Abstract
Nimesulide, a nonsteroidal anti-inflammatory drug (NSAID), has been used as an effective treatment regimen for patients aged >12 years for fever, acute pain, acute tendinitis, osteoarthritis and dysmenorrhea. It is reported to be a superior antipyretic and anti-inflammatory drug than paracetamol and aspirin, respectively, and is equal to any of the NSAIDs alone in terms of analgesia. This paper reviews the current scenario of nimesulide in adult patients, concerning clinical evidence, use in special population and expert opinion. Overall, in comparison to other NSAIDs, including coxibs, nimesulide has a promising overall efficacy, safety and tolerability profile, as well as a satisfactory benefit/risk evaluation.
Keywords: COX-2 inhibitor, fever, inflammation, NSAID, pain
BACKGROUND, HISTORY AND INDICATION
Nimesulide was first approved in Italy in 1985 and is currently available in more than 50 countries.1 It is a nonsteroidal anti-inflammatory drug (NSAID) and has been used globally as an effective treatment regimen for patients aged >12 years with fever, acute pain, acute tendinitis, osteoarthritis and dysmenorrhea.2 Nimesulide was first approved in India by the Drugs Controller General of India (DCGI) in 1995 in various dosages including nimesulide 100 mg tablet, nimesulide 50 mg/5 mL as suspension, nimesulide transdermal 1% gel for topical use and nimesulide 50 mg/100 mg as a suppository (rectal administration). In 1999, 200 mg dose of nimesulide was also introduced in the market as suppository.3
Fixed-drug combinations (FDCs) have been widely accepted as treatment options for a variety of diseases because of their superior therapeutic efficacy and safety, improved patient compliance and adherence and reduced cost to patients when compared with single-drug therapies. However, concerns have been raised regarding their irrationality and utility. FDCs have been gaining popularity in the Indian pharmaceutical market.4 Combinations of nimesulide with several other active drug compounds have been approved by DCGI.5 It was banned in 2000 in countries such as Switzerland, Spain and the US. As per an order dated 10th March 2011, nimesulide is not indicated for use in children below 12 years of age in India.
Over 90,000 people have participated in more than 200 clinical trials examining the efficacy and safety profile of nimesulide in acute and chronic inflammatory and painful disorders, the results of which showed it to be significantly better than placebo and at least comparable or in a few cases superior to other active NSAIDs (ketoprofen, naproxen, ibuprofen) in the treatment of pain and inflammation. This paper aims to review the current scenario of nimesulide in adult patients and expert opinion.
MECHANISM OF ACTION
Nimesulide is a selective inhibitor of the cyclooxygenase (COX)-2 enzyme (Fig. 1). It has unique chemical and pharmacokinetic properties.6 Nimesulide imparts its pharmacological properties through a multifactorial mechanism of action (Fig. 2). It also inhibits phosphodiesterase IV and neutrophil function and renders antioxidant effects in human chondrocytes at a pharmacologic level; inhibits overexpression of tumor necrosis factor α (TNF-α) thereby regulating the release of other hyperalgesic cytokines.7 In an in-vivo study, nimesulide decreased interleukin (IL)-1β, IL-6 and TNF-α levels in a rat model of depression.8 In patients with knee osteoarthritis, nimesulide significantly reduced the level of pro-inflammatory cytokine IL-6.9
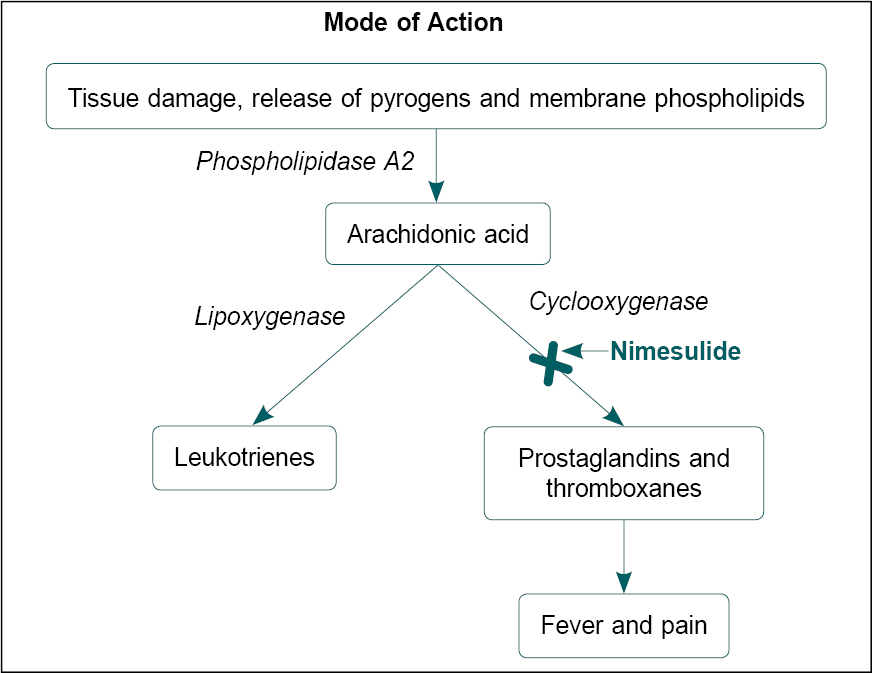
Figure 1. Mechanism of action of nimesulide.
