Published in IJCP
April 2023
Guest Editorial
Gauging the Antiobesity Potential of Oral Semaglutide in Type 2 Diabetes in the Indian Context
April 10, 2023 | Awadhesh Kumar Singh
Diabetes & Endocrinology

Abstract
“Diabesity”, which combines type 2 diabetes (T2D) and obesity has emerged as a global epidemic in the modern world. Indeed, diabesity contributes significantly to increased morbidity, and the prevalence of this global health burden has steadily increased in India over the past three decades. Importantly, only a few pharmacological agents are currently available across the world, and India in particular has access to only a limited number of such agents. However, with the advent of oral glucagon-like peptide-1 receptor agonist (GLP-1RA) after its launch in India recently as an antidiabetic agent, the horizon to combat diabesity has expanded. With the available evidence from several phase 3 trials of oral GLP-1RA (PIONEER, Peptide Innovation for Early Diabetes Treatment), it is increasingly becoming apparent that oral semaglutide could have some potential to fulfil the US Food and Drug Administration criterion of an antiobesity agent.
Keywords: Obesity, oral GLP-1RA, semaglutide, weight loss
Type 2 diabetes (T2D) is one of the largest global health emergencies of this century, ranking among the 10 leading causes of mortality along with cardiovascular disease, respiratory disease and cancer.1,2 According to the World Health Organization (WHO), noncommunicable diseases (NCDs) accounted for 74% of deaths globally in 2019, of which, diabetes resulted in 1.6 million deaths, thus becoming the 9th leading cause of death globally.2 Diabetes has steadily increased around the world including in India over the last decades and India’s contribution to the global burden is markedly increasing the overall disease burden and mortality.3 The past three decades have witnessed a global increase in overweight (body mass index [BMI] ≥25 kg/m2) from 28.8% to 36.9% in men and from 29.8% to 38.0% in women.4 Similarly, age-standardized obesity (BMI ≥30 kg/m2) has increased from 3.2% to 10.8% in men and from 6.4% to 14.9% in women.5 Studies from different parts of India have provided evidence of the rising prevalence of overweight/obesity, and indeed overweight/obesity has been found to be the most important contributor to the rising prevalence of diabetes in the country.6 Obesity is associated with a decreased life expectancy of 5 to 20 years depending upon its duration both due to the magnitude of excess weight and the emergence of associated comorbid diseases. Besides, obesity increases the prevalence of psychological, neurological, pulmonary, gastrointestinal, renal, musculoskeletal and endocrine diseases.7 To this end, antiobesity medications, when combined with lifestyle intervention, produce larger weight loss than behavioral treatment alone.8 Available evidence clearly suggests that even a modest loss of 5% to 10% is often associated with a significant reduction in obesity-related complications and increases the quality of life.9
In 2007, the United States Food and Drug Administration (US FDA) laid down certain criteria to qualify as an effective antiobesity drug. The criteria require at least one of the following to be met after 1 year of treatment. First, the “mean” efficacy criterion wherein the difference in mean weight loss between the active product and placebo-treated groups is ≥5%, and the difference should be statistically significant. Second is the “categorical” efficacy criterion where the proportion of subjects losing ≥5% of baseline body weight in the active-product group should be ≥35%, and it should be approximately double the proportion in the placebo-treated group, and the difference between groups should be statistically significant.10 To date, the US FDA has approved six pharmacological agents for chronic weight management as an adjunct to a reduced-calorie diet and increased physical activity. These include orlistat, phentermine/topiramate ER, naltrexone ER/bupropion ER, high-dose liraglutide (3.0 mg), injectable high-dose semaglutide (2.4 mg) and recently a dual glucose-dependent insulinotropic polypeptide (GIP) receptor and glucagon-like peptide-1 receptor agonists (GLP-1RAs), injectable tirzepatide (5 mg, 10 mg, 15 mg).11
