Abstract
India is the largest consumer of antibiotics in the world. High antibiotic consumption is linked to the emergence and community spread of multidrug-resistant bacteria. It is well-established that antibiotic overuse is one of the leading causes of antibiotic resistance, which is a major global public health challenge. Optimizing antibiotic usage is, thus, an essential issue. Before promoting and defining judicious antibiotic prescribing, it is crucial to analyze practitioners' diagnostic and prescribing practices. Hence, a nationwide retrospective questionnaire-based survey was conducted among 950 Indian doctors. This survey aimed to describe the approaches and practices of Indian doctors towards antibiotic use in upper respiratory tract infections (URTIs) and pyrexia of unknown origin (PUO) and compare practices with national guidelines. These are the most common reasons for primary health care consultations and significantly contribute to the overuse of antibiotics. According to the survey, amoxicillin-clavulanic acid remains the first-line antibiotic for URTI treatment. Third-generation cephalosporins were found to be the most prescribed antibiotics for PUO, uncomplicated typhoid and infections during pregnancy. Our survey results show that most of the clinicians in our study were well aware of the guidelines for antimicrobial use issued by Indian Council of Medical Research (ICMR) and the nationwide problem of antimicrobial resistance. This study provides an important insight into the prescribing practices of antibiotics among Indian doctors.
Keywords: Antibiotic usage, URTI, PUO, Questionnaire survey
High antibiotic consumption is linked to the emergence and spread of multidrug-resistant bacteria in the community.1 The number of deaths in Asia due to antimicrobial resistance (AMR) is projected to rise to 4.7 million by 2050.2 It is well-established that antibiotic misuse is one of the primary causes of antibiotic resistance; therefore, optimizing antibiotic usage is an urgent necessity. India is the largest consumer of antibiotics in the world, consuming 13 billion standard units in 2010; its per capita usage climbed by 66% between 2000 and 2010.3 Antibiotic overprescribing in primary care is mostly driven by patient expectations and demands for treatment. Other examples of inappropriate use include, prescribing a non–first-line antibiotic or an antibiotic with broad-spectrum activity for an infection that may be treated with a narrow-spectrum antibiotic.
Upper respiratory tract infection (URTI) is the most common reason for primary health care consultations and significantly contributes to the overuse of antibiotics. Primary care doctors reportedly write more than 90% of antibiotic prescriptions.1 Viruses cause most URTIs, and antibiotics neither accelerate recovery nor prevent complications. Favipiravir has been used as an empirical therapy for influenza-like illness during coronavirus disease 2019 (COVID-19) pandemic.4
Clinical manifestations of URTIs range from mild catarrhal symptoms of the upper respiratory tract to influenza-like symptoms, streptococcal angina, laryngotracheal bronchitis and lower respiratory tract symptoms. About 5% to 15% of instances of tonsillopharyngitis are caused by Group A beta-hemolytic Streptococcus (GABHS), which, according to evidence-based medicine (EBM) and national and international guidelines, necessitates the use of antibiotics to shorten the duration of the disease and lower the risk of sequelae.5
Antibiotic overuse and overprescribing in outpatient treatment may also be attributable to prescribing antibiotics for pyrexia of unknown origin (PUO) before test findings show a bacterial illness. As per ICMR, PUO is defined as “previously healthy (nonimmunosuppressed) community (urban or rural) dwelling adult (ages 19-64 years) reporting no previous medical illness or recent hospitalization (in the preceding 30 days) presenting with acute onset of fever >38.3°C (101.0°F) for >2 days and lasting up to 14 days and having received no specific treatment for this current illness with antimalarials or antibiotics”. It is associated with no organ-specific symptoms at onset.2 These primarily include malaria, arboviral infections (such as dengue), enteric fever and viral and zoonotic bacterial diseases (such as scrub typhus and leptospirosis). PUO at the public primary health care level is associated with a high rate of the previous self-treatment, diagnostic considerations that are not very specific, polypharmacy and a high rate of antibiotic prescriptions. It includes tropical infectious diseases like dengue, scrub typhus, murine typhus, leptospirosis and enteric fever, which continue to contribute substantially to the febrile disease burden throughout Southeast Asia. The paucity of data and access to affordable, sensitive and specific diagnostic methods hinder targeted and effective treatment of severe acute infections and lead to further development of AMR.6
A stewardship program adopting rational antibiotic use is required to limit irrational antibiotic use. Antimicrobial stewardship is an urgent necessity and the only proven approach to avoid human overuse and abuse of antimicrobials, one of the primary causes of AMR. In May 2015, the World Health Assembly passed a resolution endorsing a global AMR action plan.2
It is important to consider clinicians’ diagnostic and prescribing practices before encouraging and delineating prudent antibiotic prescribing. This study was conducted among doctors to assess their clinical practices and preferred antibiotic options for URTI, fever of unknown origin, typhoid, infections in pregnancy.
METHODS
Study Design and Setting
The study was a retrospective study conducted via a questionnaire-based survey among all antimicrobial-prescribing doctors between May and September 2022. It is a mixed-method study to identify targets for quality improvement in antibiotic-prescribing practices.