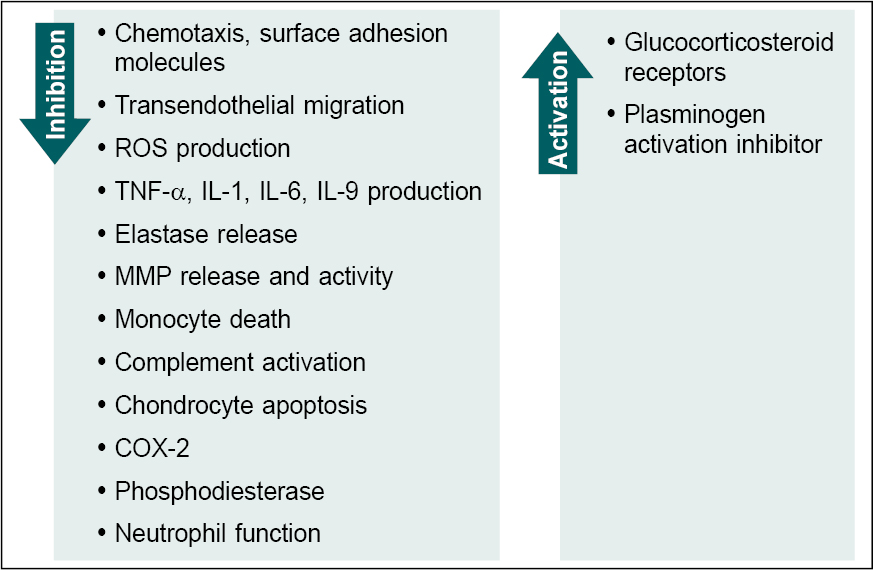
Figure 2. Multifactorial action of nimesulide.
ROS = Reactive oxygen species; TNF-a = Tumor necrosis factor a; IL = Interleukin; MMP = Matrix metalloproteinase; COX-2 = Cyclooxygenase-2.
CLINICAL EVIDENCE
Nimesulide in Fever
Multiple clinical trials have evaluated the antipyretic efficacy of nimesulide in comparison to paracetamol, diclofenac and acetylsalicylic acid. Nimesulide was found to be superior over the other antipyretic drugs in terms of safety, efficacy and tolerability, and was reported to normalize the body temperature in a short period of time.2,10-12 Nimesulide starts its action within 15 minutes of its administration in comparison with other antipyretics like paracetamol.2 In a recent Indian study of 302 adult patients with acute fever, the body temperature declined to (99.7 [1.8]°F) from the baseline body temperature of (103.2 [1.5]°F) following nimesulide treatment. Liver and renal profiles were reported to be normal from baseline to follow-up visit within 14 days; only 2% complained of nausea and dyspepsia.2
Reiner et al (1984), Reiner et al (1985) and Cunietti et al (1993) demonstrated that rectal administration of nimesulide (200 mg), diclofenac (100 mg), paracetamol (500 mg) reduced the body temperature from baseline and normalized heart rate and arterial pressure. Their efficacy was significantly comparable and well-tolerated. In comparison to the placebo, nimesulide was more efficient in fever management and shortened the duration of fever. Minor and temporary adverse effects were reported in drug-treated group with no side effects in placebo-treated group.10-12
Nimesulide in Pain
Nimesulide is an effective, safer and well tolerable therapeutic alternative for the treatment of a variety of painful inflammatory disorders. Compared to other NSAIDs such as etodolac, ibuprofen, naproxen and rofecoxib, nimesulide has a more effective, tolerable and rapid analgesic activity during the short-term usage.6,13-20 There were no significant adverse events were reported among the various anti-inflammatory drugs and nimesulide.
