Abstract
Turner syndrome is the most common chromosomal abnormality leading to gonadal failure and primary amenorrhea. While half of the cases have monosomy of chromosome X, the remaining exhibit mosaicism resulting in wide variation of phenotypic characteristics and clinical manifestations. We present a case of a 24-year-old female with mosaic variant Turner syndrome. The diagnosis was confirmed by karyotype analysis and laparoscopy.
Keywords: Turner syndrome, amenorrhea, karyotype analysis, laparoscopy
Turner syndrome is a genetic disorder that is characterized by complete or partial loss of one of the two X chromosomes in a phenotypic female. It was first described by Henry Turner in 1938. There is a wide variability of phenotypic features in a Turner syndrome patient due to the partial or complete monosomy of chromosome X, which is a result of chromosomal nondysjunction. Despite the variability, amenorrhea due to ovarian insufficiency and short stature is common to most of them. The phenotypic features exhibited by the non-mosaic 45,XO patient include short stature, webbed neck, shield chest, cubitus valgus, low hairline, short fourth metatarsal, etc. Apart from these, cardiac abnormalities like coarctation of aorta, renal abnormalities, ophthalmologic problems, sensorineural hearing loss and thyroid abnormalities are relatively common in them. In the mosaic variant of Turner syndrome, clinical manifestations may present to varying degree and sometimes the classical phenotypic features may be absent.
We present a case of mosaic variant Turner syndrome with primary amenorrhea due to ovarian insufficiency but no classical phenotypic characteristics.
Case Report
A 24-year-old female presented to the Gynecology OPD of Rajindra Hospital, Patiala with the chief complaint of failure to menstruate (primary amenorrhea). Patient explained that she was apparently well till 16 years of age when she went for gynecological consultation along with her mother for primary amenorrhea. According to her mother, they were told that the patient did not have a uterus as seen on ultrasound and were asked to consult a Gynecologist when the patient wanted to get married. So, the patient reported to Rajindra Hospital at 24 years of age with primary amenorrhea and absence of secondary sexual characters. The patient had no history of cyanosis, recurrent respiratory tract infection, cardiological problem or developmental delay. The patient had no other significant medical or surgical past history. There was no similar complaint in the family.
Suspecting Turner syndrome as the most likely cause of primary amenorrhea and absence of secondary sexual characters, we pursued our examination. On examination, patient had normal intelligence (she was a graduate). Her height and weight were 149 cm (4 feet 9 inch) and 36 kg, respectively. There was no low hairline, no low set ears, no webbed neck, no shielded chest, no cubitus valgus or genu valgum and no abnormality of nails or fingers. No abnormality was detected on thyroid gland palpation. The Tanner staging of breast was Stage 2 (breast buds present) (Fig. 1), while that for pubic hair was Stage 1 (sparse pubic hairs were present). Axillary hair were scanty. On auscultation, first and second heart sounds were normally heard and no murmur was audible. Abdomen on palpation was nontender and on local examination, urethral and vaginal openings were present. Labia majora and minora were not well developed. Based on the examination, a provisional diagnosis of primary amenorrhea was made and further investigation and examination under anesthesia were planned.
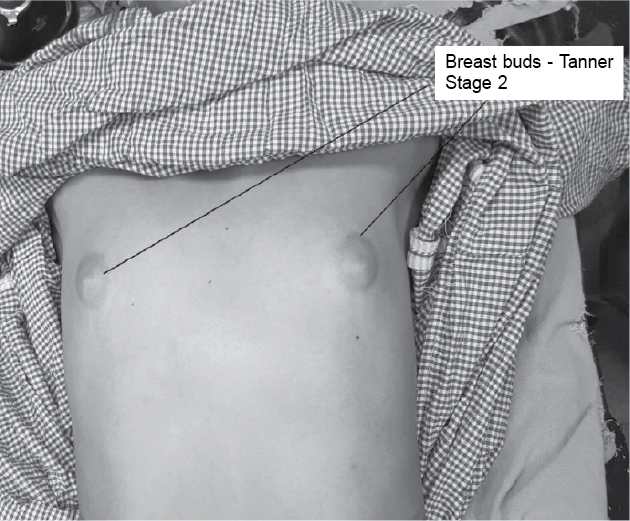
Figure 1. Breast buds - Tanner Stage 2.