Study Respondents
All qualified doctors (General Practitioners, Pediatricians, Physicians and ENTs) practicing modern medicine were eligible for the survey and were approached to participate. The survey intended to include doctors across all clinical departments, work experience and professional hierarchy to ensure the representation of different antibiotic practices.
In this study, 950 Indian doctors prescribing antibiotics participated in the questionnaire survey on treatment options for URTI and fever of unknown origin and practice toward antibiotic usage.
Data Collection
The survey tool, in the form of a closed-ended questionnaire, was developed by an expert panel comprising a physician, a microbiologist and a pharmacologist. The questionnaire was specifically focused on the study aims and tailored contextually to fit local situations.
RESULTS AND DISCUSSION
Antibiotic Use in URTI
The choice of antibiotics as first-line treatment is represented in Figure 1 and Figure 2. About 39% of the respondent clinicians chose the combination of amoxicillin-clavulanic acid as the first-line antibiotic for URTI treatment. They chose penicillins, cephalosporins, macrolides and combinations thereof as an option for treating URTI. The indication of the first-line antimicrobial choice however varies according to the established etiology. In this study, most respondents adhered to the guidelines with slight modifications depending on etiology. In 1993, Neu et al reported a literature review of 415 publications detailing the clinical use of amoxicillin-clavulanic acid spanning 1979 to 1992. A total of over 38,500 patients were treated with this combination, and clinical efficacy rates (cure or improved) were 88% and 92% in comparative and uncontrolled trials, respectively.7 However, there was an increase in the usage of specific antibiotics during the COVID-19 pandemic period. According to a survey conducted in Egypt, the most often prescribed antibiotics in 2019 were third-generation cephalosporins (42.9%), combinations of penicillins, including beta-lactamase inhibitors (16.51%), fluoroquinolones (15.25%) and carbapenems (9.16%). In 2020, the most often prescribed antibiotics were third-generation cephalosporins (30.47%), macrolides (15.12%), fluoroquinolones (14.74%) and amoxicillin-clavulanic acid, including beta-lactamase inhibitors (12.68%).8
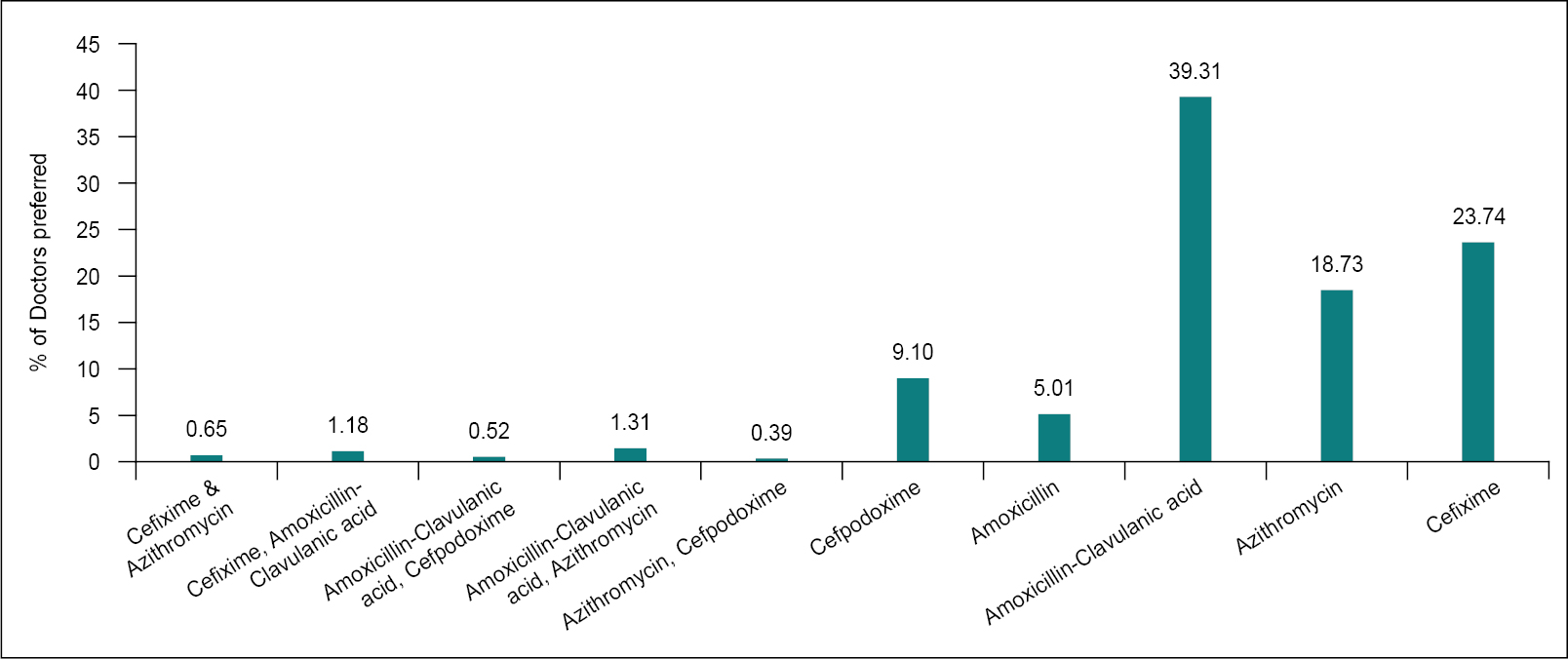
Figure 1. Percentage of antibiotics chosen as first-line for treatment of URTIs.
