Published in IJCP
February 2023
Clinical Practice Guideline
Unraveling Prostaglandin and NLRP3 Inflammasomemediated Pathways of Primary Dysmenorrhea and the Role of Mefenamic Acid and Its Combinations
February 11, 2023 | Anita Kant, Jayam Kannan, Sunita Chandra, Chandravati, Bharti kalra, Helen Mary, Prabhu Kasture, Bharati Rajshekhar, Anshu Jindal, Amuthambigai, Sachin Dalal, Reema Jain, Sujata Kulkarni, Surekha Vinay, Ummamaheshwar Sindur, Pragya Ojha, Renu Chakravarty, Harini, Archana Mayekar, Jayanta Kumar Gupta
Obstetrics and Gynecology
Abstract
Painful menstrual cramps during or around the time of the monthly cycle are known as dysmenorrhea. The estimated global prevalence in women of reproductive age ranges from 45% to 95%. It has a significant negative impact on regular activities and productivity at work. However, despite the severe consequences on quality of life, primary dysmenorrhea (PD) is underdiagnosed. Dysmenorrhea has complex pathogenesis. It involves the release of prostaglandins and activation of the nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasome and also includes the involvement of other mediators such as bradykinin, histamine and acetylcholine. Even though nonsteroidal anti-inflammatory drugs (NSAIDs) remain the most common type of pain medication, the question of which one should be the most preferred is still open to debate. The current review examines the existing evidence for the pathogenesis of PD and makes evidence based and clinical experience based recommendations for the use of mefenamic acid and its combination in the treatment of dysmenorrhea. Mefenamic acid alleviates PD by inhibiting endometrial prostaglandin formation, restoring normal uterine activity, and reducing the inflammatory response by inhibiting the NLRP3 inflammasome and reducing the release of cytokines such as interleukin (IL)-1β. It is also known to have bradykinin antagonist activity. Dicyclomine has a dual action of blocking the muscarinic action of acetylcholine in postganglionic parasympathetic effect or regions and acting directly on uterine smooth muscle by blocking bradykinin and histamine receptors to relieve spasms. According to the experts, mefenamic acid and dicyclomine act synergistically by acting on the different pathways of dysmenorrhea by blocking multifactorial agents attributed to the cause of dysmenorrhea. Hence, the combination of mefenamic acid and dicyclomine should be the preferred treatment option for dysmenorrhea.
Keywords: Dysmenorrhea, spasmolytic, mefenamic acid, dicyclomine
Dysmenorrhea is described as spasms in the lower abdomen that spreads to the thighs or lower spine, with mild to severe intensity pain occurring during the menstrual cycle. The pain is also associated with other symptoms such as vomiting, headache, back pain, diarrhea and fatigue.1
Estimated at a global prevalence of 45% to 95% in women of reproductive age,2 studies from India have reported that dysmenorrhea prevalence ranges from 50% to 87.8%.3
In the United States, an estimated $2 billion is lost every year due to missed work and productivity.4 The condition leaves a considerable impact on the quality of life, daily activity, work-related productivity and academic performance.5 However, despite the significant effects on quality of life, not only is the prevalence of primary dysmenorrhea (PD) underestimated, but additionally, most women do not frequently seek medical treatment as the common perception associated with dysmenorrheic pain is that ‘it comes as a part of menstruation’.6
Despite being a common and recurrent problem, dysmenorrhea remains undiagnosed, unattended or inadequately treated due to multiple factors attributable to the several myths and misunderstandings prevailing in society as low awareness of this particular issue, poor health-seeking behavior accepting this pain as normal, leading to the assumption that treatment is neither required nor available. Even in the clinics, this problem is not proactively inquired about, and there are no uniform standards regarding pharmacotherapy in treating dysmenorrhea.
