Abstract
Broken-heart syndrome (BHS) is an acute reversible myocardial injury of left or right ventricular myocardium in the absence of coronary occlusion. We, herein, discuss a case of a postmenopausal female presenting with angina equivalent with surface electrocardiogram (ECG) and echocardiography consistent for acute coronary syndrome. The patient was subsequently diagnosed and treated as BHS.
Keywords: Acute coronary syndrome, reversible cardiomyopathy, acute heart failure
Broken-heart syndrome (BHS) is an acute reversible condition of left or right ventricular myocardium usually preceded by a stressful trigger.1 It was first described in Japan in 1990 and is also recognized by other names like stress-induced cardiomyopathy, Takotsubo (octopus pot) cardiomyopathy and apical ballooning syndrome. The prevalence is currently estimated at 1% to 2% of patients with suspected acute coronary syndrome.2 BHS predominantly involves postmenopausal females and is commonly associated with emotional or physical stress.3 Stress cardiomyopathy represents a form of neurocardiogenic myocardial stunning for which the exact pathophysiologic mechanism is unknown. But leading hypothesis suggests catecholamine excess and sympathetic nervous system hyperactivity resulting in regional microvascular dysfunction accompanied by cellular calcium overload in susceptible individuals.4-8 Several diagnostic criteria have been proposed with the most commonly used one being the Mayo Clinic 2004 diagnostic criteria3,9 that were modified in 2008. The revised criteria include:
- Transient hypokinesis, akinesis or dyskinesis of the left ventricular mid-segments with or without apical involvement; the regional wall-motion abnormalities extend beyond a single epicardial vascular distribution; a stressful trigger is often, but not always present.
- Absence of obstructive coronary disease or angiographic evidence of acute plaque rupture.
- New electrocardiographic abnormalities (either ST-segment elevation and/or T-wave inversion) or modest elevation in cardiac troponin levels.
- Absence of pheochromocytoma or myocarditis.
BHS is a self-limiting condition, usually with rapid resolution of symptoms and left ventricular dysfunction.3 Therapy is largely supportive and tailored based on patient’s presentation. Angiotensin-converting enzyme (ACE) inhibitors and beta-blockers can be considered for treatment. Care should be taken to avoid QTc prolonging medications for their predisposition to malignant ventricular arrhythmia. Long-term prognosis is generally good but in-hospital mortality has been reported due to cardiogenic shock, left ventricular mechanical complications, malignant ventricular arrhythmia and systemic thromboembolism. Recurrences are common to the tune of 2% to 4% per year and up to 20% at 10 years, even after recovery of left ventricular functions.3
CASE REPORT
A 75-year-old diabetic female presented to the emergency department with complaints of acute onset shortness of breath and chest heaviness. She did not complain of any other abdominal or chest symptoms. Other than emotional frailty owing to social loneliness, her personal and family history were unremarkable. She was living with normal coronaries evaluated 4-year back for chest pain with ST-segment elevation on electrocardiogram (ECG). Follow-up ECG (Fig. 1A) and transthoracic echocardiography (TTE) over last few years were unremarkable. On general physical examination, patient was seen to be anxious. The heart rhythm was regular with a rate of 108 beats/min; the blood pressure was 130/80 mmHg and respiration was thoraco-abdominal with a rate of 24 breaths/min. Findings from review of the systems, other than as reported above were normal. A 12-lead surface ECG showed sinus tachycardia and ST-segment coving with T-wave inversion in precordial and limb leads (Fig. 1B). Qualitative cardiac troponin T was negative and routine biochemical analysis including complete blood count, renal and liver function tests were unremarkable. TTE revealed severe left ventricular systolic dysfunction (ejection fraction about 35% by Simpson) with marked regional wall motion abnormalities in the mid anterior, mid-septal, mid inferior and apical segments of the left ventricle (Fig. 2A and 2B). The patient was immediately shifted to the catheterization laboratory, where coronary angiography was done. Angiography revealed normal coronary arteries with mild plaquing of major epicardial vessels (Fig. 3).