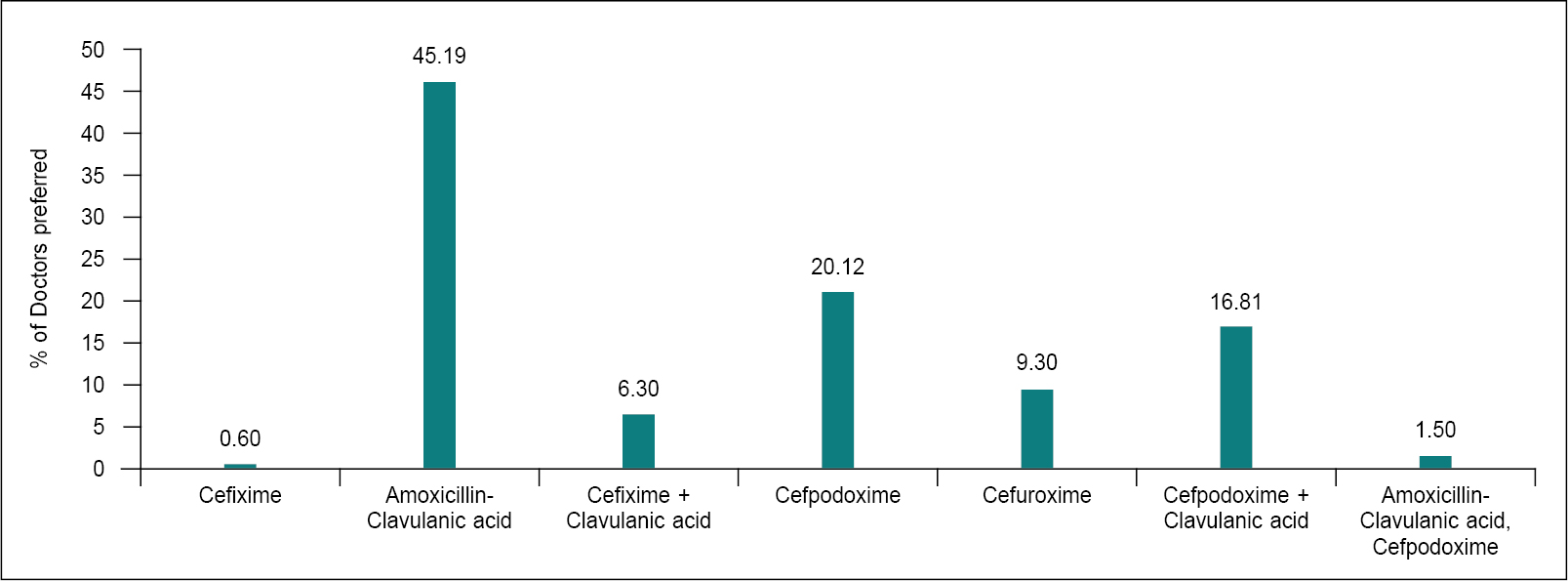
Figure 2. Preferred antibiotics in moderate URTIs.
URTIs are the most common reason for antibiotic prescription in an outpatient department (OPD). Several factors, like self-medication and pressure on providers by patients to prescribe or dispense antibiotics, etc., influence the high and unjustified antibiotic use in a common ailment. URTIs include rhinosinusitis, acute otitis media (AOM), pharyngotonsillitis and laryngitis.9 Viruses are responsible for the great majority of URTIs. Therefore, antimicrobial treatment is not always required.10 Paracetamol and ibuprofen are considered as the standard analgesics.11
The 2019 Treatment Guidelines for Antimicrobial Use in Common Syndromes issued by Indian Council of Medical Research (ICMR) recommend amoxicillin or amoxicillin-clavulanic acid (alternative) as first-line antibiotic therapy for AOM, amoxicillin or amoxicillin-clavulanic acid for sinusitis and penicillin or amoxicillin for pharyngitis. First-line therapy represents the initial antibiotics recommended for treating patients without drug allergies, and the most common class of non–first-line antibiotics prescribed was macrolides for pharyngitis and cephalosporins for sinusitis and otitis.2
Amoxicillin-clavulanic acid provides improved coverage for beta-lactamase-positive Haemophilus influenzae and drug-resistant Streptococcus pneumoniae. Alternatives (e.g., doxycycline, levofloxacin) are used only for patients allergic to amoxicillin-clavulanic acid. Alternative initial antibiotics include cefuroxime or cefpodoxime, respectively.2 For children, the second-line antibiotics include a high dose of amoxicillin-clavulanic acid combination, levofloxacin and moxifloxacin or the combination of clindamycin plus a third-generation oral cephalosporin (cefixime or cefpodoxime).
