Abstract
India is a heliophobic country; despite ample sunshine, almost 490 million people are vitamin D deficient in the country. Additionally, the Indian diet has not been successful in providing the daily need for vitamin D, leading to a vitamin D deficiency. The need to fortifying food with vitamin D has been raised several times. Besides, there have been discussions about whether vitamin D is a hormone or a vitamin? In this review, the authors have reviewed vitamin D deficiency and its status in India, assessment and screening, the role of vitamin D in various disease conditions, dosage recommendation and regimen.
Keywords: Vitamin D, heliophobic, vitamin D deficiency, hypervitaminosis
With approximately 490 million people deficient in vitamin D in a sun-rich country like India, it is important to emphasize the significance of vitamin D sufficiency. Besides, the Indian diet generally fails to provide the daily need for vitamin D, resulting in a deficiency. There is a need to fortify various foods with vitamin D through national health programs and raise awareness about the importance of diet, lifestyle and vitamin D supplementation. Another ongoing discussion is whether vitamin D is a vitamin, prohormone or hormone precursor. Hence, this silent epidemic needs to be addressed appropriately. In this article, the authors have reviewed the present status of vitamin D deficiency in India, its positioning as a vitamin or hormone, clinical implications in various conditions, and given recommendations on the use, dosage and duration of vitamin D use. An algorithm has also been suggested for managing the population’s vitamin D deficiency and sufficient conditions.
EPIDEMIOLOGY OF VITAMIN D DEFICIENCY
Vitamin D deficiency has been cited for having assumed pandemic proportions, despite being the most under-diagnosed and under-treated nutritional deficiency globally.1 In the general population across the world, vitamin D deficiency ranges from 20% to 80% and in critically ill patients, the prevalence increases with a simultaneous rise in disease severity.2 With an exponential rise in prevalence, recent observational studies have revealed that almost 40% of Europeans are vitamin D deficient, while 13% are severely deficient.3 Vitamin D deficiency or insufficiency has been reported from several countries across the globe, including the United States, South America, Canada, the Middle East, Australia, South Asia, Europe and Africa. Studies from India have reported a wide range of vitamin D deficiency, from 30% to as high as 100%. Other parts of the world, like the Middle East, China and South America, have also reported an alarmingly high prevalence of vitamin D deficiency.4
Several countries worldwide report a very high prevalence of low vitamin D status. Vitamin D [25(OH)D]
levels below 30 nmol/L occur commonly in India, Tunisia, Pakistan and Afghanistan in over 20% of the population. Studies have shown that 490 million individuals have vitamin D deficiency in India.3
Another study amongst South Asian adults revealed a high (68%) and variable prevalence of vitamin D deficiency among the adults of different South Asian countries, which was hypothesized to be linked to the high prevalence of several other health issues in this region. A country-wise comparison revealed that Sri Lanka had the lowest prevalence of vitamin D deficiency (48%), while Pakistan had the highest prevalence (73%). Bangladesh and the Indian population had about 67% prevalence of vitamin D deficiency.5
Community-based studies conducted in the last 10 years have shown a prevalence range of 50% to 94% of vitamin D deficiency in India. A high prevalence of vitamin D has been noted in the country.6 A study conducted in South India showed a high prevalence of vitamin D deficiency among pregnant women, attributed to reduced physical activity, sun exposure, darker skin complexion, lower socioeconomic status and lack of awareness.7 A single-center cross-sectional study in North India revealed a prevalence of vitamin D deficiency of 55.55% and inadequacy of 38.46%.8
While sun exposure can suffice for vitamin D sufficiency, it is a frequently witnessed nutritional deficiency. Even though India is a tropical country with abundant sunshine, it seems to be a heliophobic country as vitamin D deficiency has been prevalent. Vitamin D deficiency is commonly present in individuals of all ages, gender, race and geography.1
As per a report by the International Osteoporosis Foundation in 2009, 96% of neonates, 91% of healthy school girls, 78% of healthy hospital staff and 84% of pregnant women in North India were diagnosed with hypovitaminosis D.9
A cross-sectional multicenter study has demonstrated a high prevalence of vitamin D deficiency across India in healthy, middle-aged health care professionals.10 Vitamin D deficiency is particularly pronounced in special categories of patients, including patients with chronic renal failure and on hemodialysis, renal transplant recipients affected with liver disease or after liver transplantation, ranging from 85% to 99%.3
Besides, critically ill patients have an extremely high prevalence of vitamin D deficiency, and low vitamin D levels are also associated with increased illness severity, morbidity and mortality in patients admitted to intensive care units (ICUs) and medical and surgical ICUs.3 In a study conducted in South India, vitamin D deficiency was present in all patients with active tuberculosis despite adequate sunlight exposure in 72% of the cases.11
The Eradication of Vitamin D Deficiency
Vitamin D deficiency is a global issue manifesting several clinical signs and symptoms with major consequences. Vitamin D deficiency has a high global prevalence and leads to many health risks, including bone health, chronic pain in cancer, musculoskeletal, fibromyalgia, autoimmune conditions, cardiovascular diseases (CVDs) and even neurological system disorders. The benefits of vitamin D are undisputable in bone health. An early application of vitamin D is further supported by extraskeletal benefits.12 Considering the high prevalence of vitamin D and its overarching consequences, it is important to take steps toward eliminating vitamin D deficiency.
Iodine deficiency, at one point, was the world’s single most important preventable cause of brain damage and mental retardation. Thirty-eight million newborns in developing countries remain unprotected from the lifelong impact of brain damage associated with iodine deficiency disorders. A massive worldwide effort led to a dramatic rise in the proportion of people consuming iodized salt, from under 20% in the 1990s to about 70% by 2000.13 The Iodine Deficiency Disorder Control Programme in India is a public health success story. The successful translation of research to policy to program, sustained political commitment, the increased production of iodized salt and involvement of the private sector, the institution of legislation to ensure iodization of salt and the catalytic role played by academic institutes, civil society and international agencies are key factors in establishing the success of Iodine Deficiency Disorder Control Programme.14 Taking cues from the successful programs implemented to eradicate iodine deficiency, measures such as nutritional replenishment of iodine, robust health and nutrition education activities to create demand for iodine insufficiency and developing monitoring information systems for ensuring that iodine is sufficiently available to the population,14,15 we can facilitate evidence to policy transition to eradicate vitamin D from India.
IS VITAMIN D A VITAMIN OR HORMONE?
