ABSTRACT
An open-label prospective noncomparative study was conducted to examine the efficacy and safety of Beliv tablet, a polyherbal Ayurvedic medicine, in 30 adult patients suffering from liver disorders. Two tablets were administered daily for 56 days. Patients were evaluated at Day 0, Day 21, Day 42 and at Day 56. The primary end point of the study was a change in liver function test parameters measured by the levels of serum glutamic-oxaloacetic transaminase (SGOT), serum glutamic-pyruvic transaminase (SGPT) and total bilirubin at all assessment points. Results showed a significant reduction in the serum levels of SGOT, SGPT and total bilirubin from baseline to 21 days, 42 days and 56 days. The total icterus symptom score was also significantly reduced from 5.17 ± 2.26 (baseline) to 2.6 ± 1.48, 1.37 ± 1.13 and 0.77 ± 0.73 at Days 21, 42 and 56, respectively. The Subjective Global Assessment (SGA) decreased significantly from 3.33 ± 1.16 (Day 21) to 2.33 ± 1.16 and 1.80 ± 0.76 at Days 42 and 56, respectively. The Physician Global Assessment (PGA) score was also significantly reduced from 3.0 ± 1.02 (Day 21) to 2.07 ± 0.83 and 1.70 ± 0.79 at Days 42 and 56, respectively. A significant reduction in serum creatinine level was observed at Day 56. No adverse effects or serious adverse effects were observed during the study period. The study concluded that Beliv tablet was highly effective for the treatment of liver disorders, as evidenced by the reduction in serum levels of SGOT, SGPT, total bilirubin, icterus symptoms and PGA and SGA scores. No treatment-related side effects were reported by any of the study participants suggesting that it was safe for clinical use in humans for the treatment of liver disorders.
Keywords: Liver disorders, liver function tests, SGA score, PGA score, icterus symptom score
The liver is one of the largest organs in the human body, performing a variety of vital functions including metabolism, immunity, digestion, detoxification and vitamin storage.1 Annually, approximately 2 million people die as a result of liver diseases worldwide.1 Herbal products for liver disease have been used for a long time in India even though their therapeutic use has received considerable attention only recently.2 In Indian systems of medicine, there are over 300 formulations for the treatment of jaundice and chronic liver diseases, and over 87 medicinal plants are used in various combinations for liver diseases.3 Despite the significant numbers of preclinical studies, there is a lack of clinical trials and toxicological data on these plant products. Beliv is a polyherbal formulation. The constituent herbs include Phyllanthus niruri, Tinospora cordifolia, Picrorhiza kurroa, Azadirachta indica, Eclipta alba, Boerhaavia diffusa, Andrographis paniculata and Aloe barbadensis. The purpose of this phase IV post-marketing prospective study was to evaluate the safety and efficacy of the Beliv tablet in 30 adult volunteers suffering from liver disorders.
METHODS
This was a noncomparative open-label, single-arm clinical study for evaluating the efficacy and safety of Beliv tablets in subjects with mild to moderate liver dysfunction recruited from the outpatient clinics of the study center during their routine examination visit.
Male and female adults between the aged of 18 and 50 with abnormal liver function tests, based on complete medical history, physical examination and other laboratory tests, subjects with elevated alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels ≥1.5 times the upper limit of normal (ULN), subjects without life-threatening conditions and those who were willing to sign consent forms and were able to be present for follow-ups were included in the study.
The exclusion criteria were pregnant women or women with childbearing potential who were likely to be pregnant, subjects with severe alcoholic hepatitis with cirrhosis of liver or life expectancy less than 3 months or those with a history of severe renal failure (estimated glomerular filtration [eGFR] <60 mL/min per 1.73 m2), liver disorders from cardiac causes, hereditary metabolic causes, hemochromatosis and Wilson’s diseases or any other condition that, in the investigator’s opinion, would adversely affect the ability of the subject to complete the study or its measurements. Subjects who had participated in another clinical trial with an active intervention or drug or device with the last dose taken within 60 days were also not included in the trial.
No concomitant medication was allowed during the study. However, if participants reported any clinical symptoms during the study, the study physician prescribed the appropriate medication, which was documented.
The primary end point of the study was a change in liver function test parameters from baseline to the end of the study measured by the levels of serum glutamic-oxaloacetic transaminase (SGOT), serum glutamic-pyruvic transaminase (SGPT) and total bilirubin at all assessment points.
The secondary end points were:
- Change in icterus sign and symptom scores from baseline to end of treatment using a predefined 4-point scale.
- Change in Subjective Global Assessment (SGA) scores from baseline to end of treatment using a predefined 5-point scale.
- Change in Physician’s Global Assessment (PGA) scores from baseline to end of treatment using a predefined 5-point scale.
- Change in general health symptoms, vital signs, hematology, renal function, serum lipids and urinalysis parameters from baseline to end of treatment.
After baseline assessment at Visit 1, 30 patients were administered two Beliv tablets per day for 56 days. They were evaluated at 4 assessment points: baseline (Visit 1, Day 0), follow-ups (Visits 2 and 3, Days 21 and 42, respectively) and final (Visit 4, Day 56) visits. Informed consent was obtained from all participants at Visit 1 (Day 0). The study was carried out according to the Declaration of Helsinki, Indian Council of Medical Research (ICMR) ethical guidelines for biomedical research and International Conference on Harmonization (ICH) guidelines for Good Clinical Practice (GCP).
At Visit 1 (Day 0), all subjects were assessed for baseline data, including physical examinations, vital signs, demographic data, medical histories, concomitant medications, efficacy and safety parameters.
After 3 weeks of treatment, all subjects underwent physical examination at follow-up visits and were assessed for vital signs, liver function test, icterus sign and symptoms, PGA and SGA scores, concomitant medications and adverse events. At the final follow-up visit, patients underwent tests for complete blood count (CBC), kidney function test (KFT), lipid profile and urine analysis.