Pneumococcal resistance in nonmeningeal isolates is very low in India; hence, standard doses of amoxicillin generally suffice. Conversely, pneumococcal resistance to co-trimoxazole and macrolides is widespread.12
Antibiotic Use in Pyrexia of Unknown Origin
Around 56.5% of respondents chose cefixime as the first-line of treatment for PUO, followed by amoxicillin-clavulanic acid (18%), cefpodoxime (12%) and azithromycin (11%) (Fig. 3). Literature suggests that empirical therapy with a third-generation cephalosporin (ceftriaxone) and doxycycline or azithromycin is appropriate, while diagnostic confirmation is awaited.12
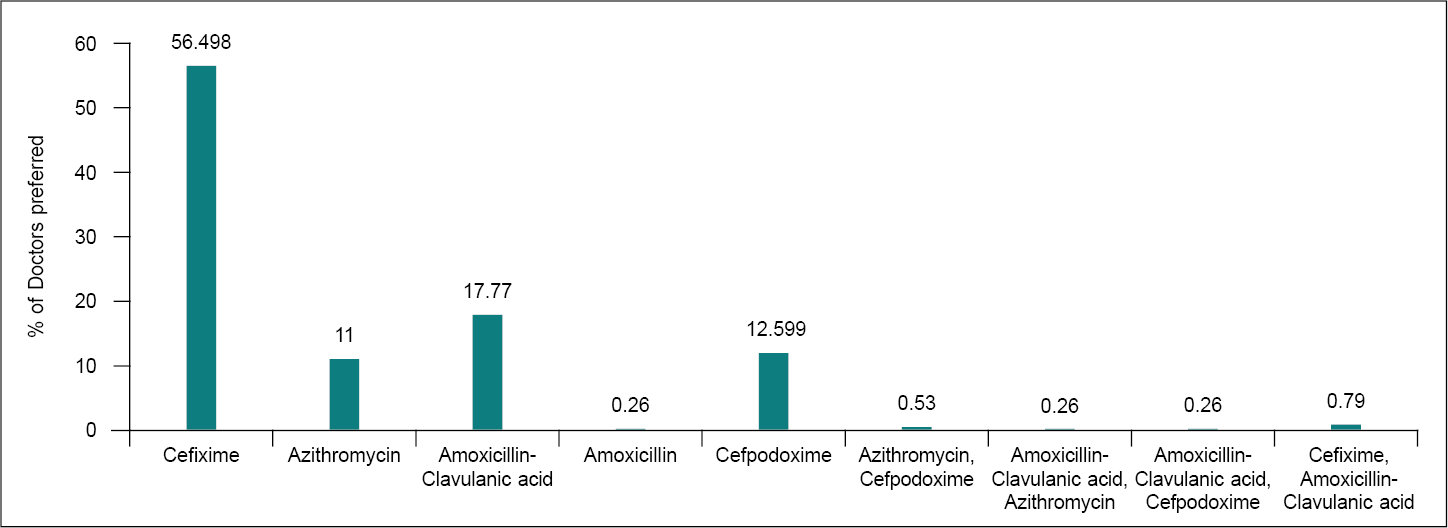
Figure 3. Choice of antibiotic in pyrexia of unknown origin.
Although azithromycin sensitivity testing has not been verified, the response has been positive in the majority of clinical studies.2
However, defervescence periods with third-generation cephalosporins are much longer compared to other classes, and bone marrow suppression is a problem with chloramphenicol. Enterobacteriaceae (Escherichia coli and Klebsiella) are increasingly resistant to quinolones (up to 80%) and third-generation cephalosporins (up to 75% due to extended-spectrum beta-lactamases [ESBL] development). Initial empiric therapy for infections caused by these organisms, pyelonephritis, severe intra-abdominal infections (IAI), etc., should consist of a drug active against ESBL producers, such as a carbapenem or a beta-lactam/beta-lactamase inhibitor for less severely ill patients. Community-acquired organisms such as Staphylococcus aureus are usually susceptible to methicillin, i.e., standard anti-staphylococcal drugs such as cloxacillin and first cephalosporins may be used. Penicillin still remains the drug of choice for pneumococcal infection.13,14
Antibiotic Use in Uncomplicated Typhoid or Paratyphoid
Antibiotic therapy shortens the clinical course of enteric fever and reduces the risk of death. The majority of survey respondents selected cephalosporins and fluoroquinolones as the conventional treatment for uncomplicated typhoid and paratyphoid (Fig. 4). Fluoroquinolones (such as ciprofloxacin) are often used for the empiric treatment of enteric fever in adults and are considered the treatment of choice for fluoroquinolone-susceptible infections. Most of the Salmonella typhi and paratyphi A infections in South Asia are found to be fluoroquinolone-nonsusceptible or nalidixic acid-resistant, which suggests that treatment failures may occur among patients treated empirically with fluoroquinolones. Resistance to quinolones is high, reaching up to 69% for S. typhi and 23% for S. paratyphi A. Trimethoprim/sulfamethoxazole, chloramphenicol and third-generation cephalosporins have modest resistance rates.2