Investigations were as under:
- Complete blood count (CBC) – Hemoglobin-11.7 g/dL, WBC-9,200/µL, platelet-4,15,000/µL, HCT-35.9%, MCV-75.6 fL, MCH-24.6 pg and MCHC-32.6 g/dL.
- Liver function test (LFT) – Total bilirubin-0.56 mg/dL, direct bilirubin-0.12 mg/dL, indirect bilirubin-0.44 mg/dL, serum glutamic oxaloacetic transaminase (SGOT)-39 IU/L, serum glutamic pyruvic transaminase (SGPT)-28 IU/L and serum alkaline phosphatase-67 IU/L.
- Renal function test (RFT) – Blood urea-18 mg% and serum creatinine-0.7 mg%.
- Fasting blood sugar (FBS) – 84 mg%.
- Thyroid profile – T3-1.53 ng/mL, T4-9.7 µg/dL and TSH-3.56 µIU/mL (normal).
- Luteinizing hormone (LH) – 12.6 mIU/mL.
- Serum prolactin (7.18 ng/mL) was normal.
- Follicle-stimulating hormone (FSH) (63.16 mIU/mL) was markedly elevated.
- Speech audiometry revealed normal hearing on right side and mild hearing loss on left side – 21.6 dB and 38.3 dB, respectively.
- Radiological investigations were carried out to determine the condition of the reproductive organs and any other organ involvement:
- Ultrasound revealed that both the ovaries were present but the uterus was severely hypoplastic.
- Magnetic resonance imaging (MRI) further confirmed the uterus to be markedly small in size, 3 × 6 cm. Endometrium was thin-lined. Ovaries were normally visualized and no other pelvic/adnexal mass or free fluid in the pelvis was seen.
- Ultrasound of whole abdomen showed no other organ abnormality.
- Echocardiography was normal.
- Chest X-ray revealed no abnormality.
- She was subjected to karyotyping which revealed two cell lines, that is, first cell line (25 cells examined) showed 45,XO (Fig. 2) while the second cell line (5 cells examined) showed 46,XX (Fig. 3).

Figure 2. Karyotyping - 45,XO (25).
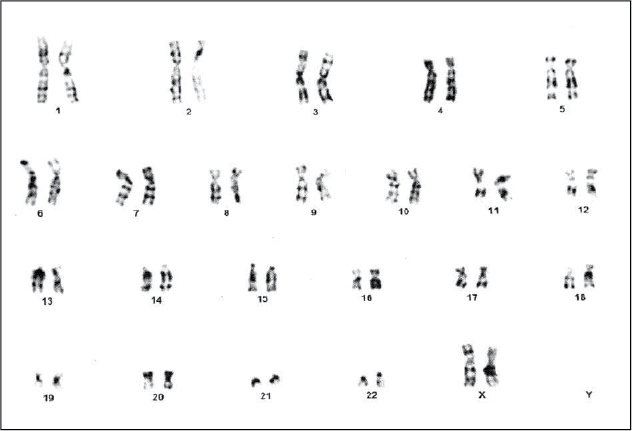
Figure 3. Karyotyping - 46,XX (5).
This mosaic chromosome complement was consistent with the diagnosis of variant Turner syndrome.
Examination under anesthesia along with diagnostic laparoscopy was performed.
Per speculum examination showed a small-sized cervix with both anterior and posterior lips (Fig. 4).