Data were abstracted and presented as a number, percentage, mean and standard deviation (SD). All efficacy and safety variables were summarized using descriptive statistics. Data comparison between baseline and follow-up visits was performed using a paired t-test or one-way analysis of variance (ANOVA), as appropriate and data were expressed as mean, SD, 95% confidence interval (CI) and p-value. A p value of 0.05 was considered statistically significant.
RESULTS
A total of 30 subjects were enrolled in the study, which included 19 (63.33%) females and 11 (36.37%) males.
All of them completed the study; hence, the data of 30 subjects was analyzed for this study. The demographic information of the participants is summarized in Table 1.
|
Table 1. Summary of Demographic Data of the Subjects (n = 30)
|
|
Parameters
|
Range
|
Number (%)
|
Mean
|
SD
|
|
Age (year)
|
21-50
|
-
|
36.33
|
8.34
|
|
Male
|
-
|
11 (36.37)
|
-
|
-
|
|
Female
|
-
|
19 (63.33)
|
-
|
-
|
|
Height (cm)
|
146-180
|
-
|
164.10
|
8.62
|
|
Weight (kg)
|
50.10-86.50
|
-
|
65.35
|
8.58
|
Efficacy Assessment
Assessment of liver function
Results showed a significant reduction in the serum levels of SGOT, SGPT and total bilirubin from baseline (V1) to 21 days (V2), 42 days (V3) and 56 days (V4). Changes were found to be statistically significant (p < 0.0001) compared to the baseline (Table 2 and Fig. 1).
|
Table 2. Comparison of Mean Change in Liver Function Parameters from Baseline to Different Follow-up Visits
|
|
Parameters
|
Visit (V)
|
Mean ± SD (n = 30)
|
Change from V1 (mean ± SD)
|
95% CI of Diff.
|
P value
|
|
SGOT (IU/L)
|
V1
V2
V3
V4
|
124.20 ± 26.55
92.03 ± 18.79
64.17 ± 8.99
32.69 ± 6.12
|
-
32.22 ± 15.17
60.08 ± 24.12
91.56 ± 26.38
|
-
20.33-44.10
48.19-71.96
79.68-103.4
|
<0.0001***
|
|
SGPT (IU/L)
|
V1
V2
V3
V4
|
109.0 ± 20.79
83.38 ± 11.46
58.21 ± 5.83
30.92 ± 7.16
|
-
25.60 ± 14.63
50.77 ± 19.75
78.06 ± 23.14
|
-
16.78-34.43
41.95-59.60
69.24-86.89
|
<0.0001***
|
|
Total bilirubin (mg/dL)
|
V1
V2
V3
V4
|
1.06 ± 0.38
0.79 ± 0.17
0.64 ± 0.14
0.55 ± 0.20
|
-
0.27 ± 0.27
0.42 ± 0.35
0.47 ± 0.50
|
-
0.10-0.445
0.24-0.593
0.302-0.647
|
<0.0001***
|
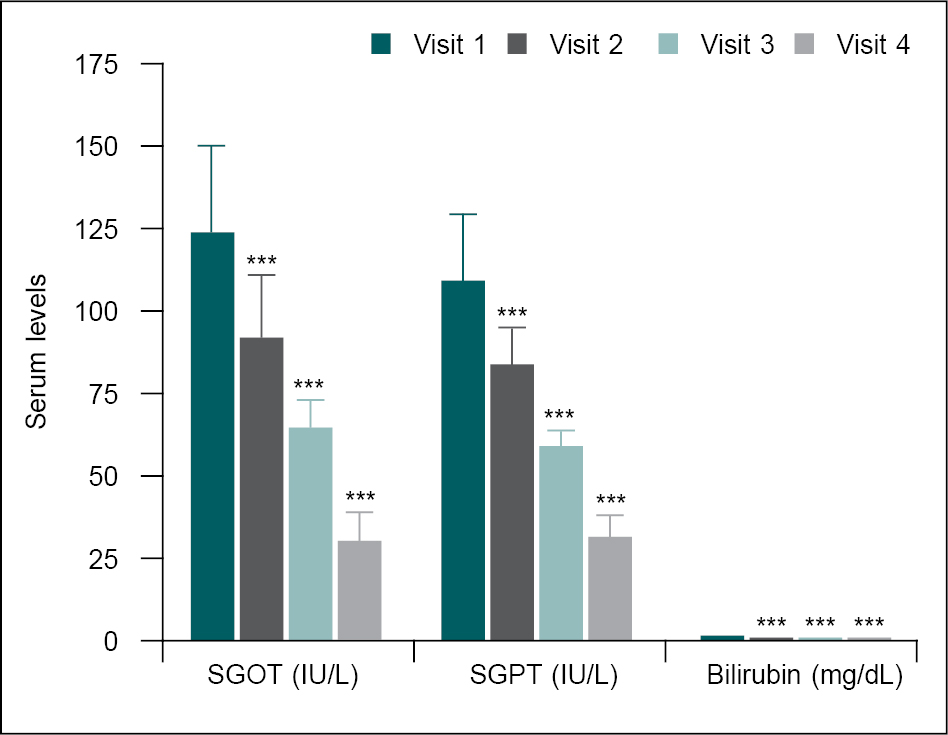
Figure 1. Comparison of mean change in liver function parameters at different visits.
Liver function parameters at baseline (V1) were compared with follow-up visits (V2, V3 and V4) using a one-way ANOVA test. Comparison: V1 vs. V2, V3 and V4. Significance level ***p < 0.001.