Nimesulide delivered rapid analgesic effect and overall positive risk/benefit profile with no or minimal side effects in the management of osteoarthritis knee pain versus placebo and other oral NSAIDs such as celecoxib and rofecoxib in a comparative, double-blind, randomized clinical trial study.6,13
Zuckermann et al have reported the clinical effectiveness of nimesulide in the management of post-hemorrhoidectomy pain and inflammation.21 Significant reduction in painful postoperative complications of various surgeries including ear, nose and throat surgery, dental surgery, oral surgery and arthroscopic knee surgery with nimesulide has also been reported.22-25
Comparison of efficacy of nimesulide with that of placebo and other analgesics such as ketoprofen (rectal administration), diclofenac (rectal administration), naproxen and niflumic acid has demonstrated the quick and effective analgesic activity of nimesulide over the other active drugs.21
A study by Pierleoni and co-workers compared the analgesic effects of nimesulide with ketoprofen in 46 patients who had undergone dental surgery. From day 2, night pain was absent in 69.5% of patients treated with nimesulide and 43.5% in patients treated with ketoprofen. Overall, 91.3% and 65.1% of patients treated with nimesulide and ketoprofen, respectively judged the efficacy as excellent or good. However, the quality of sleep improvement was consistent in both treatment groups following reduction in night pain.23
Binning evaluated the effect of nimesulide 100 mg compared to naproxen 500 mg and placebo on postoperative pain in 94 patients undergoing arthroscopic knee surgery.25 The mean pain intensity was significantly reduced in nimesulide-treated patients (10.91) compared to placebo (6.29, p < 0.001) and naproxen-treated patients (7.07, p = 0.003). Estimated median time to 50% decrease in pain intensity was significantly shorter in patients treated with nimesulide (0.82 h) compared with those receiving naproxen (1.05 h; p = 0.017) or placebo (1.33 h; p = 0.001).25
Both nimesulide (300 mg) and rofecoxib (25 mg) were equally effective in improving pain and quality of life in patients with knee osteoarthritis. However, nimesulide was superior than rofecoxib in terms of pain relief (1.2 vs. 1.65; p = 0.007), scale of Western Ontario and McMaster Universities Osteoarthritis Index (9.3 vs. 12.96; p = 0.009) after 30 days of treatment.14
In a study comparing the efficacy of nimesulide versus ibuprofen in the treatment of acute low back pain, both NSAIDs significantly improved all measured parameters of the pain and back function. However, the patients’ capacity for daily tasks was significantly improved in nimesulide-treated patients compared to ibuprofen-treated patients after 10 days (10.0 vs. 16.5; p = 0.026). In addition, nimesulide was more effective in improving lateral bending measurements (p = 0.026) with lower incidence of gastrointestinal (GI) side effects compared to ibuprofen.17
THERAPEUTIC ADJUNCT OF COVID-19
Various in-silico studies have shown nimesulide as a potential drug for the treatment of coronavirus disease 2019 (COVID-19).26-28 As it can terminate the transport function of B0AT1, a target receptor for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), it can be considered a co-adjuvant in the treatment of COVID-19.29 Selective inhibition of the COX-2 enzyme could be a potential therapeutic approach for the treatment of pain and inflammation.30 Few NSAIDs were found to have anti-inflammatory activity in mice with influenza A infection by decreasing the pro-inflammatory cytokines (TNF-α and IL-6) and increasing the levels of granulocyte-colony stimulating factors (G-CSF) in bronchoalveolar lavage fluid.31 This experimental evidence was the rationale behind recommending nimesulide as prophylactic treatment for early COVID-19 infection symptoms at home in cases with no sign of allergy or toxicity. The associated risk of hepatotoxicity is very minimal when it is taken at the prescribed time and dose. Nonetheless, treatment with nimesulide was found to be safer and well-tolerated in the recommended cohort, with just 1 patient complaining of GI pain.32
SAFETY PROFILE
COX-1 inhibition results in various adverse events such as GI discomfort, renal and urinary disorders and cardiac disorders. In-vitro studies have demonstrated nimesulide’s high selectivity for COX-2 inhibition rather than COX-1 inhibition.33 Even though GI symptoms such as dyspepsia and other nonserious complaints have been the most prevalent NSAID side effects, Bjarnason et al reported that nimesulide is superior and well-tolerated to other NSAIDs in terms of intestinal perforation, GI ulceration and bleeding.34 Kress et al and Famaey have demonstrated nimesulide as one of the most preferred antipyretic agent with no substantial GI adverse events.33,35
Laporte et al have shown that the nimesulide associated risk of upper GI bleeding was significantly lower compared to that of ibuprofen and far lower than other established NSAIDs such as ketorolac, ketoprofen and piroxicam. Ketorolac and ketoprofen showed poor GI tolerability.36
Many NSAIDs have been linked to hepatic side effects that are usually idiosyncratic reactions associated with individual vulnerability. A systematic review by Sriuttha et al demonstrated clinically significant hepatotoxic events with NSAIDs including celecoxib, etoricoxib and diclofenac. The hepatotoxicity occurred most commonly in patients treated with diclofenac in the range of 0.015 to 4.3 (×10−2), followed by celecoxib [0.13-0.38 (×10−2)] and etoricoxib [0.005-0.930 (×10−2).37 There is an extremely low prevalence of hepatic reactions (1-5 among 1,00,000 exposed) and ranges from an asymptomatic and temporary elevation of hepatic enzymes to significant hepatic injury and liver dysfunction in nimesulide treated patients compared to other NSAIDs.38,39
In almost 64,000 nimesulide-treated patients, no occurrences of hepatitis or hepatic failure were documented in clinical investigations.34
Both COX-1 and COX-2 enzymes play a major role in renal function by regulating the renin-angiotensin system and renal tubular excretion/re-absorption systems, therefore it’s very likely that nimesulide’s major COX-2 inhibition action would only affect a proportion of renal function.40,41 Nimesulide also has transitory effects on sodium and potassium excretion in healthy volunteers. In normal healthy subjects, nimesulide 200 mg BD was observed to attenuate the increase in plasma renin and aldosterone caused by furosemide, along with a reduction in urinary prostaglandin E2 (PGE2) excretion. With or without furosemide, nimesulide elevated urine flow and water excretion and decreased glomerular filtration rate and renal plasma flow.42 Most NSAIDs exhibit similar physiological effects.43 Cardiovascular events also influence its safety profile. Clinical trials have shown a minimal risk of cardiovascular events in patients receiving nimesulide.34 Serious cardiovascular events such as myocardial infarction and congestive heart failure that have lately been observed with coxibs and other NSAIDs have been seldom seen with nimesulide.