Menstruation is known to be a self-inflammatory process bringing about the sloughing of the endometrium functional lining at the end of the routine fertility cycles. In the absence of conception, the endometrium undergoes significant changes, including the release of hormones and decidualization of cells that play a key role in local inflammation within the endometrium leading up to PD. While the role of prostaglandin is established in triggering the symptoms of PD, the role of inflammation has a strong effect on the pathological process of PD.7 The complex pathophysiology of dysmenorrhea having multifactorial components needs to be addressed while choosing treatment options. Though nonsteroidal anti-inflammatory drugs (NSAIDs) remain the mainstay in the management of pain, the question of which one should be the most preferred continues to baffle clinicians. The role of spasmolytic cannot be ignored in addressing the complex pathophysiology of dysmenorrhea. It has to be chosen wisely based upon its multiple actions on the various attributable factors and synergize with the preferred NSAID to act comprehensively on all the pathophysiological factors of dysmenorrhea. Through the expert group deliberations, we have aimed to develop a consensus about the use of mefenamic acid and its combination with dicyclomine in dysmenorrhea in the light of its additional and unique mode of action on these various pathogenetic pathways of dysmenorrhea.
MATERIAL AND METHODS
The present review examines the existing evidence on the pathogenesis of PD and presents evidence based and clinical experience based recommendations on the use of mefenamic acid and mefenamic acid + dicyclomine in the treatment of dysmenorrhea.
To generate the highest possible evidence base for the use of mefenamic acid and dicyclomine in dysmenorrhea, a comprehensive review of the literature was conducted on PubMed, Cochrane database and Google Scholar. Existing guidelines, meta-analysis, systematic reviews, randomized controlled trials (RCTs), non-RCT studies and experimental studies relating to the use of mefenamic acid, dicyclomine and the pathogenesis of dysmenorrhea were searched, scanned, selected and reviewed. Only articles in English and dysmenorrhea, experimental, clinical studies or reviews on mefenamic acid and dicyclomine were included. Articles in other foreign languages or those focusing on the treatment of secondary dysmenorrhea or surgery in PD were excluded from the selection. Recommendations for the treatment of dysmenorrhea are based on the available evidence and preliminary discussions among expert groups.
A panel of 25 gynecologists from across the country was created at the initiation of the study. Evidence synthesized was documented and shared with all the members of the expert panel at least a week before the meeting, which formed the evidence base for the discussions. The discussion undertook in two separate meetings conducted on April 9, 2022 and April 23, 2022, via virtual meeting. The recommendations and deliberations of the members were recorded for further analysis, refinement and inclusion in the consensus.
The recommendation formulated by the expert panel has been included in this article. If there was little or no evidence, the panel relied on logical empiricism and consensus to generate recommendations about the rational use of mefenamic acid or its combination with dicyclomine in the treatment of dysmenorrhea.
The Impact of Dysmenorrhea
Dysmenorrhea often has a debilitating effect on a woman’s life, which becomes more concerning owing to its recurring nature. A study showed that almost 50% of the participants experienced a disruption of their daily activities, studying abilities and working productivity. Apart from absenteeism, the loss in annual productivity due to presenteeism was reported to be 7 times more than the annual productivity due to absenteeism. Further, the anticipation of severe dysmenorrhea in the upcoming month is another cause of mental stress and unproductivity in women.5
Despite being one of the most common recurring gynecological problems, most experts agreed that there is a lack of general awareness about dysmenorrhea, due to which most of the time it remains an underdiagnosed and undertreated condition. The problem frequently remains medically unaddressed as women and young girls do not get accurate and sufficient information, attempts at self-management, undervaluing the condition, and a feeling of embarrassment in talking about the problem.5
Most of the experts were of the view that there is an urgent need to enhance awareness about the condition highlighting the reasons to adolescent girls and women as to why they should seek treatment for dysmenorrhea. Several experts believed dysmenorrhea should be included as the extension topic of menstruation.
The Pathways Involved in Primary Dysmenorrhea
It is a well-established fact that dysmenorrhea is an intricate meshwork of various pathogenetic mechanisms, including behavioral and psychological factors. It is an inflammatory event driven by prostaglandin, nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasome and several other chemokines and pro-inflammatory cytokines.