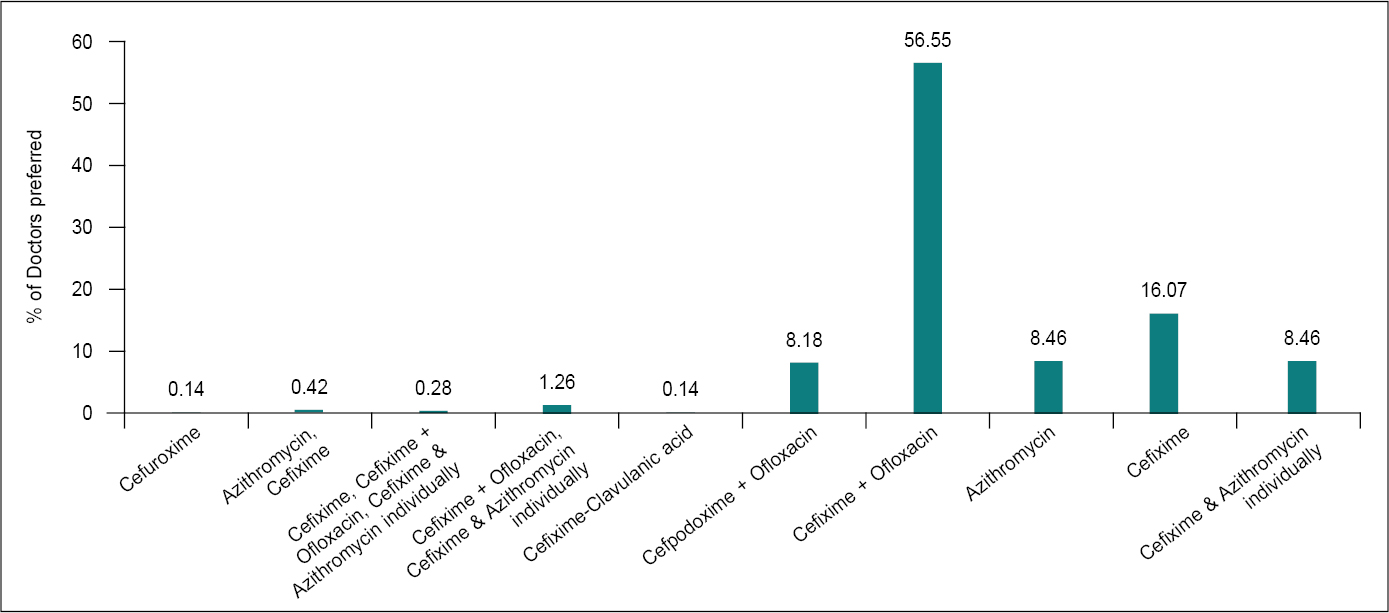
Figure 4. Preference of antibiotics in uncomplicated typhoid or paratyphoid.
Cephalosporins are a large family of antimicrobial medicines, which are commonly used to treat a variety of infectious diseases. Individual cephalosporins (such as cefixime and ceftriaxone) differ in the pathogens they are able to treat, how they are administered (orally or intravenously), and when they are produced. S. typhi and S. paratyphi A, B and C, the bacteria that cause enteric (typhoid) fever, can be treated with certain cephalosporins.15
There is limited evidence on the superiority of azithromycin over first-line antibiotics, fluoroquinolones and cephalosporins, even when used in people infected with multidrug-resistant or nalidixic acid-resistant strains of S. typhi or S. paratyphi or both. Available evidence shows that azithromycin is as good as the other comparator drugs for most outcomes and is better than fluoroquinolones in terms of reducing clinical failure and duration of hospital stay and ceftriaxone in terms of reducing relapse. Taking into consideration, the potential for the development of resistance to any new antibiotic introduced, azithromycin is used guardedly to prevent the emergence of strains resistant to the drug.12
Effectiveness of Antibiotic Combinations with ESBL Inhibitors
Antimicrobial combinations serve a crucial role in clinical therapies despite the inherent disadvantages of utilizing numerous medicines. The clinical benefits of antimicrobial combinations in certain situations have been appreciated for decades. Our survey strongly supports this (Fig. 5). Fifty-six percent of clinicians believed that the combination of an antibiotic with an ESBL inhibitor is beneficial. Beta-lactamase inhibitors (e.g., clavulanic acid, sulbactam, tazobactam) protect beta-lactams from enzymatic hydrolysis, enhancing their spectrum of activity against various ESBLs. A stronger bactericidal impact may be achieved by combining cell wall-active medicines with aminoglycosides, beta-lactams with inhibitors of beta-lactamase, and medications that inhibit different steps along a key metabolic pathway. The rationale is that combination therapy may be advantageous when resistance to a single agent develops rapidly.13
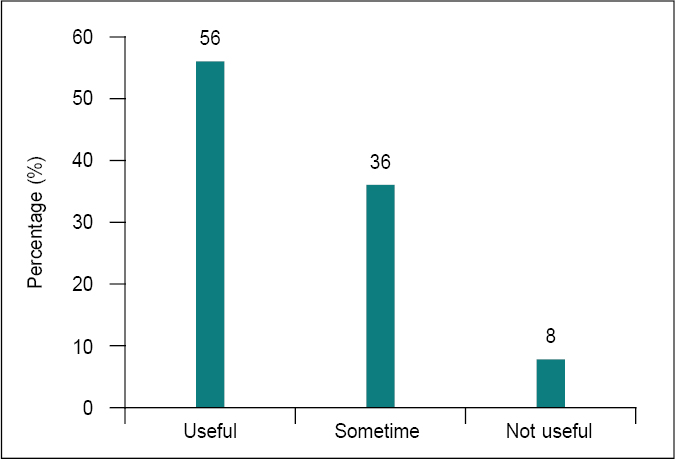
Figure 5. Effectiveness of antibiotic combination with ESBL inhibitor.