Assessment of icterus symptoms
Icterus symptoms such as fatigue, loss of appetite, itching, nausea/vomiting were assessed on a 4-point scale at all assessment points. All icterus symptom scores were significantly (p < 0.0001) reduced at Days 21 (follow-up visit), 42 (follow-up visit) and 56 (final study visit) compared to baseline (Table 3 and Fig. 2). The total icterus symptom score was also significantly reduced from 5.17 ± 2.26 (baseline) to 2.6 ± 1.48, 1.37 ± 1.13 and 0.77 ± 0.73 at Days 21, 42 and 56, respectively.
|
Table 3. Comparison of Mean Change in Icterus Symptom Scores on a 4-Point Scale from Baseline to Different Follow-up Visits (n = 30)
|
|
Parameters
|
Visit (V)
|
Mean score (mean ± SD)
|
Change from V1 (mean ± SD)
|
95% CI of Diff.
|
|
Fatigue
|
V1
V2
V3
V4
|
1.23 ± 0.77
0.77 ± 0.68
0.50 ± 0.63
0.27 ± 0.45
|
-
0.47 ± 0.57**
0.73 ± 0.78***
0.97 ± 0.81***
|
-
0.0709-0.862
0.338-1.129
0.571-1.362
|
|
Loss of appetite
|
V1
V2
V3
V4
|
1.23 ± 0.94
0.67 ± 0.66
0.33 ± 0.48
0.13 ± 0.35
|
-
0.57 ± 0.63**
0.90 ± 0.84***
1.10 ± 0.96***
|
-
0.171-0.963
0.504-1.296
0.704-1.496
|
|
Itching
|
V1
V2
V3
V4
|
0.87 ± 0.78
0.43 ± 0.50
0.27 ± 0.45
0.20 ± 0.41
|
-
0.43 ± 0.68*
0.60 ± 0.77***
0.67 ± 0.92***
|
-
0.093-0.773
0.260-0.940
0.327-1.007
|
|
Nausea/vomiting
|
V1
V2
V3
V4
|
1.83 ± 1.05
0.73 ± 0.69
0.27 ± 0.45
0.17 ± 0.38
|
-
1.10 ± 0.96***
1.57 ± 1.07***
1.67 ± 1.15***
|
-
0.673-1.527
1.139-1.994
1.239-2.094
|
|
Total score
|
V1
V2
V3
V4
|
5.17 ± 2.26
2.60 ± 1.48
1.37 ± 1.13
0.77 ± 0.73
|
-
2.57 ± 1.50***
3.80 ± 2.11***
4.40 ± 2.30***
|
-
1.904-3.830
2.837-4.763
3.437-5.363
|
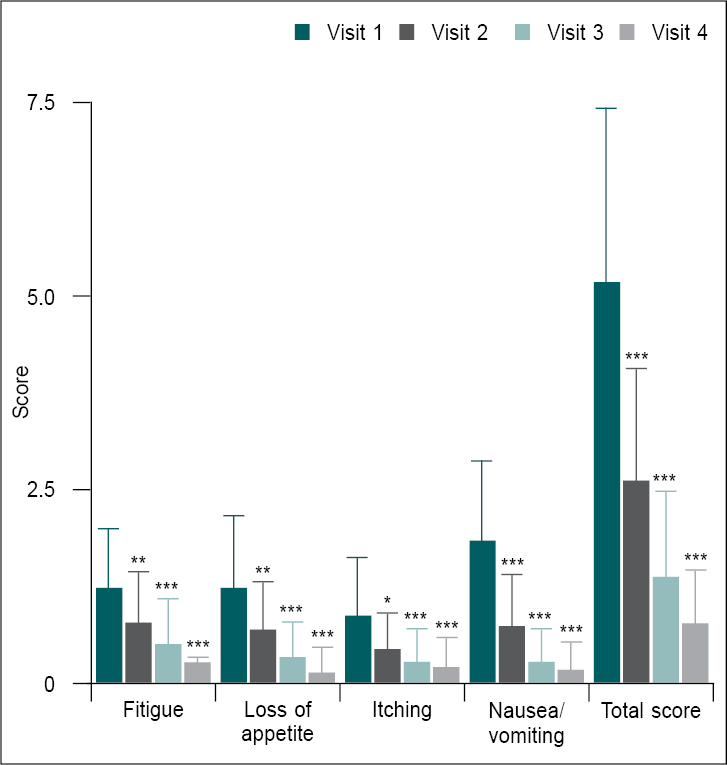
Figure 2. Comparison of mean change in icterus symptom scores from baseline (V1) to different follow-up visits (n = 30).
Icterus symptom scores at V1 were compared with follow-up visits (V2, V3 and V4) using one-way ANOVA. Comparison: V1 vs. V2, V3 and V4. Significance levels *p < 0.05,
**p < 0.01 and ***p < 0.001.
Assessment of SGA and PGA
When compared to Day 21, both the SGA and PGA scores were significantly (p < 0.0001) lower on Days 42 (follow-up visit) and 56 (final study visit) (Table 4 and Fig. 3). The SGA score decreased significantly from 3.33 ± 1.16 (Day 21) to 2.33 ± 1.16 and 1.80 ± 0.76 at Days 42 and 56, respectively. The PGA score was also significantly reduced from 3.0 ± 1.02 (Day 21) to 2.07 ± 0.83 and 1.70 ± 0.79 at Days 42 and 56, respectively.
|
Table 4. Comparison of Mean Change in SGA and PGA Scores from Day 21 to Days 42 and 56
|
|
Parameters
|
Visit (V)
|
Mean ± SD
(n = 30)
|
Change from V1 (mean ± SD)
|
95% CI of Diff.