Prostaglandins in Dysmenorrhea
Menstruation is a self-driven inflammatory process, where the release of prostaglandins is cited to be the trigger for causing PD.5
Prostaglandins are key mediators of underlying inflammation and essential for increasing abnormal uterine activity during menstruation. It is evident from the literature that women with PD have higher levels (2-7 times) of prostaglandin F2α (PGF2α) and prostaglandin E2 (PGE2). PGF2α in dysmenorrhea brings about vasoconstriction of the uterine wall and contraction of the myometrium, while PGE2 causes inflammation and contraction of the myometrium, reduced blood flow leading to uterine ischemia.7,8
NLRP3 Inflammasome
The NLRP3 inflammasome components immunolocalize to decidualized endometrial stromal cells right before menstruation.9
Activation of NLRP3 inflammasome helps activate the caspase-1, which in turn cleaves the pro-interleukin-1β and pro-interleukin-18 to active IL-1β and IL-18. Thus, before menstruation, the release of these pro-inflammatory cytokines along with leukocyte influx up-regulates the inflammatory response during menstruation, and their levels are reported to be present in high quantities in women with dysmenorrhea.1
Hence, curbing the activation and expression of NLRP3 inflammasome becomes an important approach to tackling dysmenorrhea.10 PGE2, including its receptors, also regulates the production of inflammatory cytokines such as IL-1β, IL-6 and monocyte chemotactic protein-1, inducing a vicious cycle.5
Others Mediators in Dysmenorrhea
Histamine, bradykinin and acetylcholine are considered to be the significant factors for causing dysmenorrhea by increasing uterine contractility, which leads to ischemia and pain. Additionally, bradykinin increases the release of prostaglandins and also hypersensitizes the pain fibers.5
Mefenamic Acid
Mode of action
Mefenamic acid relieves dysmenorrhea via its multiple modes of action. It has a dual action of inhibiting prostaglandin synthesis and blocking the E-type prostanoid receptors. Mefenamic acid possesses the ability to inhibit pre-existing prostaglandin, which is the reason behind its faster onset of action.5
Mefenamic acid acts as an effective and selective inhibitor of the NLRP3 inflammasome. It blocks the channels viz. volume-regulated anion channel (VRAC) and transient receptor potential melastatin 2 channel (TRPM2), thereby reducing the cellular chloride efflux and calcium influx, respectively, which in turn blocks the activation of NLRP3 inflammasome. Since the secretion of the pro-inflammatory cytokine IL-1β is mediated by the activation of NLRP3, its inhibition by mefenamic acid attenuates the levels of IL-1β and alleviates inflammation. Mefenamic acid is a bradykinin antagonist, thus possessing an extended pathway of pain relief. It can inhibit the raised uterine contractility triggered by prostaglandin-endoperoxide analogs.5
Besides reduction in pain, it also reduces menstrual blood loss by improving platelet aggregation and degranulation through increased vasoconstriction.
Safety and efficacy
Mefenamic acid exhibited a superior efficacy when compared with other NSAIDs like ibuprofen, naproxen, indomethacin and paracetamol.5,11
It is usually well-tolerated, but common side effects may include headache, dizziness, nausea, diarrhea, abdominal discomfort, heartburn and hypersensitivity reactions.5
Mefenamic acid combination with dicyclomine
Mefenamic acid relieves PD by inhibiting endometrial prostaglandin formation, restoring normal uterine activity, and reducing inflammatory response by inhibiting the NLRP3 inflammasome and decreasing the release of cytokines such as IL-1β.5 It is also known to possess bradykinin antagonism action.7
|
Expert’s Comments
All the experts unanimously recommended the use of an NSAID that may have a direct analgesic effect via inhibition of prostaglandin synthesis and reduction of blood volume of the menstrual flow. Mefenamic acid is preferred by all of them due to superior clinical efficacy as observed during their clinical practice and additionally reducing the blood volume loss due to the excess menstrual flow.