AMR poses an enormous global public health challenge. Beta-lactamase inhibitors are medications used ubiquitously in modern medicine because they can combat bacterial AMR mechanisms. Therefore, careful monitoring and prescribing of patients taking beta-lactamase inhibitors in combination with beta-lactam antimicrobials are paramount.13,15
Antibiotics to Manage Infections in Pregnancy
It is common for antibiotics to be prescribed during pregnancy for infections. Treating infections during pregnancy is important as untreated infections can have adverse effects on the mother and baby. However, the type of medication must be selected with care. Certain antibiotics are safe to take while pregnant, while others are not. Penicillins, most cephalosporins and azithromycin are considered first-choice options during pregnancy. In this survey, most of the clinicians preferred cefixime as the first choice of antibiotic to manage infections in pregnancy (Fig. 6).16 Examples of penicillin antibiotics include amoxicillin and ampicillin. Health care providers may also choose to prescribe the combination of amoxicillin-clavulanic acid. This is also considered a safe antibiotic during pregnancy. It can be used for infections where amoxicillin may not work well enough on its own.17
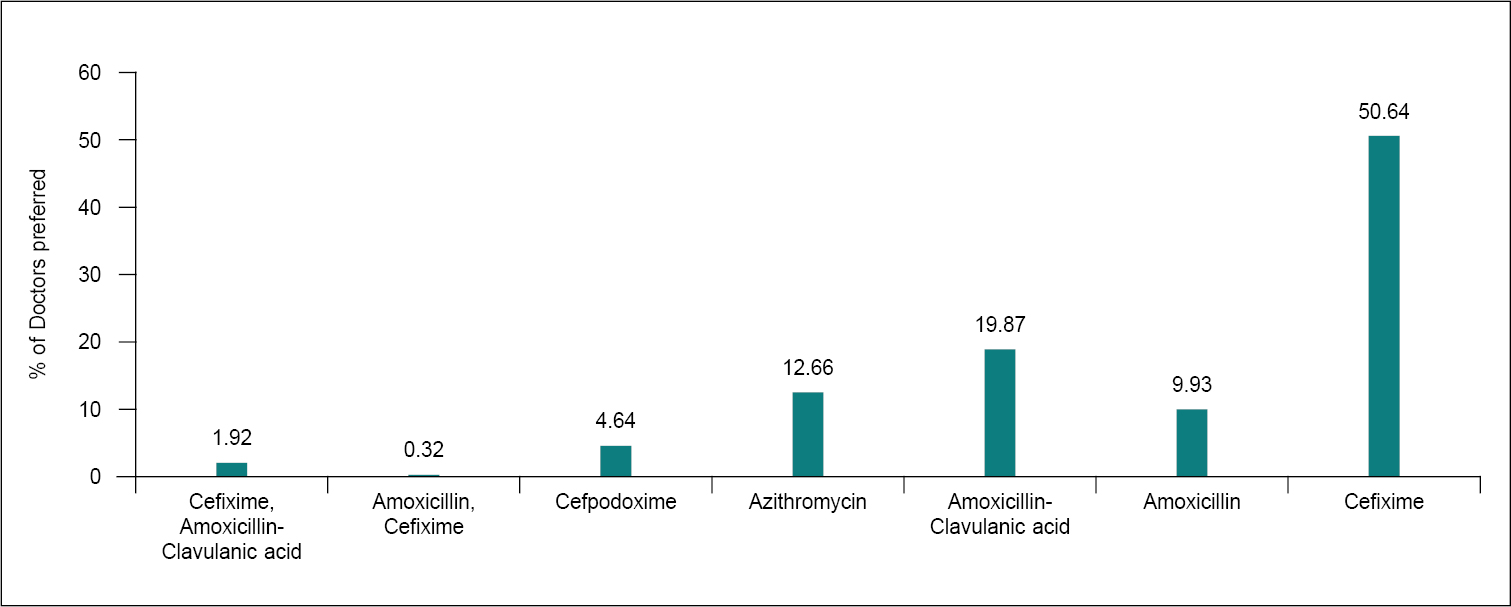
Figure 6. Prescribed antibiotics to manage infections in pregnancy.