|
P value
|
|
SGA
|
V2
V3
V4
|
3.33 ± 1.16
2.33 ± 1.16
1.80 ± 0.76
|
-
0.93 ± 0.64
1.53 ± 0.82
|
-
0.396-1.604
0.929-2.137
|
<0.0001***
|
|
PGA
|
V2
V3
V4
|
3.0 ± 1.02
2.07 ± 0.83
1.70 ± 0.79
|
-
0.93 ± 0.64
1.30 ± 1.24
|
-
0.419-1.447
0.786-1.814
|
<0.0001***
|
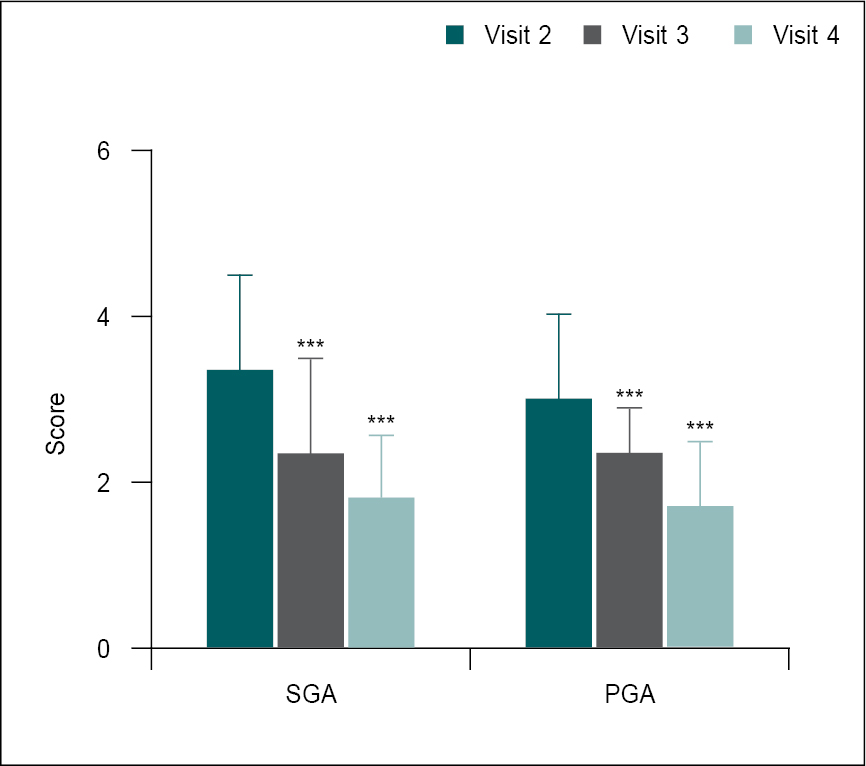
Figure 3. Comparison of mean change in SGA and PGA scores from Day 21 (V2) to Days 42 (V3) and 56 (V4) (n = 30).
SGA and PGA scores in V2 were compared with V3 and V4 using the one-way ANOVA test. Comparison: V2 vs. V3 and V4. Significance level ***p < 0.001.
Assessment of other symptoms
Other liver disorder-related symptoms, such as fever, abdominal pain, flu-like symptoms, change in skin color, dark-colored urine and clay-colored stool, were also monitored at all assessment points during the treatment period. At baseline, some subjects experienced fever (4, 13.33%), abdominal pain (3, 10%), flu-like symptoms (2, 6.67%), change in skin color (2, 6.67%), dark-colored urine (3, 10%) and clay-colored stools (6, 20%) (Table 5). After 21 days of treatment, only 1 subject (3.33%) had a change in skin color, and 4 subjects (13.33%) had clay-colored stools.
However, none of the above-mentioned symptoms were observed after 42 and 56 days of treatment with Beliv.
|
Table 5. Other Signs/Symptoms of Liver Disorders among the Participants (n = 30)
|
|
Other signs/symptoms
|
Visit 1 % (n)
|
Visit 2 % (n)
|
Visit 3 % (n)
|
Visit 4 % (n)
|
|
Fever
|
13.33 (4)
|
0
|
0
|
0
|
|
Abdominal pain
|
10 (3)
|
0
|
0
|
0
|
|
Flu-like symptoms
|
6.67 (2)
|
0
|
0
|
0
|
|
Change in skin color
|
6.67 (2)
|
3.33 (1)
|
0
|
0
|
|
Dark-colored urine
|
10 (3)
|
0
|
0
|
0
|
|
Clay-colored stool
|
20 (6)
|
13.33 (4)
|
0
|
0
|
Safety Assessment
Assessment of general health symptoms
No abnormalities were observed in the subjects’ general appearance, eyes, ear, nose, throat, abdomen, heart, chest and ECG profile during all study visits.
Assessment of vital signs
All subjects’ vital sign parameters, including heart rate, systolic and diastolic blood pressure, were measured at all assessment points. The baseline vital signs data were compared to the day obtained on Day 21, Day 42 and Day 56. No significant differences in vital signs were observed all through the treatment period (Table 6 and Fig. 4).
|
Table 6. Comparison of Mean Change in Vital Signs from Baseline to Different Follow-up Visits
|
|
Parameters
|
Visit (V)
|
Mean ± SD
(n = 30)
|
Change from V1 (mean ± SD)
|
95% CI of Diff.