Further, the experts opined that as inflammation underpins dysmenorrhea, hence treatment approaches to target underlying inflammatory process dysmenorrhea will be useful in ameliorating or reducing menstrual pain and other associated inflammatory symptoms. The rather unexplored role of NLRP3 inflammasome activation in causing dysmenorrhea symptoms piqued the interest of the experts and they were of the view that a drug that can inhibit the activation of NLRP3 inflammasome in uterine tissues and the inflammatory processes involved in dysmenorrhea will be an important asset to the armamentarium of dysmenorrhea treatment.
The novel information on NLRP3 inflammation and its role in dysmenorrhea and the inhibitory action of mefenamic acid are a value addition and should be correlated clinically.
Considering the multiple mechanisms and pathways involved in causing dysmenorrhea, experts also suggested using synergistic combination of drugs that can effectively act on different pathways thereby effectively mitigating the symptoms associated with dysmenorrhea.
|
Dicyclomine has a dual action of blocking the muscarinic action of acetylcholine in postganglionic parasympathetic effector regions and acts directly on the uterine smooth muscles by blocking the bradykinin and histamine receptors to offer relief from spasmolytic pain.5
One of the studies found that the combination of mefenamic acid and dicyclomine was well-tolerated and more effective than dicyclomine alone in moderate to severe cases of dysmenorrhea.5
Studies have demonstrated that mefenamic acid and dicyclomine show synergistic activity (Fig. 1) in patients with dysmenorrhea. The combination is not only highly effective in treating spasmodic dysmenorrhea but also well-tolerated.
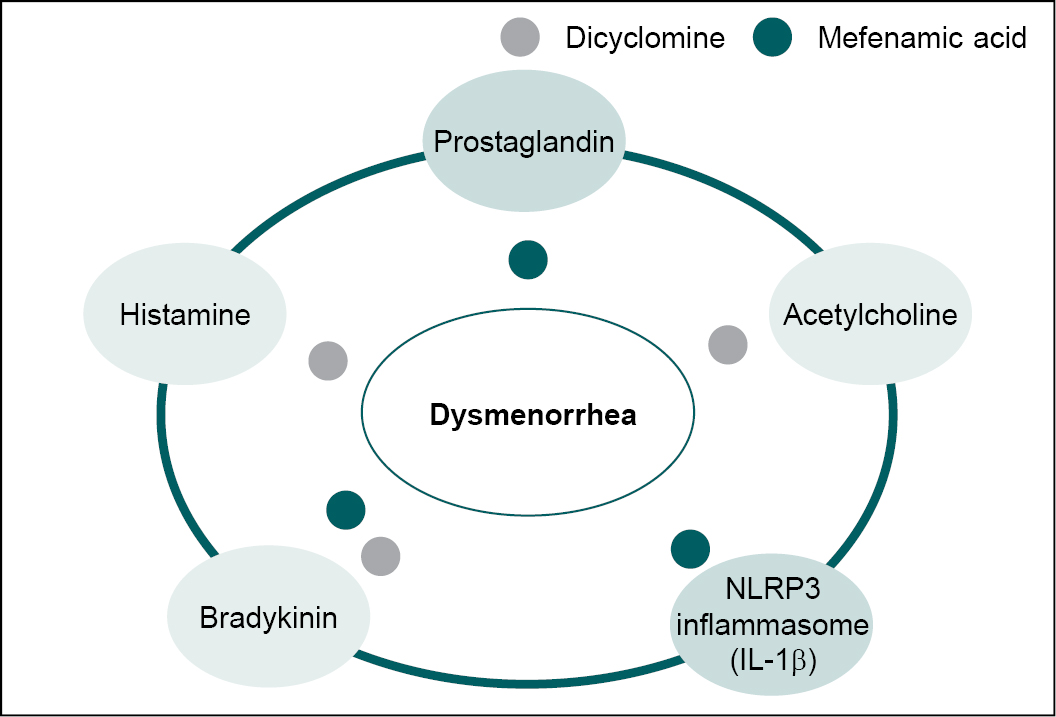
Figure 1. Synergistic action of dicyclomine and mefenamic acid on the various pathways of dysmenorrhea.
|
Expert’s Comments
We know that women with PD experience four different uterine contraction-related issues that are increased uterine resting tone, enhanced active pressure, increased number of contractions and dysrhythmic uterine activity.