On the other hand, the European Medicines Agency (EMA) has approved all of them except phentermine/topiramate and tirzepatide. Orlistat, phentermine/topiramate ER, naltrexone ER/bupropion ER and liraglutide 3.0 mg produce placebo-subtracted weight losses varying from 2.6 kg (orlistat) to 8.8 kg (phentermine-topiramate) at 1 year.12,13 Interestingly, among the six approved antiobesity drugs, randomized controlled trials (RCTs) of only tirzepatide, semaglutide 2.4 mg and phentermine + topiramate have so far fulfilled both the “mean efficacy” and “categorical efficacy” criteria of antiobesity drugs across all the phase 3 trials, as per US FDA mandate of 2007. For liraglutide 3.0 mg, 3 out of the total of 4 RCTs fulfilled both the “mean efficacy” and “categorical efficacy” criteria. Notably, only 1 RCT of naltrexone + bupropion (COR-II) fulfilled both the “mean” and “categorical” efficacy criteria, while none of the RCTs of orlistat and lorcaserin (earlier approved but recently withdrawn) fulfilled both criteria. Thus, it is apparent that some of these antiobesity drugs that only fulfilled the “categorical” efficacy criterion have also received US FDA approval as antiobesity drugs for long-term use.13,14
GLP-1RAs have been the most effective weight-lowering agents in people with T2D. This finding led to examination of higher strength GLP-1RA formulations in people with obesity regardless of T2D. Surprisingly, a significant reduction in body weight was noted. For example, injectable liraglutide 0.6-1.8 mg once daily is approved for the treatment of T2D, while liraglutide 3.0 mg is approved for obesity regardless of T2D. Similarly, injectable semaglutide 1.0 mg, a once-weekly subcutaneous GLP-1RA was first approved as a treatment for T2D in 2017, while higher strength injectable semaglutide 2.4 mg once-weekly has been approved for obesity by the US FDA, United Kingdom Medicine and Health Products Regulations Agency, and EMA in 2021 and 2022, respectively.12 Notably, the reduction in body weight was quite larger with injectable semaglutide 1.0 mg (-6.5 kg) compared to dulaglutide 1.5 mg (-3.0 kg) (Estimated treatment difference [ETD] -3.55 kg; 95% confidence interval [CI], -4.32 to -2.78 kg; p < 0.0001) in SUSTAIN-7 (Semaglutide Unabated Sustainability in Treatment of Type 2 Diabetes) trial.15 Higher strength semaglutide 2.4 mg lowered mean body weight to approximately 15% after 68 weeks of treatment (relative to about 2.4% in placebo).16 Tirzepatide at the dose of 10 mg and 15 mg dose led to a nearly 20% weight reduction in SURMOUNT-1.14
Sodium-glucose co-transporter 2 (SGLT2) inhibitors are another class of antidiabetic agents that has shown a modest weight loss in T2D. However, weight loss with SGLT2 inhibitors has been generally lesser compared to GLP-1RAs. For example, in the SUSTAIN-8 trial, once-weekly injectable semaglutide 1.0 mg had a significantly greater weight loss compared with canagliflozin 300 mg once daily (ETD -1.06 kg; 95% CI, -1.76 to -0.36; p = 0.003).17 Similarly in the PIONEER-2 trial, oral semaglutide 14 mg daily had a higher weight loss compared with empagliflozin 25 mg daily at Week 52, as evaluated by the trial product estimand (-4.7 vs. -3.8 kg, respectively; ETD -0.9 kg; 95% CI, -1.6 to -0.2; p = 0.01).18 Unfortunately, neither the injectable semaglutide (1.0 mg
and 2.4 mg) nor tirzepatide is currently available in India. The currently available GLP-1RAs available in India that include the injectable form of dulaglutide (1.5 mg) and liraglutide (1.8 mg) have shown a significant but only modest weight loss in people with T2D. Thus, it is of interest to gauge the weight-lowering potential of oral semaglutide 14 mg available in India for the treatment of T2D.