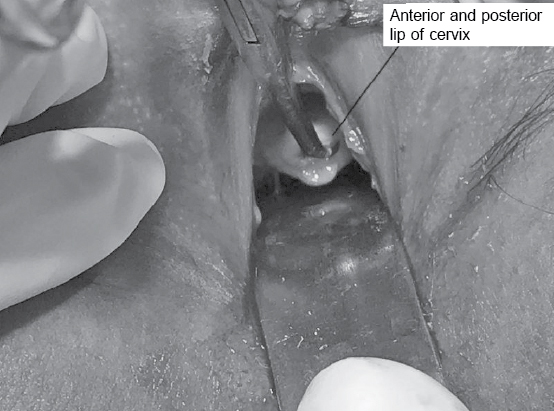
Figure 4. Per speculum examination - Note small size cervix with both anterior and posterior lips.
Per vaginal examination – vagina admitted one finger and was 9 cm in length. Dimpling of external os was felt.
Uterine sound went inside the cervix up to 3 cm. Laparoscopy performed revealed small 3 × 2 cm sized mass at the site of uterus which was suspected to be the uterine body (Fig. 5). Uterosacral ligaments were appreciated. Fallopian tubes and ovaries could not be appreciated.
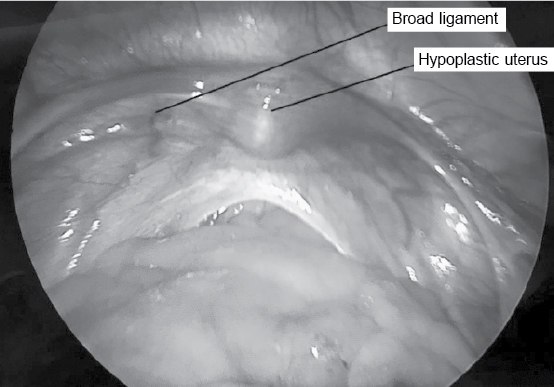
Figure 5. Diagnostic laparoscopy - Note hypoplastic uterus with broad ligaments.
Based on the karyotype analysis and diagnostic laparoscopy, final diagnosis of mosaic Turner syndrome with gonadal dysgenesis was made.
The patient and her parents were counseled about this condition and the patient was discharged on hormonal therapy, vitamin D and calcium supplementation and the advice to undergo screening tests including echocardiography, audiometry, thyroid function test, LFT, RFT, lipid profile and complete hemogram on regular basis.
Patient is coming for regular follow-up. She is married now with normal sexual life.
Discussion
Turner syndrome is one of the most common sex chromosome abnormality among the females. It affects 1 in 2,000 live born females. About 99% of Turner syndrome patients are spontaneously aborted, usually in the first trimester. More than 50% of the females with Turner syndrome have 45,XO or the non-mosaic karyotype. Many other karyotype variations including short or long arm deletion, ring X, isochromosome of long arm and mosaicism (cell lines of 45,XO and 46,XX) are also present. Short stature and gonadal insufficiency are the two clinical manifestations which are present in most of the cases irrespective of whether they are non-mosaic or mosaic variant of Turner syndrome.
Short stature is the most common reason for consultation to the doctor in the case of Turner syndrome, especially mosaic variants. The growth failure in this condition usually causes the adult female height to be about 20 cm below the average adult height of the female of that country and the average adult female height of our country is 152 cm. In our case, the patient’s height was 149 cm, indicating that this phenotypic feature was not completely affected due to mosaicism which is different from the majority of the cases which have short stature.
The other phenotypic features consistent for Turner syndrome including webbed neck, low hairline, short fourth metatarsal, cubitus valgus, peripheral edema, etc., may be absent in the mosaic variant making it difficult to diagnose clinically. The other conditions like renal abnormalities (horseshoe-shaped kidney), cardiovascular abnormalities (coarctation of aorta), sensorineural hearing loss and thyroid abnormalities which are present in most of the non-mosaic patients may be absent in the mosaic variant. In our patient, none of these conditions was present.