EXPERT OPINION
Nimesulide versus Nimesulide + Paracetamol Combination
Paracetamol is an effective antipyretic and analgesic drug but shows poor anti-inflammatory effects. It is a non-selective COX-2 inhibitor, unlike nimesulide.
In terms of analgesia or commencement of an action, the combination of nimesulide and paracetamol had no pharmacological advantage over nimesulide or paracetamol alone.44
Both show antipyretic and analgesic effect by inhibiting the biosynthesis of prostaglandin (PG) that is responsible for inducing pain and inflammation.45 Irrational drug combinations put the patients at risk of adverse effects. Although nimesulide has been reported as a superior analgesic drug over other NSAIDs in terms of safety and efficacy, its combination with paracetamol is associated with increased hepatotoxic effects, and therefore it is very crucial to make clinicians and consumers aware of this irrational combination and stop them from practicing such combination.44
Patient Profiles for Nimesulide
Arthritic conditions
In patients with a history of moderate to severe pain associated with osteoarthritis of the hip and knee, nimesulide (100 mg BD) and diclofenac (50 mg TID) had comparable overall tolerability and efficacy. However, nimesulide-treated patients had fewer GI side effects (36.3%) than the diclofenac-treated patients (47.2%). These results signify that the efficacy of nimesulide is equivalent to that of diclofenac as long-term therapy of osteoarthritis, and that nimesulide provides a superior gastric safety profile than diclofenac.46
Nimesulide was superior to other widely used NSAIDs such as celecoxib and rofecoxib in providing symptomatic relief in patients with knee osteoarthritis. It also showed a rapid analgesic effect in pain associated with walking in comparison to celecoxib, rofecoxib and etodolac. No significant adverse events were reported with any of the therapies, and the tolerability was comparable across treatment groups. These results show that nimesulide is superior or equivalent to the comparator drug in terms of rapid analgesic action, quality of life, tolerability and incidence of adverse events.13,14,16
In a study to determine the most promising/optimal dose of nimesulide for the treatment of osteoarthritis-associated pain in terms of efficacy/safety ratio, 392 patients were randomized to treatment with nimesulide (50 mg, 100 mg and 200 mg BD) and placebo for 1 month. Nimesulide-treated group showed a rapid onset of analgesia that lasted for 12 hours after the drug intake; however, a significant analgesic effect was observed in groups receiving 100 mg and 200 mg of nimesulide within 1.5 hours of drug intake.
Nimesulide 100 mg and 200 mg produced the best results. Doses of 50 mg and 100 mg were well-tolerated, while the dose of 200 mg showed a higher incidence of adverse events, although not significant. Thus, 100 mg twice a day was demonstrated to be the optimal dose of nimesulide for the treatment of osteoarthritis.15
These results provide evidence that nimesulide is a unique NSAID with analgesic properties producing the best results for the management of arthritic conditions.