|
P value
|
|
Heart rate (bpm)
|
V1
V2
V3
V4
|
67.37 ± 6.37
65.53 ± 4.64
66.47 ± 4.49
67.30 ± 4.44
|
-
1.33 ± 5.42
0.90 ± 4.16
0.07 ± 5.56
|
-
-1.665 to 5.332
-2.599 to 4.399
-3.432 to 3.565
|
0.459
|
|
SBP
(mm/Hg)
|
V1
V2
V3
V4
|
120.3 ± 7.18
121.0 ± 6.99
120.0 ± 6.74
119.6 ± 6.87
|
-
-0.67 ± 4.71
0.33 ± 4.25
0.77 ± 4.99
|
-
-5.482 to 4.149
-4.482 to 5.149
-4.049 to 5.582
|
0.877
|
|
DBP
(mm/Hg)
|
V1
V2
V3
V4
|
79.20 ± 2.94
79.60 ± 2.85
78.77 ± 3.18
79.90 ± 2.62
|
-
-0.40 ± 2.27
0.43 ± 3.37
-0.70 ± 3.57
|
-
-2.412 to 1.612
-1.579 to 2.446
-2.712 to 1.312
|
0.463
|
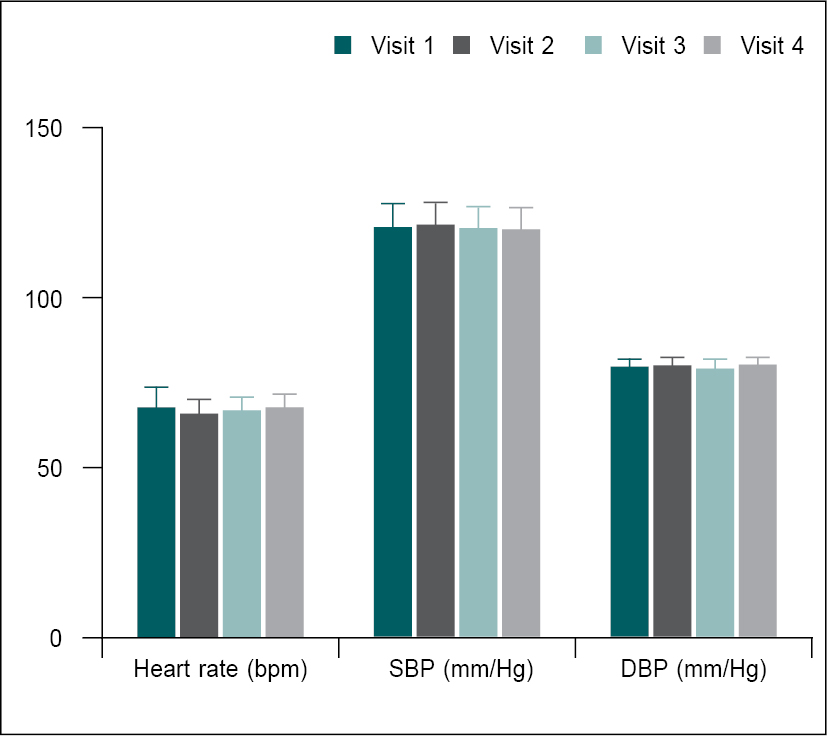
Figure 4. Comparison of mean change in vital signs from baseline to different follow-up visits (n = 30).
Vital signs data at baseline (V1) were compared to Days 21 (V2), 42 (V3) and 56 (V4) using the one-way ANOVA test. Comparison: V1 vs. V2, V3 and V4.
Assessment of complete blood profile
CBP was evaluated at the screening (V1) and end of the study (V4). There was no significant difference in the CBP parameters between screening and Day 56 data, and all values were within the normal range (Table 7 and Fig. 5).
|
Table 7. Comparison of Mean Change in CBP Parameters from Screening to Day 56
|
|
Parameters
|
Visit (V1)
|
Mean ± SD (n = 30)
|
Change from V1 (mean ± SD)
|
95% CI of Diff.
|
P value
|
|
RBC (millions/cumm)
|
V1
V4
|
5.28 ± 0.34
5.34 ± 0.47
|
-0.06 ± 0.37
|
-0.200-0.0768
|
0.370
|
|
Hb (g/dL)
|
V1
V4
|
13.53 ± 1.47
13.67 ± 1.18
|
-0.13 ± 1.05
|
-0.526-0.260
|
0.495
|
|
WBC (10^9/L)
|
V1
V4
|
7.37 ± 0.96
7.37 ± 0.51
|
0.18 ± 0.71
|
-0.086-0.443
|
0.179
|
|
Neutrophils (%)
|
V1
V4
|
51.21 ± 5.86
50.74 ± 4.23
|
0.47 ± 4.21
|
-1.102-2.040
|
0.546
|
|
Lymphocytes (%)