Multivitamins and Probiotics along with Antibiotic
Most of the clinicians (Fig. 7) in our study agreed that use of multivitamins together with antibiotics is considerable. Thirty-seven percent of clinicians favored recommending multivitamins with antibiotics, whereas 54% of professionals recommended it occasionally. Literature suggests that along with medications, additional vitamin C, vitamin D and niacin can also help treat infections. In a blinded clinical trial, intravenous vitamin C at 1,20,000 mg/day accelerated burned skin recovery. Niacin, 1,000 to 3,000 mg/day, lowers cholesterol. Doctors treating life-threatening bacterial infections have various vitamin C and niacin choices. Oral supplementation with 2,000 to 10,000 mg of vitamin C and 100 to 500 mg of time-release or “no flush” niacin is easiest.18,19 Along with medications, additional vitamin C, vitamin D and niacin can help treat respiratory infections at home. In 2005, another study found that high doses of ascorbic acid reduced antibiotic resistance in Pseudomonas aeuruginosa. Taking vitamin C, kanamycin, streptomycin and tetracyclines prevents bacterial respiratory illness.19
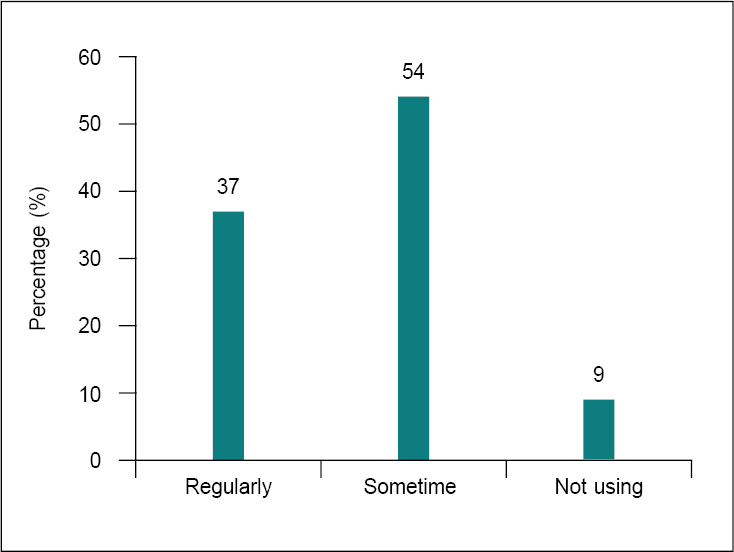
Figure 7. Frequency of using multivitamins along with antibiotics.
Antibiotics may kill a huge number of disease-causing microorganisms. However, they also frequently kill a large number of beneficial bacteria and can cause a variety of side effects, including diarrhea.20-22 These potential issues may be mitigated by probiotic supplementation. Even in the recent survey, most clinicians favored the combination of probiotics and antibiotics (57%), but only a few (30%) supported combining probiotics and antibiotics sometimes (Fig. 8). Probiotics do not render antibiotics ineffective. A randomized clinical trial investigated the efficacy of probiotic administration in addition to antibiotics. The study reported that probiotics have the ability to prevent and treat some infections.23
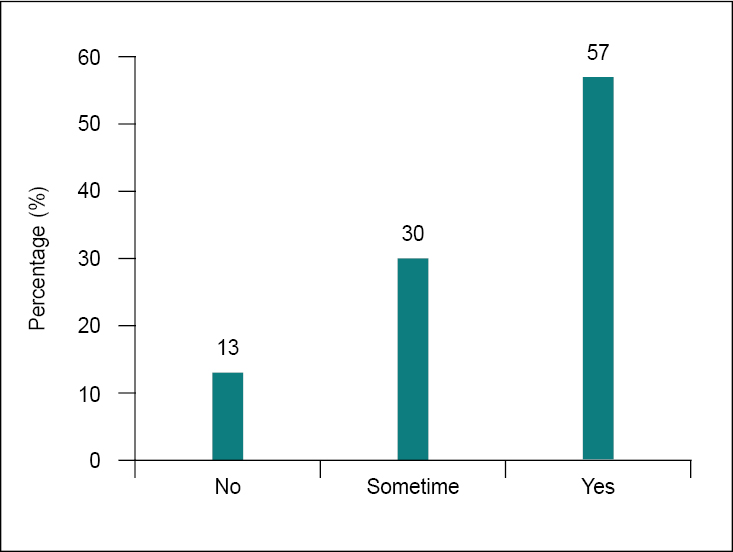
Figure 8. Frequency of co-prescribing probiotics with amoxicillin-clavulanic acid.
Efficient use of probiotics can potentially decrease patients’ exposure to antimicrobials. As antibiotics are frequently not very selective killers, the requirement and significance of consuming probiotics while taking antibiotics has been aptly described.22,24
CONCLUSION
The emergence of bacterial strains resistant to antimicrobial agents presents a growing concern worldwide. Among other factors, the irrational use of antibiotics has contributed to the progressive loss of bacterial sensitivity to antibiotics and the spreading of resistant strains of bacteria, with substantial clinical and economic impact. The clinical effectiveness of antibiotics depends partially on their correct use, depending on patients, doctors and retailers.
As per our survey, cefixime is the choice of antibiotic in PUO. In uncomplicated typhoid and paratyphoid, cefixime-ofloxacin is preferred. Amoxicillin-clavulanic acid remains the first-line of treatment for URTI. A combination of ESBL inhibitors with antibiotics is effective and preferable due to its positive role in combating the development of AMR.
Hence, it can be concluded that most of the clinicians in our study were well aware of the ICMR guidelines and the nationwide problem of AMR. Also, it provides important insight into the knowledge and adherence to the guidelines and practices regarding antibiotic resistance and usage among health care practitioners.
Acknowledgment
The authors thank Aurobindo Pharma Ltd. for their technical support in the preparation of this manuscript. The authors would also like to thank all the doctors who participated in the survey.
Conflict of Interest
The author has no conflict of interest to declare for this publication.