So, a combined NSAID + antispasmodic, such as mefenamic acid + dicyclomine will be beneficial in treatment of dysmenorrhea.
Mefenamic acid and dicyclomine exerts synergism by acting on the various pathways of dysmenorrhea thereby blocking the multifactorial agents as attributable to the causation of dysmenorrhea and should be the preferred choice of treatment in dysmenorrhea.
While discussing other combinations with mefenamic acid including drotaverine and camylofin, the experts were of the view that these drugs did not lead to any statistically significant pain relief when compared with the combination of dicyclomine and mefenamic acid.
In view of the experts, paracetamol and/or its combination were not preferred by them due to its weak analgesic and no anti-inflammatory action. Moreover, the dosage for analgesia would be higher up to 1 g per dose, which may then be associated with more adverse effects especially hepatic injury.
|
Most experts said that they have used mefenamic acid alone or in combination with dicyclomine, which is effective in alleviating the symptoms of dysmenorrhea. All the experts echoed similar recommendations and clinical practice experiences with mefenamic or mefenamic acid with dicyclomine to be the first choice of treatment depending upon the individual patient’s symptoms. They had experienced positive results/outcomes with the use of the mefenamic acid and dicyclomine combination, and none of them ever required or felt the need to use any other alternative for treating pain and other associated symptoms of dysmenorrhea.
RECOMMENDATIONS
- Women need to be in the optimum physiological and psychological conditions, and hence it becomes very important for women to address the menstrual burden and provide effective treatment. Measures such as providing educational literature, posters, and digital platforms with necessary information should be adopted and used by all gynecologists/clinicians to raise awareness about dysmenorrhea. To promote awareness about dysmenorrhea, school based and community-based initiatives can be undertaken by gynecologists/clinicians supported by NGOs, CSR functionaries of corporates, other professional bodies and community service groups. It is important to raise awareness amongst all the community members and health care professionals alike.
- The pathophysiology of dysmenorrhea is complex and multifactorial. Several pathogenetic and inflammatory mechanisms are at work in causing pain and other symptoms of PD. Prostaglandin release triggers the PD, and recent evidence has also implicated the NLRP3 activation leading to an increase in the release of pro-inflammatory cytokines playing a primary role in the underlying pathogenesis of dysmenorrhea. Apart from prostaglandins, other mediators, including bradykinin, histamine, acetylcholine, etc., also play an important role in causing PD.
- In patients with dysmenorrhea, paracetamol is not effective because of its weak analgesic and no anti-inflammatory actions. It has dose-dependent analgesic action on the prostaglandins, and since multiple factors are attributed to dysmenorrheic pain, it doesn’t give complete relief.
- High dose paracetamol (1000 mg) has analgesic action for adults with mild to moderate acute pain, but 500 mg is sub-therapeutic for this action. It does not have any NLRP3 inhibitory action and, therefore, may not be suitable to alleviate the complex inflammatory process of dysmenorrhea. Thus, 500 mg dosage is an underdose and has no action on the complex pathophysiology of dysmenorrhea.
- The additional NLRP3 inflammasome inhibitory action of mefenamic acid gives an edge and makes it an ideal choice in the treatment of dysmenorrhea marked with dysregulated NLRP3 inflammasome.
- Mefenamic acid is recommended as the first-choice drug in the treatment of dysmenorrhea. It can be given in a dosage of 500 mg twice or thrice a day, based on the patient’s requirement. It also provides the additional advantage of reducing the bleeding in associated cases of menorrhagia.
- In the case of adolescent patients, 250 mg thrice daily or 25 mg/kg/day of mefenamic acid in three divided doses is the recommended dosage.