Dysmenorrhea
Nimesulide was superior over other NSAIDs such as diclofenac, naproxen and mefenamic acid including placebo in reducing pain associated with primary dysmenorrhea.47 Investigation of the therapeutic effect of nimesulide on overall intrauterine perfusion and PGs in menstrual blood showed that nimesulide reduced the concentration of PGF2α (responsible for uterine contraction) and PGE1 or PGE2 (have relaxing/contractile effects) by 60% and 80%, respectively.48,49 Nimesulide transforms smooth muscle from the pathological condition of dysmenorrheic contracture to the physiological state of eumenorrheic contractions. It is also apparently linked with a decline in uterine artery vascular resistance.50,51
In a multicenter, randomized, double-blind clinical trial 308 women with primary dysmenorrhea were allocated to either nimesulide (3 tablets/day) or diclofenac (3 tablets/day) for the first 3 days of their menstrual cycle.52 Both nimesulide and diclofenac treatments significantly reduced pain by 82% and 79%, respectively at the second hour. However, onset of analgesia was more rapid with nimesulide starting at 30 minutes, with a decline in abdominal discomfort by 35% versus 27% in both the first and second treatment cycles, respectively. Both drugs had comparable beneficial effects headache and backache. The two treatments were well-tolerated. The prevalence of gastric discomfort was found to be significantly lower in nimesulide-treated group.
No significant changes in uterine and ovarian blood flow were observed in eumenorrheic women receiving either placebo or nimesulide (100 mg) in a double-blind, placebo-controlled clinical trial.53 In the same study, nimesulide improved symptoms and reduced uterine artery impedance (pulsatile index, PI) earlier than naproxen (500 mg, single oral dose) among women with moderate to severe dysmenorrhea. The clinical effect of both the active drugs on elevated uterine impedance was comparable. The fundus showed the most significant alterations. The ovarian branch remained unaffected. These results signify that nimesulide reduced elevated uterine vascular resistance in dysmenorrhea to normal eumenorrheic levels at a higher rate than naproxen.
Nimesulide also played a crucial role in the treatment of primary dysmenorrhea.
Post-surgical pain
Nimesulide was found to be comparatively similar to naproxen and more superior to placebo in reducing pain, inflammation and edema in patients suffering from post-hemorrhoidectomy associated pain and arthroscopic knee post-surgery associated pain. All the treatments were reported to be well-tolerated.21,25
Comparison of the analgesic effect of nimesulide with ketoprofen in patients undergoing dental surgery and third molar surgery showed that nimesulide was superior to ketoprofen in reducing pain and inflammation. The quality of sleep, mastication and swallowing function recovered more quickly among the nimesulide treated patients. Nimesulide was found to be effective in managing pain, edema and trismus after surgical removal of the third molar.23,54
Ramella and co-workers showed that treatment with nimesulide and diclofenac (rectal administration) significantly reduced pain, swelling, hyperemia, as well as the elimination of minor fever, without any adverse events among patients who underwent saphenectomy or inguinal hernioplasty.55 Based on these observations, nimesulide can be recommended in the treatment of postoperative pain, with a rapid onset of analgesic action.
Cancer pain
Two studies were compared the analgesic efficacy and tolerability of nimesulide with naproxen in patients with advanced cancer pain.19,20 Both studies revealed that 200 mg BD nimesulide and 500 mg BD naproxen have similar analgesic effects with lower incidence of adverse reactions.19,20 This indicates that nimesulide can also be used in the treatment of cancer pain.
Ear, nose and throat disease
Several multicenter clinical trials have investigated the efficacy and tolerability of nimesulide and other anti-inflammatory drugs such as seaprose-S and ketoprofen, in the management of inflammatory disorders of ear, nose and throat (ENT).22,56,57 Nimesulide efficiently reduced inflammatory signs and symptoms of ENT disorders. It had a more rapid onset of action than other NSAIDs. Both nimesulide and ketoprofen significantly reduced swelling and hyperemia in patients with postoperative inflammatory complications post-ENT surgery. However, all treatment regimens were well-tolerated with a low prevalence of mild adverse events.
Airway inflammation
Nimesulide has proven to be beneficial in regulating the inflammatory process in conditions such as rhinopharyngitis, rhinosinusitis, tubaritis, rhinitis and middle ear ailments. Nimesulide in combination with a mucolytic drug, ambroxol, has been shown to have similar anti-inflammatory effects.58 Oral nimesulide has been proven to be effective and well-tolerated in the treatment of aspirin-sensitive asthmatic patients.59
In a study conducted on patients with chronic mucus hypersecretion, nimesulide administered for 3 weeks significantly reduced sputum viscosity. No severe adverse events were reported due to nimesulide medication.60
CONCLUSION
Nimesulide is a new generation NSAID with a unique chemical structure. It has a preferentially high inhibitory effect on the COX-2 enzyme with a low preference for COX-1. This allows nimesulide its wide range of application as an anti-inflammatory agent through its multifactorial mode of action. The rapid onset of analgesic effects of nimesulide in the treatment of pain and inflammation is exerted both centrally and peripherally. Treatment with nimesulide has been proven safer, effective and well-tolerable in cases of acute pain associated with postoperative pain and inflammation, inflammation due to soft tissue injuries, odontostomatological inflammatory pain, extra-articular traumatism, osteoarthritis pain and cancer pain. The risk of liver reactions is comparable to that of other conventional NSAIDs and coxibs. In comparison to other NSAIDs, including coxibs, nimesulide has a promising overall efficacy, safety and tolerability profile, as well as a satisfactory benefit/risk evaluation. Nimesulide as antipyretic is effective and safer in fever management when used for shorter duration.