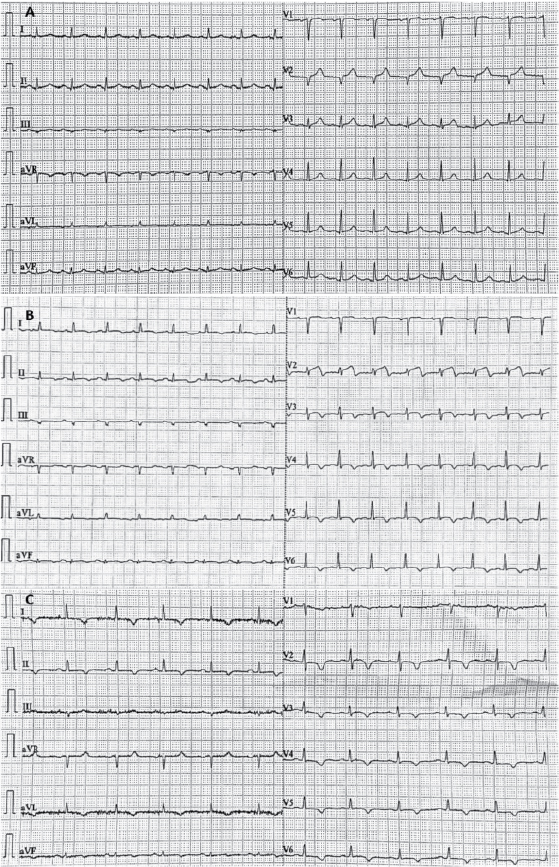
Figure 1. Serial ECGs demonstrating dynamic ST-T changes from before event (A); during event (B) and in 4 weeks follow-up period (C).
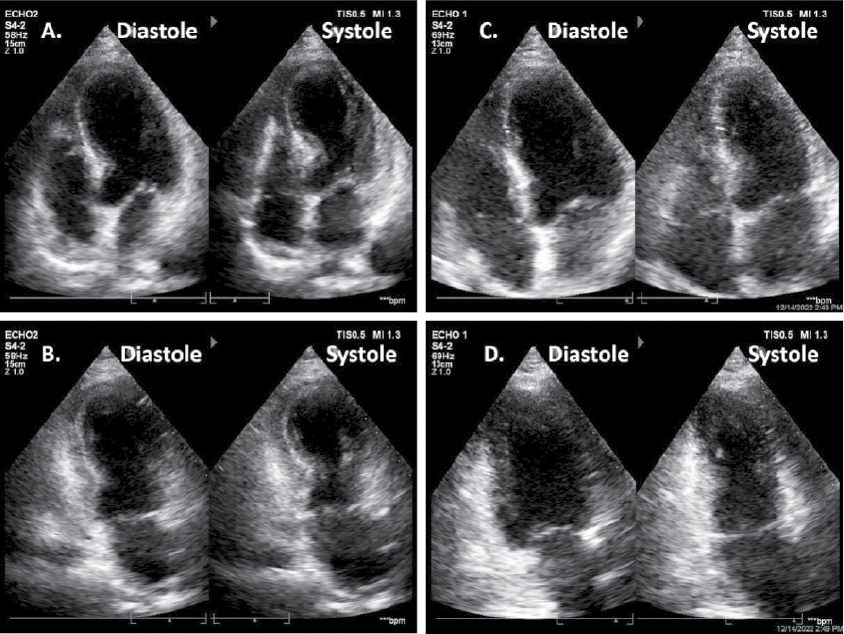
Figure 2. Echocardiogram in apical 4 chamber and apical 2 chamber views during acute event (A & B) showing apical akinesis and basal hyperkinesis leading to characteristic appearance of apical ballooning or Japanese Takotsubo (octopus pot) in systolic frame. Follow-up echocardiogram in same views after 4 weeks of follow-up (C & D) showing normalization of wall motion abnormalities.