- In case of inadequate pain relief, as may occur in moderate to severe cases, the combination of dicyclomine + mefenamic acid is recommended.
- The suggested dosage of the combination for adolescent and adult patients is 250/10 mg and 500 mg/20 mg, respectively. Adolescents with a weight of ≥60 kg can be administered the adult dosage.
|
Key Highlights
· Lack of awareness about dysmenorrhea and unmet need for social and emotional support; raises the need for spreading awareness amongst the community and health care professionals.
· Dysmenorrhea has a complex pathogenesis, involving release of prostaglandins, activation of NLRP3 inflammasome, and involvement of other mediators-bradykinin, histamine and acetylcholine.
· NSAIDs remain the mainstay in the management of dysmenorrhea and mefenamic acid is the most preferred choice.
· An ideal treatment would act on the different pathways causing dysmenorrheic pain and other associated symptoms.
· The combination of mefenamic acid and dicyclomine is the most preferred fixed-dose drug combinations (FDCs) because of its actions on the various pathways of dysmenorrhea and its synergism.
· Paracetamol is not effective as it has a weak analgesic effect and no anti-inflammatory actions.
|
Acknowledgments
The authors would like to acknowledge and appreciate Ms Pooja S Banerjee, for her medical writing support in developing the consensus.
Disclosure
The authors report no conflicts of interest in this work.
REFERENCES
- Barcikowska Z, Rajkowska-Labon E, Grzybowska ME, Hansdorfer-Korzon R, Zorena K. Inflammatory markers in dysmenorrhea and therapeutic options. Int J Environ Res Public Health. 2020;17(4):1191.
- Karout S, Soubra L, Rahme D, Karout L, Khojah HMJ, Itani R. Prevalence, risk factors, and management practices of primary dysmenorrhea among young females. BMC Women’s Health. 2021;21(1):392.
- Omidvar S, Bakouei F, Amiri FN, Begum K. Primary dysmenorrhea and menstrual symptoms in Indian female students: prevalence, impact, and management. Glob J Health Sci. 2016;8(8):53632.
- Itani R, Soubra L, Karout S, Rahme D, Karout L, Khojah HMJ. Primary dysmenorrhea: pathophysiology, diagnosis, and treatment updates. Korean J Fam Med. 2022;43(2):101-8.
- Rao KA, Chandra S, Kasture PN. Role of prostaglandins in pathogenesis of dysmenorrhea and place of mefenamic acid and dicyclomine in its management. Indian J Clin Pract. 2022;32(10):27-34.
- Kho KA, Shields JK. Diagnosis and management of primary dysmenorrhea. JAMA. 2020;323(3):268-9.
- Kannan J, Chandra S, Rao K, Kasture P. NLRP3 inflammasome in the pathogenesis of dysmenorrhea and role of mefenamic acid as its inhibitor. Indian J Clin Pract. 2022;32(12):25-7.
- Guimarães I, Póvoa AM. Primary dysmenorrhea: assessment and treatment. Rev Bras Ginecol Obstet. 2020;42(8):501-7. English.
- Azlan A, Salamonsen LA, Hutchison J, Evans J. Endometrial inflammasome activation accompanies menstruation and may have implications for systemic inflammatory events of the menstrual cycle. Hum Reprod. 2020;35(6):1363-76.
- Tang B, Liu D, Chen L, Liu Y. NLRP3 inflammasome inhibitor MCC950 attenuates primary dysmenorrhea in mice via the NF-κB/COX-2/PG pathway. J Inflamm (Lond). 2020;17:22.