Disclosures
Source of funding: This initiative was funded by Dr Reddy’s Laboratories Limited, Hyderabad, India.
Conflict of interest: Anup Uttam Petare and Krishna Chaitanya Veligandla are employees of Dr Reddy’s Laboratories Limited, Hyderabad. All other authors have no conflict of interest to declare.
REFERENCES
- European Medicines Agency. Nimesulide. Available at: https://www.ema.europa.eu/en/medicines/human/referrals/nimesulide-0#overview-section. Last accessed February 28, 2022.
- Arulrhaj S, Tiwaskar M, Sabharwal M, Saikia R, Majid S, Rathod R, et al. Effectiveness of nimesulide in acute fever management in adults: retrospective electronic medical records database study outcome in outpatient department. J Assoc Physicians India. 2021;69(7):11-2.
- List of New Drugs Approved in India from 1991 to 2000. Available at: https://cdsco.gov.in/opencms/resources/UploadCDSCOWeb/2018/UploadApprovalNewDrugs/1991-2000.pdf. Last accessed February 28, 2022.
- Gautam CS, Saha L. Fixed dose drug combinations (FDCs): rational or irrational: a view point. Br J Clin Pharmacol. 2008;65(5):795-6.
- Fixed dose combinations approved by DCG (I) since 1961 to 31st December 2019. Available at: https://cdsco.gov.in/opencms/opencms/system/modules/CDSCO.WEB/elements/download_file_division.jsp?num_id=NzM3OQ. Last accessed February 28, 2022.
- Rainsford KD; Members of the Consensus Report Group on Nimesulide. Nimesulide - a multifactorial approach to inflammation and pain: scientific and clinical consensus. Curr Med Res Opin. 2006;22(6):1161-70.
- Bennett A, Villa G. Nimesulide: an NSAID that preferentially inhibits COX-2, and has various unique pharmacological activities. Expert Opin Pharmacother. 2000;1(2):277-86.
- Luo W, Luo Y, Yang J. Proteomics-based screening of the target proteins associated with antidepressant-like effect and mechanism of nimesulide. Sci Rep. 2020;10(1):11052.
- Bianchi M, Broggini M, Balzarini P, Franchi S, Sacerdote P. Effects of nimesulide on pain and on synovial fluid concentrations of substance P, interleukin-6 and interleukin-8 in patients with knee osteoarthritis: comparison with celecoxib. Int J Clin Pract. 2007;61(8):1270-7.
- Reiner M, Cereghetti S, Haeusermann M, Monti T. Antipyretic activity of nimesulide suppositories: double blind versus diclofenac and placebo. Int J Clin Pharmacol Ther Toxicol. 1985;23(12):673-7.
- Reiner M, Massera E, Magni E. Nimesulide in the treatment of fever: a double-blind, crossover clinical trial. J Int Med Res. 1984;12(2):102-7.
- Cunietti E, Monti M, Viganó A, D’Aprile E, Saligari A, Scafuro E, et al. Nimesulide in the treatment of hyperpyrexia in the aged. Double-blind comparison with paracetamol. Arzneimittelforschung. 1993;43(2):160-2.
- Bianchi M, Broggini M. A randomised, double-blind, clinical trial comparing the efficacy of nimesulide, celecoxib and rofecoxib in osteoarthritis of the knee. Drugs. 2003;63 Suppl 1:37-46.
- Herrera JA, González M. Comparative evaluation of the effectiveness and tolerability of nimesulide versus rofecoxib taken once a day in the treatment of patients with knee osteoarthritis. Am J Ther. 2003;10(6):468-72.
- Bourgeois P, Dreiser RL, Lequesne MG, Macciocchi A, Monti T. Multi-centre double-blind study to define the most favourable dose of nimesulide in terms of efficacy/safety ratio in the treatment of osteoarthritis. Eur J Rheum Inflamm. 1994;14(2):39-50.
- Lücker PW, Pawlowski C, Friedrich I, Faiella F, Magni E. Double-blind, randomised, multi-centre clinical study evaluating the efficacy and tolerability of nimesulide in comparison with etodalac in patients suffering from osteoarthritis of the knee. Eur J Rheumatol Inflamm. 1994;14(2):29-38.
- Pohjolainen T, Jekunen A, Autio L, Vuorela H. Treatment of acute low back pain with the COX-2-selective anti-inflammatory drug nimesulide: results of a randomized, double-blind comparative trial versus ibuprofen. Spine (Phila Pa 1976). 2000;25(12):1579-85.