There has been a discussion amongst clinicians and researchers about whether vitamin D is a prohormone or hormone precursor, as it has to be synthesized by the sunlight for activation. Unlike other vitamins such as A, B and C, vitamin D is the only vitamin that the body can make. It plays an important part in the neurobiological pathways and cascading effects associated with mental health. Other evidence has also shown that it has a crucial role in various endocrine pathways, reiterating that vitamin D deficiency is a hormonal deficiency. Besides, vitamin D is also involved in brain development on a hormone level. Hence, vitamin D fits more appropriately in the criteria of a hormone replacement rather than a vitamin supplement.12
Technically, the term vitamin D is misleading, as it cannot be considered a true vitamin. The body has the capacity to synthesize its own cholecalciferol (D3). Hence, it is more accurately referred to as a steroid hormone or an oxysterol. The International Union of Pure and Applied Chemistry’s Commission on the Nomenclature of Biological Chemistry defines vitamin D3 as a ‘steroid or secosteroid’16 (Refer Box).
|
Box. Vitamin D: Vitamin vs. Hormone
|
|
Evidence suggesting that vitamin D is a hormone
|
|
Prohormone
|
Vitamin D is a prohormone, the body converts the substance Vitamin D into a hormone for it to be used.12
It is a key regulator of bone metabolism and calcium and phosphorus homeostasis through negative feedback with the parathyroid hormone.17
|
|
Hormone precursor
|
Vitamin D is a hormone precursor, as it must be synthesized by the sunlight for activation.16
|
|
Synthesized by the body
|
The only vitamin that can be made by the body, unlike other vitamins.18
The endogenous synthesis occurs by ultraviolet light exposure of 7-dehydrocholesterol within the microvessels of the skin resulting in its conversion into cholecalciferol.18
|
Vitamin D Formulations
Vitamin D preparations may be administered orally, subcutaneously, intraperitoneally or intravenously. They may be administered daily, sometimes in divided doses or intermittently in large bolus doses, usually 3 times per week. The timing of the dose is also important which may be at night, as the gut calcium load is at a minimum at night.19
Vitamin D is available in two forms in India: Vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). A market assessment in India has shown that over 99.9% of the preparations contain vitamin D3 in the form of alfacalcidol, calcitriol or cholecalciferol. Most of the preparations have vitamin calcitriol or alfacalcidol. About 10% of preparations contain cholecalciferol.20
The most common formulation for oral administration is in the form of tablets and capsules. Alfacacidiol and calcitriol are commonly available as tablets and capsules; cholecalciferol is in the form of granules in sachets. Other dosage forms include syrups and soft gel capsules. More than 75% of alfacalcidol preparations also contain calcium. Most of the calcitriol preparations are combined with zinc or zinc sulfate. Several vitamin D preparations also contain various other minerals/vitamins such as magnesium, cupric, boron, methylcobalamin, vitamin E, vitamin K, vitamin C, pyridoxine, folic acid, beta-carotene, glutamic acid, manganese, omega-3 and docosapentaenoic acid.20
Tablets and capsules contain 10-10,000 IU of vitamin D and are usually administered on a daily basis. Cholecalciferol soft gel capsules, syrup and sachets containing granules of vitamin D (60,000 IU) are given weekly. Vitamin D2 preparation is available in tablet form in two formulations containing 20 IU and 100 IU, respectively.20
The parenteral route has been shown to be effective and safe in patients with hypovitaminosis D caused by severe intestinal malabsorption. Intramuscular vitamin D is the preferred treatment for malabsorption disorders like intestinal bowel disorders, pancreatic insufficiency, short-bowel syndrome, gluten enteropathy, post-bariatric surgery and any need to total parenteral nutrition.21 Table 1 depicts the different preparations of vitamin D formulations available in India.
|
Table 1. Different Formulations of Vitamin D
|
|
Formulation
|
Vitamin D strength
|
Administration
|
|
Tablets/Capsules
|
10-10,000 IU
|
Daily
|
|
Sublingual tablets
|
10-10,000 IU
|
Daily
|
|
Soft gel capsules/granules/syrup/tablet
|
60,000 IU
|
Weekly
|
|
Intramuscular injection
|
6,00,000 IU/mL
|
Weekly
|
|
Soft gelatin capsules/tablets
|
10-10,000 IU
|
Daily
|
Recommended Vitamin D Levels in the Body
Maintaining adequate serum levels of 25(OH)D for skeletal and extraskeletal physiologic effects is important. Studies have shown that the desirable and safe levels of serum 25(OH)D levels should be 30-100 ng/mL because, at the level of 30 ng/mL, the intestinal calcium absorption reaches its highest level and parathyroid hormone (PTH) levels are continually reduced until this level of 25(OH)D is reached.6
Vitamin D deficiency is described as a serum/plasma level of 25(OH)D below 75 nmol/L (or 30 ng/mL). A threshold of <25 or <30 nmol/L (or 10/12 ng/mL) can raise the risk of osteomalacia and nutritional rickets, thus determining severe vitamin D deficiency.3
The clinical practice guidelines of the Endocrine Society Task Force on vitamin D have described a cut-off level of 50 nmol/L as vitamin D deficient.22 In addition, various societies and expert bodies worldwide define 50 nmol/L as the vitamin D requirement of all normal, healthy individuals, primarily considering bone health.
We recommend that desired serum level of vitamin D should be between 40 and 60 ng/mL, and hence, appropriate vitamin D dose should be administered.