The available evidence from some of the head-to-head, active comparator trials of GLP-1RAs (for example- AWARD-6 between liraglutide vs. dulaglutide, PIONEER-4 between oral semaglutide vs. injectable liraglutide, and PIONEER-10 between oral semaglutide 14 mg vs. dulaglutide 0.75 mg) suggest that amongst the three agents available in India, weight reduction is largest with oral semaglutide 14 mg followed by injectable forms of liraglutide 1.8 mg and dulaglutide 1.0 mg, respectively. Moreover, in PIONEER-1, 44% of patients on oral semaglutide 14 mg had ≥5% weight loss at 26 weeks compared with 16% of patients on placebo (trial-product estimand), which satisfies the US FDA “categorical” efficacy criterion (≥35% of participants on active drug having ≥5% weight loss). Since PIONEER-1 was conducted for only 26 weeks it did not fulfil the 52 weeks trial duration criteria laid down by the US FDA. However, in the PIONEER-4 trial, the percentage of patients with ≥5% weight loss at 52 weeks was 49.3% versus 12% in the placebo arm, alike SUSTAIN-1 of injectable semaglutide 1.0 mg once weekly (Fig. 1). These findings hint towards an antiobesogenic potential of oral semaglutide 14 mg similar to injectable semaglutide 1.0 mg. Indeed, both ≥5% and ≥10% weight reduction with oral semaglutide 14 mg is almost like the injectable semaglutide 1.0 mg, despite a relatively lesser baseline body weight in the former trial (Fig. 2). Finally, in a post-hoc analysis of PIONEER 1-5 and PIONEER-8 that evaluated the composite 3 (glycated hemoglobin [HbA1c] reduction ≥1% and body weight loss ≥5%), found 1% to 8% of patients on placebo, 11% on sitagliptin 100 mg, 18% on liraglutide 1.8 mg, 20% on empagliflozin 25 mg and 27% to 41% on oral semaglutide having achieved this composite. Moreover, the odds of achieving this composite 3 with oral semaglutide 14 mg were significantly (p < 0.0001) greater versus all other comparators.19
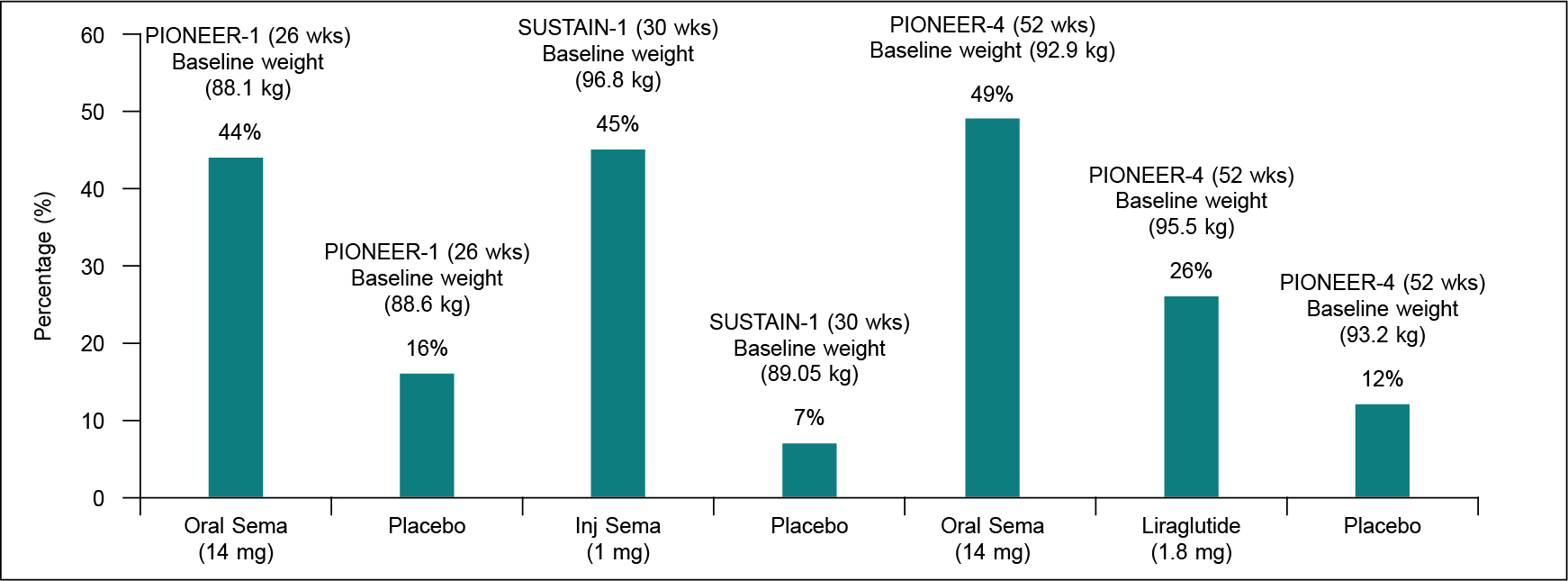
Figure 1. Comparative weight loss (≥5%) with oral (14 mg) and injectable semaglutide (1 mg) vs. placebo.