|
V1
V4
|
36.78 ± 5.71
37.50 ± 3.92
|
-0.72 ± 4.25
|
-2.306-0.868
|
0.362
|
|
Monocytes (%)
|
V1
V4
|
7.44 ± 1.02
7.31 ± 0.78
|
0.14 ± 0.83
|
-0.172-0.446
|
0.373
|
|
Eosinophils (%)
|
V1
V4
|
3.96 ± 1.06
3.89 ± 0.97
|
0.07 ± 0.93
|
-0.275-0.422
|
0.70
|
|
Basophils (%)
|
V1
V4
|
0.60 ± 0.20
0.56 ± 0.14
|
0.04 ± 0.19
|
-0.030-0.110
|
0.26
|
|
Platelet count
(x1000/cumm)
|
V1
V4
|
336.7 ± 50.27
337.1 ± 51.45
|
-0.37 ± 18.61
|
-7.315-6.582
|
0.91
|
|
ESR (mm 1st hour)
|
V1
V4
|
21.33 ± 11.26
21.80 ± 8.06
|
-0.47 ± 5.51
|
-2.525-1.592
|
0.646
|
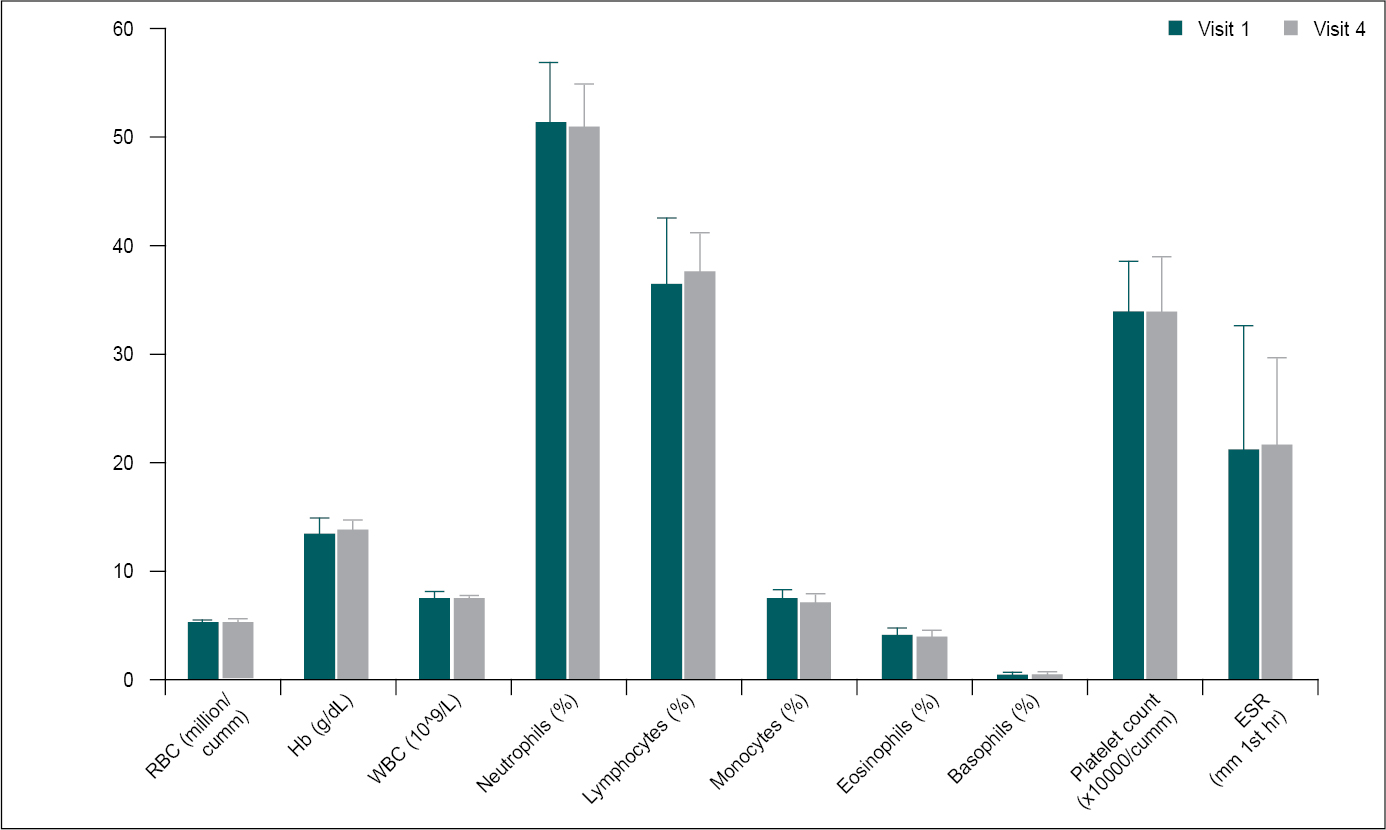
Figure 5. Comparison of mean change in CBP parameters from screening to Day 56 (n = 30).
CBP parameters at screening (V1) were compared to Day 56 (V4) using paired t-test. Comparison: V1 vs. V4.
Assessment of renal function
Serum levels of blood urea nitrogen (BUN) and creatinine were measured at baseline and Day 56 to assess renal function. The serum levels of BUN and creatinine were in the normal range at both assessment points, but the reduction in serum creatinine level at Day 56 was statistically significant (p < 0.05) (Table 8 and Fig. 6).
|
Table 8. Comparison of Mean Change in Renal Function Parameters from Baseline to Day 56
|
|
Parameters
|
Visit (V)
|
Mean ± SD
(n = 30)
|
Change from V1
(mean ± SD)
|
95% CI of Diff.
|
P value
|
|
BUN (mg/dL)
|
V1
V4
|
11.18 ± 3.65
10.73 ± 2.94
|
0.45 ± 4.53
|
-1.236-2.143
|
0.587
|
|
Serum creatinine (mg/dL)
|
V1
V4
|
0.94 ± 0.25
0.81 ± 0.17
|
-0.13 ± 0.32
|
0.0116-0.250
|
0.033*
|
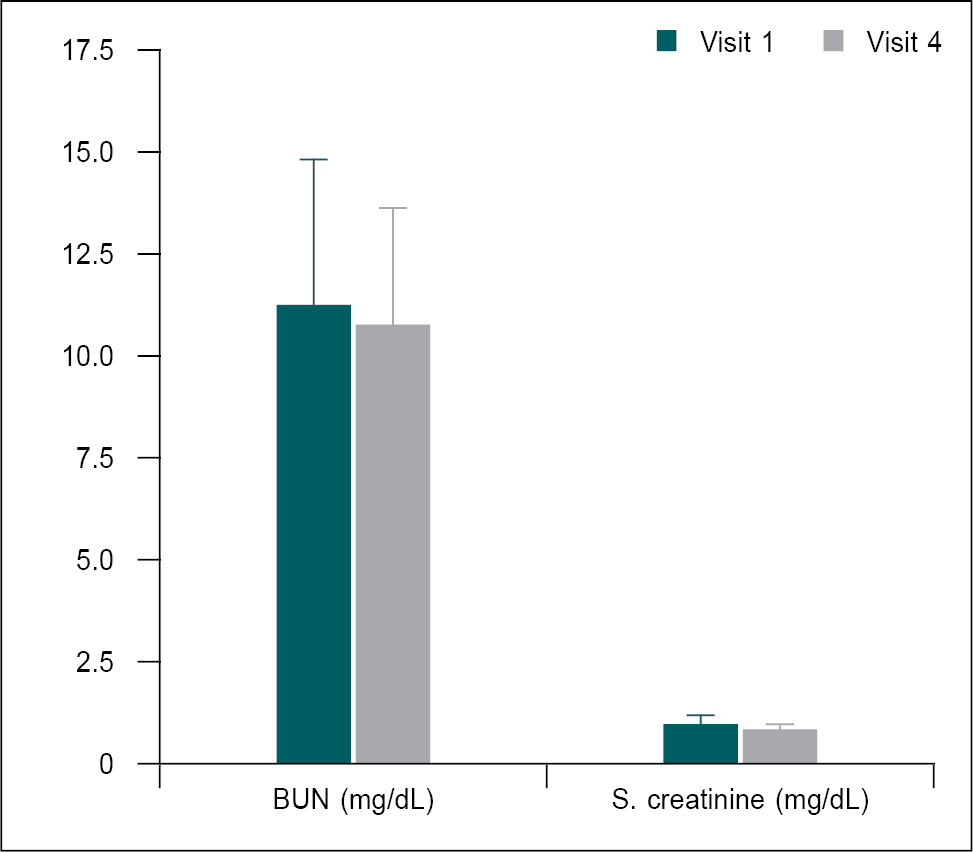
Figure 6. Comparison of mean change in renal function parameters from the baseline (V1) to the Day 56 (V4) (n = 30).