INDICATIONS FOR VITAMIN D
Musculoskeletal Effects of Vitamin D Deficiency
Rickets and osteomalacia
Vitamin D, a secosteroid hormone, is crucial for calcium absorption and bone mineralization absorption, which positively associates with bone mineral density.23 It is known that prolonged and severe vitamin D deficiency causes rickets in children and osteomalacia in adults. Results from several clinical studies have shown that vitamin D supplementation can improve muscle strength contributing to a reduction in the incidence of falls.23
It has been established that insufficient vitamin D intake over a prolonged period can result in bone demineralization. Vitamin D deficiency reduces calcium absorption, eventually reducing the calcium concentrations in the circulation. Vitamin D deficiency also causes secondary hyperparathyroidism, which structurally weakens the bone and increases the risk of fractures.23
Maternal vitamin D deficiency passes on to the infant who is at high risk of presenting in the first few days or months of life with hypocalcemic complications, including seizures, tetany and dilated cardiomyopathy. Vitamin D deficiency in older children can result in motor delay, proximal myopathy and dental complications. Osteomalacia in girls can result in pelvic deformities and lead to obstructed labor later in life.24
Vitamin D deficiency triggers the release of PTH, which enhances urinary phosphate excretion, resulting in hypophosphatemia and osteomalacia.25 Low calcium intake and/or low vitamin are the primary reason for body calcium deprivation across the world, and their combined deficiency accelerated bone demineralization.24
Osteoporosis
Osteoporosis is defined as a bone mineral density (BMD) 2.5 standard deviations below the mean of healthy young individuals. Also, there is a direct relationship between BMD and fracture risk. Hence, vitamin D plays a key role in osteoporosis and fractures.23
The results of vitamin D deficiency are secondary hyperparathyroidism and bone loss, resulting in osteoporosis and fractures, mineralization defects, which may lead to osteomalacia in the long-term, and muscle weakness, causing falls and fractures. Several randomized controlled trials have shown that vitamin D and calcium showed a significant reduction in the incidence of fracture.26
Research has shown that vitamin D supplementation improves BMD and enhances muscle function, leading to reduced falls. It can also modulate the effect of pro-inflammatory cytokines on bone metabolism.23
Vitamin D sufficiency is, therefore, pivotal for normal skeletal development both in utero and in childhood and sustaining bone health in the adult population. A deficiency of vitamin D in mothers can lead to a considerable reduction in infant bone mineral acquisition, which is persistent in nature.27
Sarcopenia
Sarcopenia refers to a loss of skeletal muscle mass with aging. It is marked by the progressive and generalized loss of skeletal muscle mass and strength with a risk of adverse outcomes such as physical disability, poor quality of life and death. Studies have shown a positive correlation between serum 25(OH)D concentration and muscle function. A decline in serum 25(OH)D concentration with rising age leads to reduced bone density, leading to a higher risk of falling and bone fractures.28
Studies have shown that older people with vitamin D deficiency may be at risk of sarcopenia, a geriatric syndrome featured by the progressive loss of skeletal muscle mass and strength frequently complicated by adverse events, including falls, disability, hospitalization and death.29
MOAN syndrome
MOAN refers to the musculo-osteo-arthro-neuropathic syndrome. Osteoporosis is often related to sarcopenia and osteoarthritis and is attributed to calcium and vitamin D deficiency, which may eventually result in metabolic neuropathy and myopathy. While conditions such as sarcopenia or loss of muscle mass or strength or function may occur as a primary condition, which may result in secondary such as osteoporosis and arthritis. Even though the associations have been shown to occur individually, it is important to address MOAN syndrome as one entity. Vitamin D may play an important role in preventing and managing the MOAN syndrome.30
Mitogenic Disorders
Strong preclinical data have demonstrated that vitamin D is linked with cell cycle control and cancer.31 It was shown in a Cochrane systematic review that cancer mortality was modestly reduced by vitamin D supplementation in individuals receiving a mean daily dose of 1,146 IU compared to no supplementation during a mean follow-up for more than 6 years.32 Several large randomized controlled trials, including the VITAL trial, showed that a vitamin D supplementation group had a nonsignificant trend of reduction in total cancer mortality.
The vitamin D endocrine system influences all cells and most cytokines of the immune system.33 As per the results of the LUNG-ViDA trial, vitamin D supplementation may have a modest impact in improving expiratory lung function.34
Some studies have also shown a strong link between poor vitamin D status and increased risk of infection or autoimmune diseases such as multiple sclerosis, inflammatory bowel diseases, or type 1 diabetes mellitus.35 A causal relationship between increased risk of multiple sclerosis and genetically low serum 25(OH)D levels has also been confirmed.
Besides, pregnant women frequently report poor vitamin D status compared to nonpregnant women of the same age. Studies have shown that vitamin D supplementation may lead to a considerable reduction in maternal mortality and improve the health of the baby.36
Psoriasis
Vitamin D has a significant role in the treatment of psoriasis, owing to its activity in the proliferation and maturation of keratinocytes. Significant associations between low vitamin D status and psoriasis have been systematically observed. A bi-directional relationship between low vitamin D status and psoriasis is also important for delineating the risk profile for comorbidities that may result from psoriasis, including type 2 diabetes and metabolic syndrome.37 Clinicians should consider general vitamin D supplementation in individuals who are at high risk of vitamin D deficiency, such as psoriatic patients.
Glucometabolic, Cardiometabolic and Barometabolic Disorders
Cardiovascular diseases
Vitamin D deficiency has been implicated in cardiometabolic disorders, including obesity, type 2 diabetes mellitus, CVD and polycystic ovary syndrome.38
Epidemiologic studies have revealed that low vitamin D levels are linked with an increased risk of cardiovascular events. 25(OH)D plays a role in diabetic CVD, myocardial infarction (MI), heart failure and peripheral vascular disease. Decreased vitamin D levels are strongly associated with a high prevalence of coronary artery disease, a 3-times rise in the rate of myocardial ischemia among individuals with hypertension. Besides, the rate of cardiovascular complications, including MI and heart failure, was higher in individuals with vitamin D deficiency who were followed for about 5 years.39
It was seen in a clinical trial that vitamin D treatment may be effective in patients with cardiometabolic disorders having vitamin D deficiency.40 Another study has shown that vitamin D supplementation given at a dose of 50,000 IU for 16 weeks brought about a considerable reduction in triglyceride level.41
Another Iranian study demonstrated an inverse link between plasma vitamin D levels and cardiometabolic risk factors in children and adolescents.42 Supplementation also significantly reduces blood pressure in elderly women.43 A systematic review and meta-analysis have shown that high vitamin D levels in middle-aged and elderly populations are related to a significant reduction in CVD, type 2 diabetes and metabolic syndrome.44