Apart from these clinical features, gonadal insufficiency is usually present in most of the patients with Turner syndrome. It is due to early degeneration of the oocytes so that at birth only few oocytes are present and the ovaries look like fibrotic streaks. This gonadal failure is determined by raised FSH, ultrasound or diagnostic laparoscopy. In our patient, FSH was elevated and ovaries were not appreciated at all on diagnostic laparoscopy. Also, due to lack of estrogen production, secondary sexual characters like breast development, pubic hair growth, etc., do not develop.
Despite the fact that gonadal insufficiency is common in both mosaic and non-mosaic variants, some studies do indicate that normal puberty and spontaneous menarche occur more commonly in living mosaic variants (including those with some cell lines having 45,XO while others having 46,XX or 47,XXX or 46,XiXp) as compared to the non-mosaic (42-70% and 10%, respectively). Although these mosaic variants do conceive but there is increased risk of spontaneous abortions, stillbirths and congenital abnormalities.
A number of complications can develop in these patients as they grow old, including hypertension, cardiac complications (especially aortic root dissection), thyroid abnormalities, sensorineural hearing loss, increased risk of fractures due to osteoporosis and liver cirrhosis. Although these conditions are relatively more common and serious in the non-mosaic variant but all the Turner syndrome patients should be screened periodically for these conditions for timely diagnosis and thus decrease the morbidity and mortality associated with it. Apart from this, patients are given psychological supportand hormone therapy staring from adolescence and continuing till 50-60 years after which continuation of therapy depends on risk factors for developing complications like deep-vein thrombosis, cardiovascular complications, etc. So, a multidisciplinary team should carry out the management of girls with Turner syndrome right from diagnosis to adulthood.
Suggested Reading
- Zinn AR, Page DC, Fisher EM. Turner syndrome: the case of the missing sex chromosome. Trends Genet. 1993;9(3):90-3.
- Gravholt CH. Clinical practice in Turner syndrome. Nat Clin Pract Endocrinol Metab. 2005;1(1):41-52.
- Hook EB, Warburton D. The distribution of chromosomal genotypes associated with Turner’s syndrome: live birth prevalence rates and evidence for diminished fetal mortality and severity in genotypes associated with structural X abnormalities or mosaicism. Hum Genet. 1983;64(1):24-7.
- Suri M, Kabra M, Jain U, Sanders V, Saxena R, Shukla A, et al. A clinical and cytogenetic study of Turner syndrome. Indian Pediatr. 1995;32(4):433-42.
- Ogata T, Matsuo N. Turner syndrome and female sex chromosome aberrations: deduction of the principal factors involved in the development of clinical features. Hum Genet. 1995;95(6):607-29.
- Jones KL. Smith’s Recognizable Patterns of Human Malformations. 5th Edition, New York: WB Saunders Company; 1997.
- Mamidi RS, Kulkarni B, Singh A. Secular trends in height in different states of India in relation to socioeconomic characteristics and dietary intakes. Food Nutr Bull. 2011;32(1):23-34.
- Lippe BM. Primary ovarian failure. In: Kaplan SA (Ed.). Clinical Pediatric Endocrinology. Philadelphia: WB Saunders Company; 1990. pp. 325-66.
- Sybert VP. Phenotypic effects of mosaicism for a 47,XXX cell line in Turner syndrome. J Med Genet. 2002;39(3):217-20.
- Lippe B, Westra SJ, Boechat MI. Ovarian function in Turner syndrome: recognizing the spectrum. In: Hibi I, Takano K (Eds.). Basic and Clinical Approach to Turner Syndrome. Elsevier Science Publishers; 1993. pp. 117-22.
- Kaneko N, Kawagoe S, Hiroi M. Turner’s syndrome - review of the literature with reference to a successful pregnancy outcome. Gynecol Obstet Invest. 1990;29(2):81-7.
- Gravholt CH. Medical problems of adult Turner’s syndrome. Horm Res. 2001;56 Suppl 1:44-50.
- Gonzalez L, Witchel SF. The patient with Turner syndrome: puberty and medical management concerns. Fertil Steril. 2012;98(4):780-6.