Renal function parameters at baseline (V1) were compared with Day 56 (V4) using the paired t-test. Comparison: V1 vs. V4. Significance level *p < 0.05.
Assessment of Serum Lipids
Serum cholesterols, including total cholesterol, triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), very-low-density lipoprotein (VLDL) cholesterols, as well as the Chol:HDL ratio, were measured at baseline and Day 56. The serum levels of total cholesterol, TG, LDL, VLDL cholesterols, and the Chol:HDL ratio were reduced and HDL increased at Day 56 when compared to baseline (Table 9 and Fig. 7).
|
Table 9. Comparison of Mean Change in Serum Lipids from Baseline to Day 56
|
|
Parameters
|
Visit (V)
|
Mean ± SD (n = 30)
|
Change from V1 (mean ± SD)
|
95% CI of Diff.
|
P value
|
|
Total cholesterol (mg/dL)
|
V1
V4
|
164.3 ± 28.44
153.9 ± 22.91
|
10.39 ± 13.25
|
5.443-15.34
|
0.0002***
|
|
TG (mg/dL)
|
V1
V4
|
109.50 ± 30.21
98.55 ± 24.17
|
10.95 ± 11.40
|
6.692-15.20
|
<0.0001***
|
|
HDL (mg/dL)
|
V1
V4
|
67.09 ± 4.03
70.45 ± 3.44
|
5.78 ± 10.97
|
1.681-9.873
|
0.0073**
|
|
LDL (mg/dL)
|
V1
V4
|
94.10 ± 17.34
89.42 ± 11.14
|
4.68 ± 19.22
|
-2.496-11.86
|
0.193
|
|
VLDL (mg/dL)
|
V1
V4
|
28.46 ± 7.33
25.37 ± 4.38
|
3.09 ± 8.86
|
-0.217-6.401
|
0.066
|
|
Chol:HDL ratio (mg/dL)
|
V1
V4
|
2.46 ± 0.45
2.19 ± 0.35
|
-0.075 ± 0.58
|
-0.290-0.141
|
0.484
|

Figure 7. Comparison of mean change in serum lipids from the baseline to Day 56 (n = 30).
Serum lipid parameters at baseline (V1) were compared with Day 56 (V4) using the paired t-test. Comparison: V1 vs. V4. Significance levels **p < 0.01 and ***p < 0.001.
Despite the fact that all serum cholesterol levels were within the normal range, there was a statistically significant change in total cholesterol, TG and HDL levels from baseline.
Urinalysis
Urine analysis of all participants was performed at baseline and Day 56. Urinalysis parameters showed no significant difference between two evaluation points.
Adverse events
No adverse effects or serious adverse effects were observed during the study period. However, three participants at Visit 2, two participants at Visits 3 and 4 experienced mild abdominal discomfort; however, this effect was not related to Beliv treatment.
REVIEW OF LITERATURE
Beliv is a polyherbal formulation, which has been used to treat various liver disorders. It contains T. cordifolia, P. kurroa, A. indica, E. alba, B. diffusa, A. paniculata, A. barbadensis and P. niruri, which have been shown to be hepatoprotective in studies. These herbs also have anti-inflammatory, antimicrobial, antiviral, antiparasitic, antidiabetic, immunomodulator activity, antioxidant and antitumor properties, which are complementary to the hepatoprotective activity and are desirable in hepatic disorders.