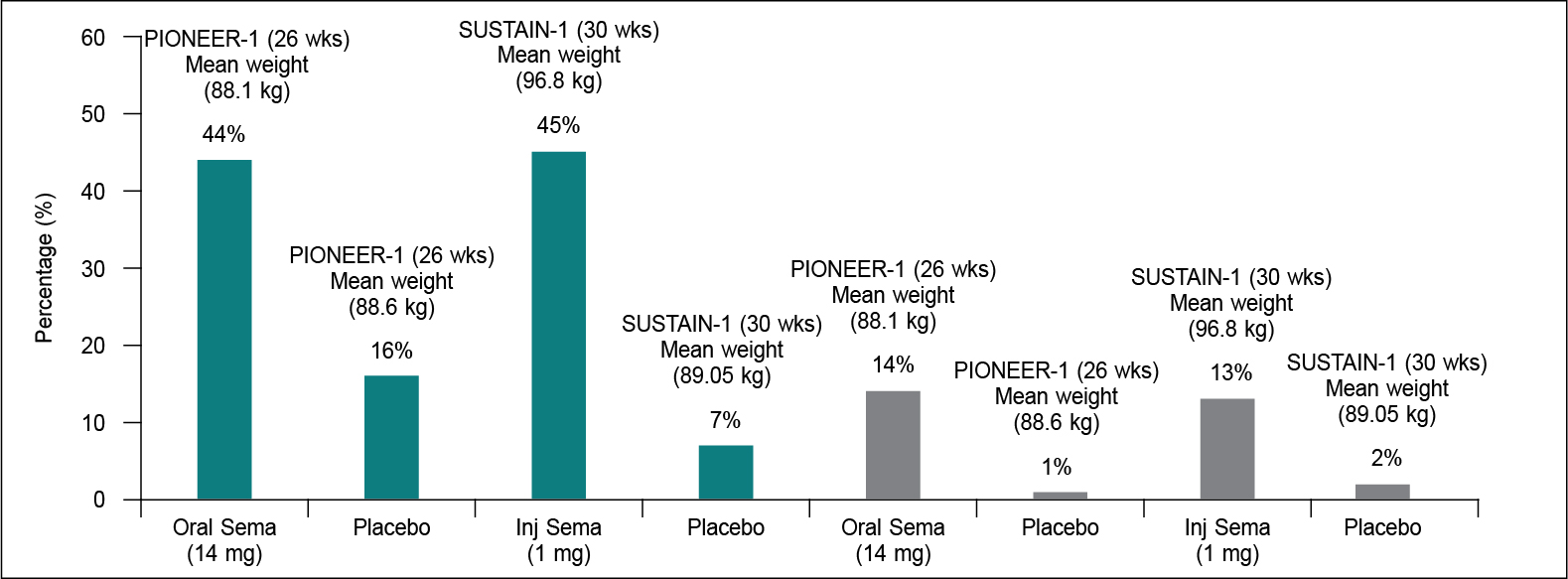
Figure 2. Comparative weight loss of ≥5% (green color) and ≥10% (grey color) with oral (14 mg) and injectable semaglutide (1 mg) vs. placebo.
In summary, oral semaglutide has been approved by the Drugs Controller General of India (DCGI) in 2020 and has been introduced in the Indian market in January 2022 as an antidiabetic agent.20 Despite a potential antiobesogenic effect, it was not launched in India as an antiobesity agent. However, it does appear to fit into the US FDA “categorical” efficacy criterion as an antiobesity agent based on the findings from the PIONEER-1 and PIONEER-4 trials. Notwithstanding, amongst all antidiabetic agents available in India, oral semaglutide seems to have the highest potential for weight loss in people with T2D. Further phase 3 global trials of higher strength oral semaglutide such as 25 mg and 50 mg in people with obesity (OASIS-1 and OASIS-2) are currently ongoing,21,22 while a top line result from a similar study recently conducted in people with T2D with obesity (PIONEER-PLUS) found both higher strength of oral semaglutide to have superior efficacy not only in terms of glucose-lowering but with an excellent weight lowering effect compared to oral semaglutide 14 mg with an acceptable side effects.23
REFERENCES
- International Diabetes Federation. IDF Diabetes Atlas. 9th Edition. Brussels, Belgium: International Diabetes Federation; 2019.