Figure 3. Coronary angiogram demonstrating normal epicardial coronary arteries.
The patient was treated with beta-blockers, ACE inhibitors, dual antiplatelets, statins, diuretics and antidepressants. She was discharged in a stable condition after 3 days without any complications. She was re-evaluated after 4 weeks. At the follow-up visit, all her symptoms had disappeared. ECG showed repolarization abnormalities in the form of T-wave inversion consistent for previous event (Fig. 1C). Echocardiography showed normalization of the left ventricular systolic function (ejection fraction about 60% by Simpson) with no residual wall motion abnormality (Fig. 2C and 2D).
DISCUSSION
Our patient was an elderly postmenopausal female, diagnosed with stress-induced cardiomyopathy or BHS after fulfilling Mayo Clinic diagnostic criteria. Retrospectively, her previous coronary event 4 years back, was likely the same event precipitated by extreme stress that went undiagnosed. As compared to acute coronary syndrome patients, the cardiac enzymes of BHS typically demonstrate mild elevation and return to baseline faster. Our patient did not have raised cardiac troponin despite marked ECG changes and significant wall abnormality in TTE.
Her cardiac functions normalized after both the events. BHS is a self-limiting condition, usually with rapid resolution of symptoms and ventricular dysfunction. The precise mechanism that cause this neurocardiogenic myocardial stunning are unknown but it has been postulated that catecholamine and hormones play important role in the pathogenesis; hence, it is also namedas Takotsubo endocrinopathy.10
CONCLUSION
BHS is an acute reversible cardiac disorder with a transient left or right ventricular wall motion abnormality. We should keep a high index of suspicion in postmenopausal women who present with acute coronary syndrome like symptoms after an intense emotional stress. The in-hospital mortality rate seems to be low, and there is a low risk for recurrence.
REFERENCES
- Hurst RT, Prasad A, Askew JW 3rd, Sengupta PP, Tajik AJ. Takotsubo cardiomyopathy: a unique cardiomyopathy with variable ventricular morphology. JACC Cardiovasc Imaging. 2010;3(6):641-9.
- Gianni M, Dentali F, Grandi AM, Sumner G, Hiralal R, Lonn E. Apical ballooning syndrome or Takotsubo cardiomyopathy: a systematic review. Eur Heart J. 2006;27(13):1523-9.
- Medina de Chazal H, Del Buono MG, Keyser-Marcus L, Ma L, Moeller FG, Berrocal D, et al. Stress cardiomyopathy diagnosis and treatment: JACC state-of-the-art review. J Am Coll Cardiol. 2018;72(16):1955-71.
- Del Buono M, Carbone S, Abbate A. Comment on Stiermaier et al. Prevalence and prognostic impact of diabetes in Takotsubo syndrome: insights from the international, multicenter GEIST Registry. Diabetes Care 2018;41:1084-8. Diabetes Care. 2018;41(7):e121.
- Pelliccia F, Kaski JC, Crea F, Camici PG. Pathophysiology of Takotsubo syndrome. Circulation. 2017;135(24):2426-41.
- Sonnino C, Van Tassell BW, Toldo S, Del Buono MG, Moeller FG, Abbate A. Lack of soluble circulating cardio-depressant factors in Takotsubo cardiomyopathy. Auton Neurosci. 2017;208:170-2.
- Akashi YJ, Goldstein DS, Barbara G, Ueyama T. Takotsubo cardiomyopathy: a new form of acute, reversible heart failure. Circulation. 2008;118(25):2754-62.
- Wittstein IS, Thiemann DR, Lima JA, Baughman KL, Schulman SP, Gerstenblith G, et al. Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med. 2005;352(6):539-48.
- Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J. 2008;155(3):408-17.
- Kalra S, Lakhani OJ, Chaudhary S. Takotsubo endocrinopathy. Eur Endocrinol. 2020;16(2):97-9.