Vitamin D deficiency is highly prevalent and may lead to arterial hypertension. Vitamin D exerts its effect by suppressing renin and PTH levels and its anti-inflammatory and vasculoprotective properties. Low vitamin D levels may be an independent risk factor for incident arterial hypertension. Results of cross-sectional studies have shown that low vitamin D is linked with higher systolic blood pressure and a higher incidence of hypertension.39
Real-world data has shown that high vitamin D levels are associated with a favorable serum lipid profile. While there have been mixed results from different studies, some randomized controlled studies have indicated that vitamin D supplementation provided a statistically significant rise in low-density lipoprotein cholesterol (LDL-C), with a tendency towards a rise in total cholesterol. Besides, the effect of vitamin D supplements on serum LDL-C levels is more pronounced in obese individuals.45 Supplementation of vitamin D (1,000 mg) for a month in healthy school children demonstrated a significant improvement in the high-density lipoprotein (HDL) level.46
Type 2 diabetes
Poor vitamin D levels may act as one of the driving factors for the development of type 2 diabetes. This has been corroborated by the fact that low vitamin D status is linked with an increased risk of glucose intolerance or diabetes. Longitudinal studies have exhibited that poor vitamin D level is associated with an increased incidence of type 2 diabetes. Changes in calcium and vitamin D homeostasis are related to insulin resistance, decreased β-cell function, metabolic syndrome, glucose intolerance and diabetes.39 Vitamin D supplementation (60,000 IU) for 1 year period significantly reduced the glycemic parameters, including fasting blood glucose (FBG), postprandial blood glucose (PPBG) and glycated hemoglobin (HbA1c).47
Obesity and overweight
Studies have shown that fluctuation in serum 25(OH)D concentrations may be associated with a myriad of problems. It has been shown that an increase in body mass index (BMI) levels per unit is associated with a 1.15% lower concentration of 25(OH)D, with adjusted age and sex. Other evidence points towards an association between obesity, high concentration of PTH and 1,25-dihydroxyvitamin D [1,25(OH)2D] with low serum 25(OH)D concentrations. Additionally, changes in behavior, low synthetic capacity, intestinal absorption and a changed metabolism play a major role in controlling vitamin D levels. Obese individuals are exposed to less sunlight, resulting in reduced synthesis of vitamin D. Besides, it has been suggested that weight loss is associated with improved serum vitamin D concentration in overweight or obese women.39
However, a few studies have also suggested that there are no evident benefits of vitamin D supplementation in cardiometabolic disorders.38
Miasma-related Disorders
Vitamin D deficiency has been shown to occur frequently in patients with chronic airway inflammatory and infectious diseases, correlated with increased disease severity. Research has shown that vitamin D modulates ongoing abnormal immune responses in chronic respiratory diseases and is shown to restrict bacterial and viral colonization into the lungs.48
Various studies have highlighted the association between low levels of vitamin D deficiency with increased risk of asthma and other respiratory disease symptoms, including lower lung function. Several studies involving clinical trials of vitamin D supplementation on asthma symptoms have reported positive results.48
Among the published clinical studies, a hospital-based cross-sectional study has reported that increased tumor necrosis factor (TNF)-α levels and low serum vitamin D concentrations are related with airway obstruction in chronic obstructive pulmonary disease (COPD) patients.49 It has been assessed that around 40% to 90% of cystic fibrosis patients are vitamin D deficiency, with vitamin D serum levels below 30 ng/mL. Almost 15% to 20% have vitamin D levels below 15 ng/mL. While patients usually are given vitamin D supplementation when they present exocrine pancreatic insufficiency, but it normally appears at birth or during the first months of life. Hence, supplementation usually starts with the diagnosis.50
ASSESSMENT AND SCREENING
With the increasing awareness about the importance of vitamin D for maintaining musculoskeletal health, there has been a rise in vitamin D testing. However, uniform screening for vitamin D deficiency is not recommended in the general population as it is an expensive test. However, given that certain individuals are at high risk of vitamin D deficiency, it is important to perform targeted testing of the susceptible population.51
Once the treatment is initiated, timely retesting following 3 to 6 months of treatment must confirm adequate treatment and prevent potential toxic over-treatment. Vitamin D levels undergo seasonal variations and hence should be considered when tests and interpretations are conducted. In addition, risk factors for vitamin D deficiency should be considered to ensure targeted testing of patients.52 The screening of vitamin D deficiency should be considered in the case of osteoporosis, osteomalacia, chronic kidney disease, hepatic failures, malabsorption syndromes, hyperparathyroidism, chronic treatment with medications that influence vitamin D metabolism, pregnant and lactating women, institutionalized or hospitalized patients, older adults (>65 years) in general, older adults with a history of falls or a history of nontraumatic fractures, granuloma-forming disorders, chronic autoimmune diseases, obesity, dark skin pigmentation, different types of cancer, certain cardiovascular disorders, diabetes mellitus and its comorbidities, some neurological conditions and recurrent acute respiratory tract infections; in symptomatic patients with musculoskeletal pain; before initiating osteoporosis treatment with antiresorptive medications.51
Clinical Presentation of Vitamin D Deficiency
The common clinical presentations of vitamin D deficiency are listed in Table 2.53
|
Table 2. Clinical Manifestations of Vitamin D Deficiency
|
| |
Infants
|
Children
|
Adults
|
|
Symptoms
|
Difficult breathing, irritability
|
Repeated infection, bone pain, difficulty in squatting/standing/walking
|
Bone pain, muscle pain, muscle weakness, difficulty in squatting/standing/walking, frequent falls
|
|
Signs
|
Seizures, tetany stridor
|
Hypotonia, delayed dentition, enamel hypoplasia, dental caries, wrist widening, Rachitic rosary, knock knees, bowlegs
|
Waddling gait, anterior tibial weakness, rib cage tenderness, fractures, pelvic deformities, kyphoscoliosis
|
| |
Dilated cardiomyopathy, craniotabes, open anterior fontanelle and frontoparietal bossing
|
|
|
Markers to Assess Vitamin D Deficiency
Total serum 25(OH)D concentration is the primary marker for assessing vitamin D status, reflecting vitamin D supply from all sources, including endogenous vitamin D synthesis in the skin, diet, supplements and mobilization from tissue stores. Patients suffering from vitamin D deficiency and other and other related health issues, such as bone diseases, require testing of additional laboratory parameters, including serum calcium, phosphate, alkaline phosphatase, PTH, creatinine and magnesium to guide further the diagnostics and treatment of these patients.51
Vitamin D deficiency is also accompanied by normal blood levels for calcium and phosphorus, high normal or increased levels of PTH, normal to high levels of total alkaline phosphatase, a low 24-hour urine calcium excretion rate and low levels of total 25(OH)D. Patients suffering from severe and chronic vitamin D deficiency may present with overt hypocalcemia and/or hypophosphatemia.54 Based on risk factors and seasonal alteration in the serum levels of vitamin D, it is recommended that:
- Targeted testing based on risk factors is performed.
- Seasonal variations in vitamin D levels are considered before ordering testing for patients.
- Retesting for individuals on therapy is ordered after 3 months following the initiation of vitamin D treatment.