- Kasture P, Mehta K, Gowda A. Inflammasome, inflammation, infection, and mefenamic acid. J Assoc Physicians India. 2022;70(3):11-2.
|
Contributors List
|
|
|
Anita Kant
|
Dept. of Obstetrics and Gynecology, Asian Institute of Medical Sciences, Faridabad, Haryana
|
|
Jayam Kannan
|
Dept. of Obstetrics and Gynecology, Garbba Rakshambigai Fertility Centre, Chennai, Tamil Nadu
|
|
Sunita Chandra
|
Dept. of Obstetrics and Gynecology, Rajendra Nagar Hospital & IVF Center and Morpheus Lucknow Fertility Center, Lucknow, Uttar Pradesh
|
|
Chandravati
|
Dept. of Obstetrics and Gynecology, Krishna Medical Centre, Lucknow, Uttar Pradesh
|
|
Bharti Kalra
|
Dept. of Obstetrics and Gynecology, Bharti Hospital, Karnal, Haryana
|
|
Helen Mary
|
Dept. of Obstetrics and Gynecology, St. John's Medical College Hospital, Bengaluru, Karnataka
|
|
Prabhu Kasture
|
Deputy Director-Medical Services and Pharmacovigilance, Blue Cross Laboratoires Pvt Ltd., Mumbai, Maharashtra
|
|
Bharati Rajshekhar
|
Dept. of Obstetrics and Gynecology, Vaatsalya Hospital, Hassan, Karnataka
|
|
Anshu Jindal
|
Dept. of Obstetrics and Gynecology, IVF Specialist, Jindal Hospital and Fertility Center, Meerut, Uttar Pradesh
|
|
Amuthambigai
|
Dept. of Obstetrics and Gynecology, Uma Hospital, Tindivanam, Tamil Nadu
|
|
Sachin Dalal
|
Dept. of Obstetrics and Gynecology, IVF Specialist, Madhu Hospital and Embrion IVF & Fortis Hospital, Mumbai, Maharashtra
|
|
Reema Jain
|
Dept. of Obstetrics and Gynecology, Laparoscopic Surgeon and Infertility Specialist, Vaishnavi Nursing Home, Gurugram, Haryana
|
|
Sujata Kulkarni
|
Dept. of Obstetrics and Gynecology, Seeta Nursing Home, Nashik, Maharashtra
|
|
Surekha Vinay
|
Dept. of Obstetrics and Gynecology, Infertility Specialist and Laproscopic Surgeon, Gipson Clinic, Kurnool, Andhra Pradesh
|
|
Ummamaheshwar Sindur
|
Dept. of Obstetrics and Gynecology, Laparoscopic Surgeon, Sindur Hospital, Vijayapura, Karnataka
|
|
Pragya Ojha
|
Dept. of Obstetrics and Gynecology, VAMA - Centre for Women Health, Varanasi, Uttar Pradesh
|
|
Renu Chakravarty
|
Dept. of Obstetrics and Gynecology, Chakravarty Nursing Home, Panchkula, Haryana
|
|
Harini
|
Dept. of Obstetrics and Gynecology, Chikkaballapur Institute of Medical Sciences, Karnataka
|
|
Archana Mayekar
|
Dept. of Obstetrics and Gynecology, VN Desai Municipal General Hospital, Mumbai, Maharashtra
|
|
Jayanta Kumar Gupta
|
Dept. of Obstetrics and Gynecology, Apollo Multispecialty Hospitals, Kolkata, West Bengal
|
|
Ruchika Garg
|
Dept. of Obstetrics and Gynecology, Garg Clinic, Lucknow, Uttar Pradesh
|
|
Tushar Palve
|
Dept. of Obstetrics and Gynecology, Cama and Albless Hospital, Mumbai, Maharashtra
|
|
Nithya Vaidya
|
Dept. of Obstetrics and Gynecology, Yashada Nursing Home and Lifewave Hospital, Mumbai, Maharashtra
|
|
Nupur Chandan
|
Dept. of Obstetrics and Gynecology, Surya Clinic, Dhanbad, Jharkhand
|
|
Sonam Kumari
|
Infertility Consultant, Aveta Test Tube Baby Centre, Jharkhand
|
|
Vaishali Chaudhary
|
Dept. of Obstetrics and Gynecology, Siddhivinayak Hospital, Jalgaon, Maharashtra
|
|