- Dreiser RL, Riebenfeld D. A double-blind study of the efficacy of nimesulide in the treatment of ankle sprain in comparison with placebo. Drugs. 1993;46 Suppl 1:183-6.
- Toscani F, Gallucci M, Scaricabarozzi I. Nimesulide in the treatment of advanced cancer pain. Double-blind comparison with naproxen. Drugs. 1993;46 Suppl 1:156-8.
- Gallucci M, Toscani F, Mapelli A, Cantarelli A, Veca G, Scaricabarozzi I. Nimesulide in the treatment of advanced cancer pain. Double-blind comparison with naproxen. Arzneimittelforschung. 1992;42(8):1028-30.
- Zuckermann M, Panconesi R, Scaricabarozzi I, Nava ML, Bechi P. Clinical efficacy and tolerability of nimesulide compared with naproxen in the treatment of posthaemorrhoidectomy pain and inflammation. Drugs. 1993;46 Suppl 1:177-9.
- Coscarelli S, Scaricabarozzi I, Nava ML, Alajmo E. A comparison of nimesulide and ketoprofen in the prevention and treatment of painful postoperative inflammatory complications of ear, nose and throat surgery. Drugs. 1993;46 Suppl 1:174-6.
- Pierleoni P, Tonelli P, Scaricabarozzi I. A double-blind comparison of nimesulide and ketoprofen in dental surgery. Drugs. 1993;46 Suppl 1:168-70.
- Ragot JP, Monti T, Macciocchi A. Controlled clinical investigation of acute analgesic activity of nimesulide in pain after oral surgery. Drugs. 1993;46 Suppl 1:162-7.
- Binning A. Nimesulide in the treatment of postoperative pain: a double-blind, comparative study in patients undergoing arthroscopic knee surgery. Clin J Pain. 2007; 23(7):565-70.
- Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583(7816):459-68.
- Cava C, Bertoli G, Castiglioni I. In silico discovery of candidate drugs against Covid-19. Viruses. 2020;12(4):404.
- Zhou Y, Hou Y, Shen J, Huang Y, Martin W, Cheng F. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020;6:14.
- Scalise M, Indiveri C. Repurposing nimesulide, a potent inhibitor of the B0AT1 subunit of the SARS-CoV-2 receptor, as a therapeutic adjuvant of COVID-19. SLAS Discov. 2020;25(10):1171-3.
- FitzGerald GA, Patrono C. The coxibs, selective inhibitors of cyclooxygenase-2. N Engl J Med. 2001;345(6):433-42.
- Carey MA, Bradbury JA, Rebolloso YD, Graves JP, Zeldin DC, Germolec DR. Pharmacologic inhibition of COX-1 and COX-2 in influenza A viral infection in mice. PLoS One. 2010;5(7):e11610.
- Suter F, Consolaro E, Pedroni S, Moroni C, Pastò E, Paganini MV, et al. A simple, home-therapy algorithm to prevent hospitalisation for COVID-19 patients: a retrospective observational matched-cohort study. EClinicalMedicine. 2021;37:100941.
- Famaey JP. In vitro and in vivo pharmacological evidence of selective cyclooxygenase-2 inhibition by nimesulide: an overview. Inflamm Res. 1997;46(11):437-46.
- Bjarnason I, Bissoli F, Conforti A, Maiden L, Moore N, Moretti U, et al. Adverse reactions and their mechanisms from nimesulide. In: Rainsford KD (Ed.). Nimesulide - Actions and Uses. Basel: Birkhauser; 2005. pp. 315-415.
- Kress HG, Baltov A, Basinski A, Berghea F, Castellsague J, Codreanu C, et al. Acute pain: a multifaceted challenge – the role of nimesulide. Curr Med Res Opin. 2016;32(1):23-36.
- Laporte JR, Ibáñez L, Vidal X, Vendrell L, Leone R. Upper gastrointestinal bleeding associated with the use of NSAIDs: newer versus older agents. Drug Saf. 2004;27(6):411-20.
- Sriuttha P, Sirichanchuen B, Permsuwan U. Hepatotoxicity of nonsteroidal anti-inflammatory drugs: a systematic review of randomized controlled trials. Int J Hepatol. 2018;2018:5253623.
- Trechot P, Gillet P, Gay G, Hanesse B, Netter P, Castot A, et al. Incidence of hepatitis induced by non-steroidal anti-inflammatory drugs (NSAID). Ann Rheum Dis. 1997;55(12):936.
- Walker AM. Quantitative studies of the risk of serious hepatic injury in persons using nonsteroidal antiinflammatory drugs. Arthritis Rheum. 1997;40(2):201-8.