HYPERVITAMINOSIS D
An increased administration of vitamin D supplements may sometimes result in a higher risk of exogenous hypervitaminosis D, displaying symptoms of hypercalcemia, also referred to as vitamin D toxicity. Hypervitaminosis may develop as a result of using vitamin D analogs. Hypervitaminosis D with hypercalcemia may also be a manifestation of excessive production of 1,25(OH)2D in granulomatous disorders, in lymphomas and during idiopathic infantile hypercalcemia.55
PREVENTION, TREATMENT AND MANAGEMENT
Sunlight
Insufficient sun exposure is the main reason for vitamin D insufficiency. The ultraviolet portion of the sunlight enters the skin and is converted to pre-vitamin D3. Pre-vitamin D3 rapidly converts within the plasma membrane to vitamin D3, which is released in the extracellular space. Factors affecting the cutaneous synthesis of vitamin D are sunscreen use, skin pigmentation, time of day, year’s season, latitude and aging. Limited sensible exposure to sunlight or ultraviolet B radiation is more effective in raising blood levels of 25(OH)D than 1,000 IU vitamin D3 taken daily for most skin types.56
Vitamin D-fortified Foods
Vitamin D-fortified foods usually contain 100 IU per serving of vitamin D. It has been reported that consumption of vitamin D-fortified, especially milk, increased vitamin D intake and effectively increased 25(OH)D levels. Cereal, juices, other dairy products and margarine are other food items that can be fortified with vitamin D.1 Considering limited dietary sources of vitamin D, enhancing foods with vitamin D is a probable mode for ensuring increased consumption and improved vitamin D status.57 Vitamin D food fortification is also an effective method to improve 25(OH)D levels, prevent vitamin D deficiency and improve the intelligence quotient in the pediatric population.58 Besides, it has been seen that milk fortified with ergocalciferol has effectively eliminated rickets as an important health problem, and when used in high doses, ergocalciferol can effectively treat osteomalacia in adults.59
Pharmacological Therapy
Vitamin D supplementation
Sunlight and the skin are the primary sources of vitamin D; changes in sunlight exposure to the skin may alter vitamin D levels. There are several reasons for the low levels of vitamin D in the South Asian population, including dark skin, individuals whose skin is not exposed to sunlight because of clothing, sunscreen use or lack of outdoor activities. Besides, in winter, ultraviolet irradiation is reduced, particularly in temperate climates and air pollution leads to further vitamin D deficiency. Exclusive sunlight exposure is not adequate to achieve recommended vitamin D levels, and supplementation is necessary.53 Vitamin D is included in the WHO Essential Medicines List 2021 and the Indian National List of Essential Medicines 2022 as micronutrient supplements.60
To ensure that non-specific symptoms of vitamin D insufficiency are not over-treated, patients are recommended to use over-the-counter vitamin D supplements or fortified food supplements such as cereals.52 An empiric vitamin D supplementation without testing is suggested for patients without any risk factors or signs of deficiency but are suspected of having insufficient sun exposure or dietary intake.54
Vitamin D supplementation can be used to treat almost all individuals except those with an increased sensitivity to vitamin D treatment, a rare condition. Daily, weekly or monthly vitamin D supplementation at equivalent doses can adequately lead to a similar increase in 25(OH)D serum concentrations, while a daily vitamin D dosing is preferred.51
Individuals with an assessment of vitamin D level to be below 20 ng/mL should be treated with vitamin D supplementation. When aiming for a minimum 25(OH)D concentration of at least 30 ng/mL, a daily vitamin D supplement dose of about 1,000 IU per day is sufficient for all individuals, irrespective of the season. 100 IU of vitamin D per day increases serum 25(OH)D concentrations by about 1 ng/mL is one of the factors modulating the individual treatment response. Evaluation of treatment success should be considered in individuals with insufficient and deficient vitamin D levels after 3 months of treatment initiation.51
DOSAGE AND DURATION
Currently, bolus dosing with long dosing intervals is not used in the general population because of an increased risk of adverse effects such as falls and fractures related to them.61 A 2017 metadata analysis showed that vitamin D leads to a significant benefit in case of acute respiratory infection with daily or weekly dosing but not with longer dosing intervals.62 In the case of intensive care, an upfront loading dose, followed by a daily dose, is important to improve vitamin D levels quickly.3
Vitamin D dosing regimen should be such that it can maintain stable availability of several vitamin D metabolites. Vitamin D supplementation is necessary as only sunlight exposure and dietary intake are usually insufficient in most individuals. Currently, there is no international consensus on the standard level of vitamin D supplementation, and recommendations vary in different categories ranging from a daily dose of 400 to 2,000 IU.3
Endocrine Society has given a safe upper level for vitamin D supplementation as 10,000 IU, while the Institute of Medicine (IOM) and the European Food and Safety Authority recommend a value under 4,000 IU per day (100 µg). Most countries have recommended the upper level as 2,000 IU per day (50 µg) for adults.3
However, clinical studies on dose-response relationships or toxicity studies have shown that there are no adverse effects following an intake of 1,250 µg (50,000 IU) once every 2 weeks for several years. High doses do not cause hypercalcemia or other evidence of hypervitaminosis D.63 Clinical studies conducted on smaller populations have also shown that even a daily consumption of up to 250 µg (10,000 IU) of vitamin D over long periods did not result in any adverse events in healthy adults.
Considering that the recommended daily dose of vitamin D supplementation of up to 2,000 IU may lead to severe side effects in the general population. Since, 800 IU is the lowest dose associated with a bone benefit, a recommended daily dose of 20 to 50 µg is reasonable.64 A daily vitamin D of 800 IU is adequate to achieve a target of at least 20 ng/mL in most healthy individuals. However, 2000 IU is sufficient to achieve a level at least 30 ng/mL. Clinical data has also pointed out that a level higher than 20 ng/mL may be needed for optimal risk reduction in several cases.3
The results of a study showed that after a normal level of 25(OH)D is reached, the daily maintenance dose of 2,000 IU was inadequate to achieve and stabilize the level of 25(OH)D at ≤30 ng/mL. The daily dosage has increased from 400 to 2,000 IU per day to maintain normal levels. However, an adequate maintenance dose is required to prevent recurrent deficiency, irrespective of the initial dosage. National Health Service in the United Kingdom has recommended a daily dose ranging between 800 to 2,000 IU per day but opined to raise the dose up to 4,000 IU daily. However, the study supported the fact that supplementation is the only source of vitamin D, and obese individuals require 2 to 3 times higher doses compared to individuals with normal weight.65 Table 3 shows the recommended doses of vitamin D by different societies and official bodies.
|
Table 3. Daily Vitamin D Dosage Recommended by Different Societies and Organizations
|
|
Recommendation from
|
Vitamin D recommendation
|
|
United States RDA
|
200 IU/day in men and women ≤50 years
|
|
Vitamin D Council
|
2,000 IU vitamin D daily ≤60 years
|
|
IOF
|
800 to 1,000 IU/day in men and women
|
|
ICMR-NIN
|
600 IU/day in adult men and women
800 IU/day in men and women >60 years
|
RDA = Recommended Dietary Allowance; IOF = International Osteoporosis Foundation; ICMR-NIN = Indian Council of Medical Research-National Institute of Nutrition.
Based on several studies, it has been suggested that vitamin D deficiency is defined as 25(OH)D below 20 ng/mL, insufficiency as 25(OH)D of 21 to 29 ng/mL, and sufficiency as a level of 25(OH)D of 30 to 100 ng/mL.22 Figure 1 gives an algorithm giving a step-by-step approach to assessing and managing vitamin D deficiency.