REFERENCES
- Van de Sande-Bruinsma N, Grundmann H, Verloo D, Tiemersma E, Monen J, Goossens H, et al.; European Antimicrobial Resistance Surveillance System Group; European Surveillance of Antimicrobial Consumption Project Group. Antimicrobial drug use and resistance in Europe. Emerg Infect Dis. 2008;14(11):1722-30.
- Aggarwal S, Walia K, Madhumathi J, Gopalkrishnan R, Ohri VC, et al. Treatment guidelines for antimicrobial use in common syndromes. 2nd edition; Indian Council of Medical Research: New Delhi, India; 2019.
- Davey P, Sneddon J, Nathwani D. Overview of strategies for overcoming the challenge of antimicrobial resistance. Expert Rev Clin Pharmacol. 2010;3(5):667-86.
- Thanesekaraan V, Samaria JK, Kant S, Koul P, Mishra N, Tampi PS, et al. Consensus statement on: favipiravir as an empirical therapy for influenza-like illness during COVID-19 pandemic. J Indian Med Assoc. 2020;118(9):70-6.
- Filipetto FA, Modi DS, Weiss LB, Ciervo CA. Patient knowledge and perception of upper respiratory infections, antibiotic indications and resistance. Patient Prefer Adherence. 2008;2:35-9.
- Wangrangsimakul T, Althaus T, Mukaka M, Kantipong P, Wuthiekanun V, Chierakul W, et al. Causes of acute undifferentiated fever and the utility of biomarkers in Chiangrai, northern Thailand. PLoS Negl Trop Dis. 2018;12(5):e0006477.
- Neu HC, Wilson AP, Grüneberg RN. Amoxycillin/clavulanic acid: a review of its efficacy in over 38,500 patients from 1979 to 1992. J Chemother. 1993;5(2):67-93.
- Hussein RR, Rabie ASI, Bin Shaman M, Shaaban AH, Fahmy AM, Sofy MR, et al. Antibiotic consumption in hospitals during COVID-19 pandemic: a comparative study. J Infect Dev Ctries. 2022;16(11):1679-86.
- Zeng L, Zhang L, Hu Z, Ehle EA, Chen Y, Liu L, et al. Systematic review of evidence-based guidelines on medication therapy for upper respiratory tract infection in children with AGREE instrument. PLoS One. 2014;9(2):e87711.
- Hersh AL, Jackson MA, Hicks LA; American Academy of Pediatrics Committee on Infectious Diseases. Principles of judicious antibiotic prescribing for upper respiratory tract infections in pediatrics. Pediatrics. 2013;132(6):1146-54.
- Perrott DA, Piira T, Goodenough B, Champion GD. Efficacy and safety of acetaminophen vs ibuprofen for treating children’s pain or fever: a meta-analysis. Arch Pediatr Adolesc Med. 2004;158(6):521-6.
- Bhargava A, Ralph R, Chatterjee B, Bottieau E. Assessment and initial management of acute undifferentiated fever in tropical and subtropical regions. BMJ. 2018;363:k4766.
- Crump JA, Sjölund-Karlsson M, Gordon MA, Parry CM. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev. 2015;28(4):901-37.
- Eliopoulos GM, Moellering RC Jr. Antibiotic synergism and antimicrobial combinations in clinical infections. Rev Infect Dis. 1982;4(2):282-93.
- Ferrara P, Cutrona C, Sbordone A. Which treatment for upper respiratory tract infections? Ital J Pediatr. 2015;41(Suppl 2):A31.
- Niebyl JR, Simpson JL. Chapter 8: Drugs and environmental agents in pregnancy and lactation: embryology, teratology, epidemiology. Obstetrics: Normal and Problem Pregnancies. Elsevier; 2007. pp. 184-214.
- Bracken MB, Holford TR. Exposure to prescribed drugs in pregnancy and association with congenital malformations. Obstet Gynecol. 1981;58(3):336-44.
- Santos F, Oraichi D, Bérard A. Prevalence and predictors of anti-infective use during pregnancy. Pharmacoepidemiol Drug Saf. 2010;19(4):418-27.
- Cursino L, Chartone-Souza E, Nascimento AMA. Syergic interaction between ascorbic acid and antibiotics against Pseudomonas aeruginosa. Brazilian Arch Biol Technol. 2005;48(3):379-84.
- Linday LA, Dolitsky JN, Shindledecker RD. Nutritional supplements as adjunctive therapy for children with chronic/recurrent sinusitis: pilot research. Int J Pediatr Otorhinolaryngol. 2004;68(6):785-93.
- Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behavior. Nat Rev Neurosci. 2012;13(10):701-12.
- Rao M, Duseja A. Probiotics in health and disease: a review. J Ayurv Integrat Med. 2015;6(1):7.
- Butler CC, Lau M, Gillespie D, Owen-Jones E, Lown M, Wootton M, et al. Effect of probiotic use on antibiotic administration among care home residents: a randomized clinical trial. JAMA. 2020;324(1):47-56.
- Heidari F, Abbaszadeh S, Mirak SEM. Evaluation effect of combination probiotics and antibiotics in the prevention of recurrent urinary tract infection (UTI) in women. Biomed Pharmacol J. 2017;10(2).