- Rodriguez F, Llinás, Moreno C, Salazar FJ. Role of cyclooxygenase-2-derived metabolites and NO in renal response to bradykinin. Hypertension. 2001;37(1):129-34.
- Roig F, Llinás MT, Löpez R, Salazar FJ. Role of cyclooxygenase-2 in the prolonged regulation of renal function. Hypertension. 2002;40(50:721-8.
- Steinhäuslin F, Munafo A, Buclin T, Macciocchi A, Biollaz J. Renal effects of nimesulide in furosemide-treated subjects. Drugs. 1993;46 Suppl 1:257-62.
- Schlondorff D. Renal complications of nonsteroidal anti-inflammatory drugs. Kidney Int. 1993;44(3):643-53.
- Ahmed M, Upadhyaya P, Seth V. Comparison of analgesic effects of nimesulide, paracetamol, and their combination in animal models. Indian J Pharmacol. 2010;42(6):354-7.
- Gautam CS, Aditya S. Irrational drug combinations: need to sensitize undergraduates. Indian J Pharmacol. 2006;38(3):167-70.
- Huskisson EC, Macciocchi A, Rahlfs VW, Bernstein RM, Bremner AD, Doyle DV, et al. Nimesulide versus diclofenac in the treatment of osteoarthritis of the hip or knee: An active controlled equivalence study. Curr Ther Res. 1999;60(5):253-65.
- Bianchi M, Ehrlich GE, Facchinetti F, Huskisson EC, Jenoure P, La Marca A, et al. Clinical applications of nimesulide in pain, arthritic conditions and fever. In: Rainsford KD (Eds.). Nimesulide - Actions and Uses. Basel: Birkhauser; 2005. pp. 245-313.
- Baracat EC, Motta ELA, Lima GR. Clinical evaluation of the efficacy and tolerability of nimesulide versus piroxicam in the therapeutic of primary dysmenorrhea [Avaliacaoclinica da eficacia e tolerabilidade do nimesulide versus piroxicam naterapeutica da dismenorreiaprimaria]. Jornal Brasileiro de Ginecologia. 1991;101(10):467-70.
- Chiantera A, Tesauro R, Di Leo S, Meli S, Scaricabarozzi I. Nimesulide in the treatment of pelvic inflammatory diseases. A multicentre clinical trial conducted in Campania and Sicily. Drugs. 1993;46 Suppl 1:134-6.
- Pulkkinen MO. Alterations in intrauterine pressure, menstrual fluid prostaglandin F levels, and pain in dysmenorrheic women treated with nimesulide. J Clin Pharmacol. 1987;27(1):65-9.
- Pulkkinen MO. Is there a rationale for the use of nimesulide in the treatment of dysmenorrhoea? Drugs Today. 2001;37:31-8.
- Facchinetti F, Piccinini F, Sgarbi L, Renzetti D, Volpe A. Nimesulide in the treatment of primary dysmenorrhoea: a double-blind study versus diclofenac. Drugs Today. 2001;37:39-45.
- Pirhonen J, Pulkkinen M. The effect of nimesulide and naproxen on the uterine and ovarian arterial blood flow velocity. A Doppler study. Acta Obstet Gynecol Scand. 1995;74(7):549-53.
- Pouchain EC, Costa FW, Bezerra TP, Soares EC. Comparative efficacy of nimesulide and ketoprofen on inflammatory events in third molar surgery: a split-mouth, prospective, randomized, double-blind study. Int J Oral Maxillofac Surg. 2015;44(7):876-84.
- Ramella G, Costagli V, Vetere M, Capra C, Casella G, Sogni A, et al. Comparison of nimesulide and diclofenac in the prevention and treatment of painful inflammatory postoperative complications of general surgery. Drugs. 1993;46 Suppl 1:159-61.
- Ottaviani A, Mantovani M, Scaricabarozzi I. A multicentre clinical study of nimesulide in inflammatory diseases of the ear, nose and throat. Drugs. 1993;46 Suppl 1:96-9.
- Banchini G, Scaricabarozzi I, Montecorboli U, Ceccarelli A, Chiesa F, Ditri L, et al. Double-blind study of nimesulide in divers with inflammatory disorders of the ear, nose and throat. Drugs. 1993;46 Suppl 1:100-2.
- Bellussi L, Passàli D. Treatment of upper airways inflammation with nimesulide. Drugs. 1993;46 Suppl 1:107-10.
- Bianco S, Robuschi M, Petrigni G, Scuri M, Pieroni MG, Refini RM, et al. Efficacy and tolerability of nimesulide in asthmatic patients intolerant to aspirin. Drugs. 1993;46 Suppl 1:115-20.
- Sofia M, Molino A, Mormile M, Stanziola A, Scaricabarozzi I, Carratù L. Nimesulide in the treatment of chronic bronchitis. Drugs. 1993;46 Suppl 1:111-4.