Figure 1. The clinical approach to managing individuals with vitamin D insufficiency.
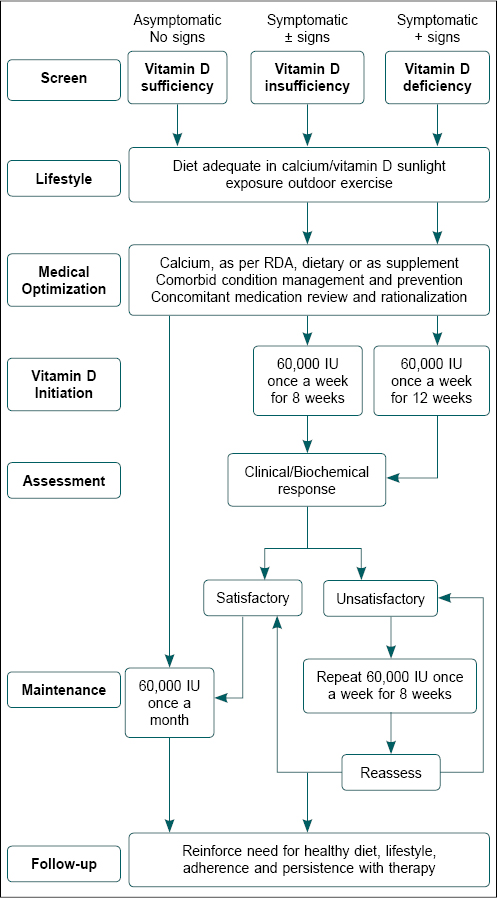
7 Ds of Vitamin D Supplementation
Demand
- Vitamin D deficiency has assumed pandemic proportions.
- Indian studies show prevalence ranging from 50% to 94%.
- India is a heliophobic country leading to an inadequate exposure to sunlight, resulting in insufficient levels of vitamin D.
- Vitamin D deficiency has been implied in multiple systems; cardiometabolic disorders, essential for bone health in children and adults, and MOAN syndrome.
- Included in WHO Model List of Medicines 2021 and National List of Essential Medicines 2022.
Dosage
- Asymptomatic individuals: 800 to 2,000 IU per day.
- Symptomatic, vitamin D insufficient: Up to 60,000 once a week.
- Symptomatic, vitamin D deficient: Up to 60,000 IU once a week.
Duration
- Asymptomatic individuals: 800 to 2000 IU per day.
- Symptomatic vitamin D insufficient: Up to 60,000 IU
- Initiation dose: Once a week for 8 weeks
- If unsatisfactory, once a week for 8 weeks
- Maintenance dose: Up to 60,000 IU, once a month.
- Symptomatic vitamin D deficient: Up to 60,000 IU
- Initiation dose: Once a week for 12 weeks
- If unsatisfactory, once a week for 8 weeks
- Maintenance dose: Up to 60,000 IU, once a month.
Dynamics
- The recommendation for supplementation should be indication based.
- Vitamin D supplementation dependent on season, age, body weight and other risk factors including susceptible individuals including elderly >65 years, hospitalized individuals, women planning pregnancy and in patients with osteoporosis with an increased risk of falls. OR
- Patient should be referred for vitamin D supplementation
- Lack of improvement
- Malabsorption
- Hypercalcemia
- Associated kidney disease or hepatic disease
- In individuals post-bariatric surgery.
Delivery
- Available in the form of tablet, capsules, soft gel capsules and syrups.
- Preferred drug formulation may be customized based on ease of administration and the patient compliance.
Desirability
- In the general population, desired serum levels following supplementation with initiation and maintenance dose should be 40 to 60 ng/mL.
- Vitamin D insufficient levels: ≥20 to <30 ng/mL.
- Vitamin D deficient levels: <20 ng/mL.
Direction
- Education and awareness of general practitioners on vitamin D deficiency
- Desired serum levels, recommendations for indications, loading and maintenance dosages needed.
- Reinforce need for healthy diet, lifestyle, adherence and persistence with therapy.
- Public awareness about
- Importance of sunlight exposure for vitamin D production.
- Lifestyle factors, diet, vitamin D-fortified food.
- Vitamin D supplements availability, required dosage and formulations.
AWARENESS OF VITAMIN D DEFICIENCY
It is known that a lack of knowledge and awareness of vitamin D deficiency acts as a potential risk factor for vitamin D deficiency.66 A study on children showed that most individuals with vitamin D deficiency are unaware of vitamin D sources.67 There is a need for an improved campaign for awareness, knowledge and attitudes regarding vitamin D.
Several factors work at the heart of a successful eradication program. In case of vitamin D deficiency, it can be integrated with other programs running under the Ministry of Health and Family Welfare, such as maternal and child health programs (Janani-Shishu Suraksha Yojana, Janani Suraksha Yojana, Pradhan Mantri Surakshit Matritva Abhiyan, etc.), national program for health care of the elderly and noncommunicable disease control programs. Health care professionals need educational programs to create awareness about vitamin D assessment and supplementation.
CONCLUSION
Vitamin D deficiency could be symptomatic or subclinical, with a continuously rising prevalence. Hence, there is a clear need to educate the public and health care professionals about the prevention of deficiency through supplementation, diagnosis through targeted testing, especially in high-risk individuals, and adequate treatment, maintenance and monitoring.
Vitamin D is implicated in bone health, attributing to increased fractures and susceptibility to falls in individuals who are vitamin D deficient. Unarguably, vitamin D has also emerged as an independent risk factor for various CVDs, including MI and coronary heart disease.
Considering vitamin D prevalence in the general population and its clinical manifestations, we recommend the following dosage and duration of vitamin D: (a) Asymptomatic individuals – 800 to 2,000 IU per day, or 60,000 IU once every 30 days. (b) Symptomatic vitamin D insufficient – Up to 60,000 IU - initiation dose is once a week for 8 weeks and maintenance dose up to 60,000 IU, once a month. (c) Symptomatic vitamin D deficient – Up to 60,000 IU - initiation dose is once a week for 12 weeks and maintenance dose up to 60,000 IU, once a month.
REFERENCES
- Ritu G, Gupta A. Vitamin D deficiency in India: prevalence, causalities, and interventions. Nutrients. 2014;6(2):729-75.
- Chen KW, Chen CW, Yuan KC, Wang IT, Hung FM, Wang AY, et al. Prevalence of vitamin D deficiency and associated factors in critically ill patients: a multicenter observational study. Front Nutr. 2021;8:768804.
- Amrein K, Scherkl M, Hoffmann M, Neuwersch-Sommeregger S, Köstenberger M, Tmava Berisha A, et al. Vitamin D deficiency 2.0: an update on the current status worldwide. Eur J Clin Nutr. 2020;74(11):1498-513.