- World Health Organization. The top 10 causes of death. Available from: http://www.who.int/en/news-room/fact-sheets/detail/the-top-10-causes-of-death
- Indian Council of Medical Research, Public Health Foundation of India, and Institute for Health Metrics and Evaluation. India: Health of the Nation’s States - The India State-Level Disease Burden Initiative. New Delhi: ICMR, PHFI and IHME; 2017.
- Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384(9945):766-81.
- NCD Risk Factor Collaboration (NCD-RisC). Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet. 2016;387(10026):1377-96.
- Kutty VR, Dilip TR, Archana AR, Gopinathan S, Ramanathan M. Shifting pattern of diabetes among the elderly in India: evidence from the national sample survey organization’s data, 2004-2014. Int J Non-Commun Dis. 2018;3(2):67-74.
- Müller TD, Blüher M, Tschöp MH, DiMarchi RD. Anti-obesity drug discovery: advances and challenges. Nat Rev Drug Discov. 2022;21(3):201-23.
- Shi Q, Wang Y, Hao Q, Vandvik PO, Guyatt G, Li J, et al. Pharmacotherapy for adults with overweight and obesity: a systematic review and network meta-analysis of randomised controlled trials. Lancet. 2022;399(10321):259-69.
- Ryan DH, Yockey SR. Weight loss and improvement in comorbidity: differences at 5%, 10%, 15%, and over. Curr Obes Rep. 2017;6(2):187-94.
- Guidance for Industry Developing Products for Weight Management. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). 2007. Available from: http://www.fda.gov/cder/guidance/index.htm
- US Food and Drug Administration. FDA approves novel, dual-targeted treatment for type 2 diabetes. Accessed March 30, 2023.
- Khera R, Murad MH, Chandar AK, Dulai PS, Wang Z, Prokop LJ, et al. Association of pharmacological treatments for obesity with weight loss and adverse events: a systematic review and meta-analysis. JAMA. 2016;315(22):2424-34.
- Singh AK, Singh R. Pharmacotherapy in obesity: a systematic review and meta-analysis of randomized controlled trials of anti-obesity drugs. Expert Rev Clin Pharmacol. 2020;13(1):53-64.
- Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, et al; SURMOUNT-1 Investigators. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-16.
- Muzurovic E, Dragnic S, Medenica S, Smolovic B, Bulajic P, Mikhailidis DP. Weight-centric pharmacological management of type 2 diabetes mellitus - An essential component of cardiovascular disease prevention. J Diabetes Complications. 2020;34(8):107619.
- Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al; STEP 1 Study Group. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
- Lingvay I, Catarig AM, Frias JP, Kumar H, Lausvig NL, le Roux CW, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(11):834-44.
- Rodbard HW, Rosenstock J, Canani LH, Deerochanawong C, Gumprecht J, Lindberg SØ, et al; PIONEER 2 Investigators. Oral semaglutide versus empagliflozin in patients with type 2 diabetes uncontrolled on metformin: the PIONEER 2 trial. Diabetes Care. 2019;42(12):2272-81.
- Singh AK, Singh R, Misra A. Oral semaglutide in type 2 diabetes mellitus: comprehensive review, critical appraisal and clinical consideration of its use in India. Diabetes Metab Syndr. 2022;16(3):102436.
- Joshi SR, Rajput R, Chowdhury S, Singh AK, Bantwal G, Das AK, et al. The role of oral semaglutide in managing type 2 diabetes in Indian clinical settings: addressing the unmet needs. Diabetes Metab Syndr. 2022;16(6):102508.
- Research study to investigate how well semaglutide tablets taken once daily work in people who are overweight or living with obesity (OASIS 1). Available at: https://clinicaltrials.gov/ct2/show/NCT05035095. Accessed April 1, 2023.
- Research study to investigate how well semaglutide tablets taken once daily work in East Asian people who are overweight or living with obesity (OASIS 2). Available at: https://clinicaltrials.gov/ct2/show/NCT05132088. Accessed April 1, 2023.
- Available at: News Details (novonordisk.com). Accessed April 1, 2023.
|