019;37(2):278-80.
- Wangdi K, Kasturiaratchi K, Nery SV, Lau CL, Gray DJ, Clements ACA. Diversity of infectious aetiologies of acute undifferentiated febrile illnesses in south and Southeast Asia: a systematic review. BMC Infect Dis. 2019;19(1):577.
- Varghese GM, Abraham OC, Mathai D, Thomas K, Aaron R, Kavitha ML, et al. Scrub typhus among hospitalised patients with febrile illness in South India: magnitude and clinical predictors. J Infect. 2006;52(1):56-60.
- Tsaganos T, Tseti IK, Tziolos N, Soumelas GS, Koupetori M, Pyrpasopoulou A, et al. Randomized, controlled, multicentre clinical trial of the antipyretic effect of intravenous paracetamol in patients admitted to hospital with infection. Br J Clin Pharmacol. 2017;83(4):742-50.
- Arulrhaj S, Tiwaskar M, Sabharwal M, Saikia R, Majid S, Rathod R, et al. Effectiveness of nimesulide in acute fever management in adults: retrospective electronic medical records database study outcome in outpatient department. J Assoc Physicians India. 2021;69(7):11-2.
- Lakshmi RMMVN, Dharma TV, Sudhaharan S, Surya SMV, Emmadi R, Yadati SR, et al. Prevalence of scrub typhus in a tertiary care centre in Telangana, South India. Iran J Microbiol. 2020;12(3):204-8.
- Phimda K, Hoontrakul S, Suttinont C, Chareonwat S, Losuwanaluk K, Chueasuwanchai S, et al. Doxycycline versus azithromycin for treatment of leptospirosis and scrub Antimicrob Agents Chemother. 2007;51(9):3259-63.
- Shelke YP, Deotale YS, Maraskolhe DL. Spectrum of infections in acute febrile illness in central India. Indian J Med Microbiol. 2017;35(4):480-4.
- Susilawati TN, McBride WJ. Acute undifferentiated fever in Asia: a review of the literature. Southeast Asian J Trop Med Public Health. 2014;45(3):719-26.
- Mørch K, Manoharan A, Chandy S, Chacko N, Alvarez-Uria G, Patil S, et al. Acute undifferentiated fever in India: a multicentre study of aetiology and diagnostic accuracy. BMC Infect Dis. 2017;17(1):665.
- Singhi S, Chaudhary D, Varghese GM, Bhalla A, Karthi N, Kalantri S, et al; Indian Society of Critical Care Medicine Tropical fever Group. Tropical fevers: management guidelines. Indian J Crit Care Med. 2014;18(2):62-9.
- Mave V, Chandanwale A, Kagal A, Khadse S, Kadam D, Bharadwaj R, et al. High burden of antimicrobial resistance and mortality among adults and children with community-onset bacterial infections in India. J Infect Dis. 2017;215(8):1312-20.
- Robinson ML, Kadam D, Kagal A, Khadse S, Kinikar A, Valvi C, et al. Antibiotic utilization and the role of suspected and diagnosed mosquito-borne illness among adults and children with acute febrile illness in Pune, India. Clin Infect Dis. 2018;66(10):1602-9.
- Francino MP. Antibiotics and the human gut microbiome: dysbioses and accumulation of resistances. Front Microbiol. 2016;6:1543.