- Sarmah D, Sharma B. Vitamin D deficiency: is it real? Need to re-evaluate in context to the latest research findings. Int J Adv Med. 2016;3(2):142-4.
- Siddiqee MH, Bhattacharjee B, Siddiqi UR, Meshbahur Rahman M. High prevalence of vitamin D deficiency among the South Asian adults: a systematic review and meta-analysis. BMC Public Health. 2021;21(1):1823.
- Aparna P, Muthathal S, Nongkynrih B, Gupta SK. Vitamin D deficiency in India. J Family Med Prim Care. 2018;7(2):324-30.
- Ravinder SS, Padmavathi R, Maheshkumar K, Mohankumar M, Maruthy KN, Sankar S, et al. Prevalence of vitamin D deficiency among South Indian pregnant women. J Family Med Prim Care. 2022;11(6):2884-9.
- Kalra S, Kalra B, Khandelwal SK. Vitamin D status in patients with musculoskeletal symptoms in Haryana, India. J Med Nutr Nutraceut. 2012;1(1):50-3.
- Vishwanath P, Kulkarni P, Prashant A. Vitamin D deficiency in India: are we overconcerned? Int J Health Allied Sci. 2014;3(2):77-8.
- Beloyartseva M, Mithal A, Kaur P, Kalra S, Baruah MP, Mukhopadhyay S, et al. Widespread vitamin D deficiency among Indian health care professionals. Arch Osteoporos. 2012;7:187-92.
- Sasidharan PK, Rajeev E, Vijayakumari V. Tuberculosis and vitamin D deficiency in Kerala, India. Medicine Update. 2012;22:331-5.
- Ellison DL, Moran HR. Vitamin D: vitamin or hormone? Nurs Clin North Am. 2021;56(1):47-57.
- Sustainable Elimination of Iodine Deficiency. 2006. Available from: https://data.unicef.org/resources/sustainable-elimination-of-iodine-deficiency/. Accessed December 19, 2022.
- Pandav CS, Yadav K, Srivastava R, Pandav R, Karmarkar MG. Iodine deficiency disorders (IDD) control in India. Indian J Med Res. 2013;138(3):418-33.
- Kapil U. Successful effects toward elimination iodine deficiency disorders in India. Indian J Community Med. 2010;35(4):455-68.
- Demer LL, Hsu JJ, Tintut Y. Steroid hormone vitamin D. Circulat Res. 2018;122(11):1576-85.
- Cesari M, Incalzi RA, Zamboni V, Pahor M. Vitamin D hormone: a multitude of actions potentially influencing the physical function decline in older persons. Geriatr Gerontol Int. 2011;11(2):133-42.
- Hinkle J, Cheever K. Brunner and Suddarth’s Textbook of Medical-Surgical Nursing. 14th Edition, Philadelphia: Wolters Kluwer; 2018.
- Sweny P. Optimum route of administration of vitamin D in renal failure. Nephrol Dial Transplant. 1995;10 Suppl 4:
29-32.
- Lhamo Y, Chugh PK, Tripathi CD. Vitamin D supplements in the Indian market. Indian J Pharm Sci. 2016;78(1):41-7.
- Bilezikian JP, Formenti AM, Adler RA, Binkley N, Bouillon R, Lazaretti-Castro M, et al. Vitamin D: dosing, levels, form, and route of administration: does one approach fit all? Rev Endocr Metab Disord. 2021;22(4):1201-18.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al; Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-30.
- Laird E, Ward M, McSorley E, Strain JJ, Wallace J. Vitamin D and bone health: potential mechanisms. Nutrients. 2010;2(7):693-724.
- Uday S, Högler W. Nutritional rickets and osteomalacia in the twenty-first century: revised concepts, public health, and prevention strategies. Curr Osteoporos Rep. 2017;15(4):293-302.
- Sahay M, Sahay R. Rickets-vitamin D deficiency and dependency. Indian J Endocrinol Metab. 2012;16(2):164-76.
- Lips P, Schoor NVM. The effect of vitamin D on bone and osteoporosis. Best Pract Res Clin Endocrinol Metab. 2011;25(4):585-91.
- Wacker M, Holick MF. Vitamin D - Effects on skeletal and extraskeletal health and the need for supplementation. Nutrients. 2013;5(1):111-48.
- Uchitomi R, Oyabu M, Kamei Y. Vitamin D and sarcopenia: potential of vitamin D supplementation in sarcopenia prevention and treatment. Nutrients. 2020;12(10):3189.
- Remelli F, Vitali A, Zurlo A, Volpato S. Vitamin D deficiency and sarcopenia in older persons. Nutrients. Nutrients. 2019;11(12):2861.
- Kalra S, Kumar V, Kapoor N. The MOAN (Musculo-Osteo-Arthro-Neuropathic) syndrome. J Pak Med Assoc. 2022;72(2):373-4.
- Feldman D, Krishnan AV, Swami S, Giovannucci E, Feldman BJ. The role of vitamin D in reducing cancer risk and progression. Nat Rev Cancer. 2014;14(5):342-57.
- Bjelakovic G, Gluud LL, Nikolova D, Whitfield K, Krstic G, Wetterslev J, et al. Vitamin D supplementation for prevention of cancer in adults. Cochrane Database Syst Rev. 2014;(6):CD007469.
- Bouillon R, Marcocci C, Carmeliet G, Bikle D, White JH, Dawson-Hughes B, et al. Skeletal and extraskeletal actions of vitamin D: current evidence and outstanding questions. Endocr Rev. 2019;40(4):1109-51.
- Chen FY, Xiao M, Ling B, Liu L, Chen L. Vitamin D does not improve lung function decline in COPD: a meta-analysis. Eur Rev Med Pharmacol Sci. 2019;23(19):8637-44.
- Murdaca G, Tonacci A, Negrini S, Greco M, Borro M, Puppo F, Gangemi S. Emerging role of vitamin D in autoimmune diseases: an update on evidence and therapeutic implications. Autoimmun Rev. 2019;18(9):102350.
- Palacios C, De-Regil LM, Lombardo LK, Peña-Rosas JP. Vitamin D supplementation during pregnancy: updated meta-analysis on maternal outcomes. J Steroid Biochem Mol Biol. 2016;164:148-55.
- Barrea L, Savanelli MC, Di Somma C, Napolitano M, Megna M, Colao A, et al. Vitamin D and its role in psoriasis: an overview of the dermatologist and nutritionist. Rev Endocr Metab Disord. 2017;18(2):195-205.
- Marquina C, Mousa A, Scragg R, de Courten B. Vitamin D and cardiometabolic disorders: a review of current evidence, genetic determinants and pathomechanisms. Obes Rev. 2019;20(2):262-77.
- Mahmood LAG, Al Saadi R, Matthews L. Vitamin D deficiency and cardiometabolic syndrome: is the evidence solid? Arch Med Health Sci. 2017;5(2):229-36.
- Pittas AG, Chung M, Trikalinos T, Mitri J, Brendel M, Patel K, et al. Systematic review: vitamin D and cardiometabolic outcomes. Ann Intern Med. 2010;152(5):307-14.
- Salekzamani S, Mehralizadeh H, Ghezel A, Salekzamani Y, Jafarabadi MA, Bavil AS, et al. Effect of high-dose vitamin D supplementation on cardiometabolic risk factors in subjects with metabolic syndrome: a randomized controlled double-blind clinical trial. J Endocrinol Invest. 2016;39(11):1303-13.
- Qorbani M, Heidari-Beni M, Ejtahed HS, Shafiee G, Goodarzi F, Tamehri Zadeh SS, et al. Association of vitamin D status and cardio-metabolic risk factors in children and adolescents: the CASPIAN-V study. BMC Nutr. 2021;7(1):71.
- Pfeifer M, Begerow B, Minne HW, Nachtigall D, Hansen C. Effects of a short-term vitamin D(3) and calcium supplementation on blood pressure and parathyroid hormone levels in elderly women. J Clin Endocrinol Metab. 2001;86(4):1633-7.
- Parker J, Hashmi O, Dutton D, Mavrodaris A, Stranges S, Kandala NB, et al. Levels of vitamin D and cardiometabolic disorders: systematic review and meta-analysis. Maturitas. 2010;65(3):225-36.
- Wang H, Xia N, Yang Y, Peng DQ. Influence of vitamin D supplementation on plasma lipid profiles: a meta-analysis of randomized controlled trials. Lipids Health Dis. 2012;11:42.
- Tavakoli F, Namakin K, Zardast M. Vitamin D supplementation and high-density lipoprotein cholesterol: a study in healthy school children. Iran J Pediatr. 2016;26(4):e3311.
- Kuchay MS, Laway BA, Bashir MI, Wani AI, Misgar RA, Shah ZA. Effect of vitamin D supplementation on glycemic parameters and progression of prediabetes to diabetes: a 1-year, open-label randomized study. Indian J Endocrinol Metab. 2015;19(3):387-92.
- Gaudet M, Plesa M, Mogas A, Jalaleddine N, Hamid Q, Al Heialy S. Recent advances in vitamin D implications in chronic respiratory diseases. Respir Res. 2022;23(1):252.
- Ilyas M, Agussalim A, Megawati M, Massi N, Djaharuddin I, Bakri S, et al. Relationship between vitamin D level and serum TNF-α concentration on the severity of chronic obstructive pulmonary disease. Open Access Maced J Med Sci. 2019;7(14):2298-304.
- Mangas-Sánchez C, Garriga-García M, Serrano-Nieto MJ, García-Romero R, Álvarez-Beltrán M, Crehuá-Gaudiza E, et al. Vitamin D status in pediatric and young adult cystic fibrosis patients. Are the new recommendations effective? Nutrients. 2021;13(12):4413.
- Pludowski P, Takacs I, Boyanov M, Belaya Z, Diaconu CC, Mokhort T, et al. Clinical Practice in the prevention, diagnosis and treatment of vitamin D deficiency: A Central and Eastern European Expert Consensus Statement. Nutrients. 2022;14(7):1483.
- Zhao S, Gardner K, Taylor W, Marks E, Goodson N. Vitamin D assessment in primary care: changing patterns of testing. London J Prim Care (Abingdon). 2015;7(2):15-22.
- Kalra S. Vitamin D deficiency: pragmatic suggestions for prevention and treatment. J Pak Med Assoc. 2017;67(7):1116-8.
- Kennel KA, Drake MT, Hurley DL. Vitamin D deficiency in adults: when to test and how to treat. Mayo Clin Proc. 2010;85(8):752-8.
- Marcinowska-Suchowierska E, Kupisz-Urbanska M, Lukaszkiewicz J, Pludowski P, Jones G. Vitamin D toxicity - A clinical perspective. Front Endocrinol (Lausanne). 2018;9:550.
- Holick MF. Vitamin D and sunlight: strategies for cancer prevention and other health benefits. Clin J Am Soc Nephrol. 2008;3(5):1548-54.
- O’Mahony L, Stepien M, Gibney MJ, Nugent AP, Brennan L. The potential role of vitamin D enhanced foods in improving vitamin D status. Nutrients. 2011;3(12):1023-41.
- Al Khalifah R, Alsheikh R, Alnasser Y, Alsheikh R, Alhelali N, Naji A, et al. The impact of vitamin D food fortification and health outcomes in children: a systematic review and meta-regression. Syst Rev. 2020;9(1):144.
- Houghton LA, Vieth R. The case against ergocalciferol (vitamin D2) as a vitamin supplement. Am J Clin Nutr. 2006;84(4):694-7.
- WHO Model List of Essential Medicines 2021 and National List of Essential Medicines 2022.
- Sanders KM, Stuart AL, Williamson EJ, Simpson JA, Kotowicz MA, Young D, et al. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA. 2010;303(18):1815-22.
- Martineau AR, Jolliffe DA, Hooper RL, Greenberg L, Aloia JF, Bergman P, et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017;356:i6583.
- Pietras SM, Obayan BK, Cai MH, Holick MF. Vitamin D2 treatment for vitamin D deficiency and insufficiency for up to 6 years. JAMA Intern Med. 2009;169(19):1806-18.
- Hanley DA, Cranney A, Jones G, Whiting SJ, Leslie WD, Cole DE, et al; Guidelines Committee of the Scientific Advisory Council of Osteoporosis Canada. Vitamin D in adult health and disease: a review and guideline statement from Osteoporosis Canada. CMAJ. 2010;182(12):E610-8.
- Sadat-Ali M, Al-Anii FM, Al-Turki HA, AlBadran AA, AlShammari SM. Maintenance dose of vitamin D: how much is enough? J Bone Metab. 2018;25(3):161-4.
- Alamoudi LH, Almuteeri RZ, Al-Otaibi ME, Alshaer DA, Fatani SK, Alghamdi MM, et al. Awareness of vitamin D deficiency among the general population in Jeddah, Saudi Arabia. J Nutr Metab. 2019;2019:4138187.
- Alshamsan FM, Bin-Abbas BS. Knowledge, awareness, attitudes and sources of vitamin D deficiency and sufficiency in Saudi children. Saudi Med J. 2016;37(5):579-83.