The hepatoprotective property of T. cordifolia, commonly known as Giloy, was examined in an experimental study in hepatic damage caused by carbon tetrachloride (CCl4). The liver enzymes (SGPT, SGOT, serum alkaline phosphatase [ALP]) and serum bilirubin returned to normal levels following administration of extract of T. cordifolia.4 It also exhibited antifibrotic effect in liver injury models, which has been hypothesized to occur via activation of Kupffer cells, the liver macrophages.5
In another study of an animal model of lead-induced liver injury, the aqueous extracts of stem and leaves of T. cordifolia lowered the levels of transaminases and ALP and increased the antioxidant enzymes superoxide dismutase and catalase.6
Administration of hydroalcoholic extract of Kutki or P. kurroa for 4 weeks led to reversal of the fatty infiltration in a study of experimental nonalcoholic fatty liver disease (NAFLD). The liver fat content also reduced - 38.33 for 200 mg/kg and 29.44 for 400 mg/kg of P. kurroa compared to 130.07 mg/g of liver tissue in the high-fat diet group.7 In a randomized placebo-controlled trial, patients with acute viral hepatitis (HBsAg negative) were treated with the root powder of P. kurroa in doses of 375 mg thrice a day for 2 weeks. A significant decrease in liver enzymes (SGPT 86 IU vs. 221 IU; SGOT 44 IU vs. 83 IU) and total serum bilirubin (4.15 mg/dL vs. 6.78 mg/dL) was seen compared to placebo. A rapid and marked improvement in symptoms of anorexia, nausea and malaise was also noted.8 P. kurroa has been shown to reduce the ethanol-induced oxidative stress because of its antioxidant activity.9
The hepatoprotective activity of A. indica, commonly called Neem, has been demonstrated in a study of paracetamol-induced liver damage in rats and has been attributed to its antioxidant activity as evident by the increase in the levels of glutathione-dependent enzymes in the liver and activity of superoxide dismutase and catalase.10,11 The leaf extract of A. indica ameliorated the hepatotoxicity induced by antitubercular drugs in rats.12,13
- diffusa or punarnava has been widely used for its hepatoprotective property.14 In a preclinical study, oral administration of an alcoholic extract of the whole plant improved the levels of serum ALT, TGs, cholesterol and total lipid in CCl4-induced hepatotoxicity. Enhancement in normal bile flow was indicative of a strong choleretic activity.15 It is also protective against paracetamol- and ethanol-induced hepatotoxicity.16
- barbadensis or Aloe vera has been called the “miracle plant” because of its multitude of beneficial actions. It contains enzymes that aid digestion and functions of the liver such as detoxification.17 Several animal model studies have suggested its hepatoprotective and antifibrotic effects.18 In mice fed a high-fat and fructose diet, A. vera significantly reduced the levels of cholesterol and improved the activities of superoxide dismutase and catalase indicative of the antioxidant effects of A. vera.19
The antihepatotoxic, antilithic, anti-HIV and anti-hepatitis B properties of P. niruri have been explored in literature.20 The aqueous extracts of the herb has been demonstrated to have radical scavenging activity including hepatoprotective activity. It also has protective effect against paracetamol-induced hepatotoxicity.21 The efficacy of P. niruri in patients with mild to moderate alcoholic hepatitis was evaluated in a randomized, double-blind, parallel-arm placebo-controlled trial. After 4 weeks of treatment with P. niruri, an improvement in the total antioxidant levels was observed. It also increased the appetite. The liver function parameters showed an improvement, but this was not statistically significant.22
The extract of E. alba or Bhringraj was protective against CCl4-induced acute liver damage and paracetamol-induced hepatotoxicity in animal studies.23 Luteolin, wedelolactone and apigenin are the three major active phytoconstituents of the herb. These compounds have been shown to in vitro inhibit RNA-dependent RNA polymerase (RdRp) activity of hepatitis C virus (HCV) replicase; they also exhibited anti-HCV replication activity in cell culture systems thereby reducing the HCV RNA titer. These properties suggest E. alba as a potential complementary treatment of hepatitis C.24 In animals with high-fat diet, E. alba reduced mononuclear infiltration, fat deposition and necrotic foci in the liver and also stimulated hepatocyte regeneration.25
Similar to the above-mentioned herbs, A. paniculata or Kalmegh was also protective against hepatotoxicity induced by CCl4, paracetamol and ethanol. This hepatoprotective activity was exerted via andrographolide, its major antihepatotoxic component.26 In patients with infective hepatitis, Kalmegh administered as a decoction improved the symptoms and liver biochemistry by reducing levels of serum bilirubin, ALP and the serum aminotransferases (SGOT and SGPT). This led to cure in 80% of the patients.27 Its antiviral activity has been demonstrated in patients with acute viral hepatitis.
A marked improvement in symptoms and liver enzymes was seen in patients receiving Kalmegh compared to the control group; 80.81% vs. 33.46%, respectively after 1 month, which was statistically significant.28
CONCLUSION
The results of the present clinical study demonstrated that the polyherbal formulation Beliv is highly effective for the treatment of liver disorders, as evidenced by the reduction in serum levels of SGOT, SGPT, total bilirubin, icterus symptoms and PGA and SGA scores. Furthermore, during the 56-day treatment period, no significant changes in vital signs, hematological profile, lipid profile, renal function or urinalysis parameters were observed. No treatment-related side effects were reported by any of the study participants suggesting that Beliv tablet is highly effective and safe for clinical use in humans for the treatment of liver disorders. All these effective actions are attributed to synergistic actions of its hepatoprotective ingredients.
Conflict of Interest
The author has no conflict of interest.
Disclosures
None.
REFERENCES
- Ghosh N, Ghosh R, Mandal V, Mandal SC. Recent advances in herbal medicine for treatment of liver diseases. Pharm Biol. 2011;49(9):970-88.
- Dhiman RK, Chawla YK. Herbal medicines for liver diseases. Dig Dis Sci. 2005;50(10):1807-12.
- Thyagarajan SP, Jayaram S, Gopalakrishnan V, Hari R, Jeyakumar P, Sripathi MS. Herbal medicines for liver diseases in India. J Gastroenterol Hepatol. 2002;17(Suppl 3):S370-6.
- Upadhyay AK, Kumar K, Kumar A, Mishra HS. Tinospora cordifolia (Willd.) Hook. f. and Thoms. (Guduchi) - validation of the Ayurvedic pharmacology through experimental and clinical studies. Int J Ayurveda Res. 2010;1(2):112-21.
- Nagarkatti DS, Rege NN, Desai NK, Dahanukar SA. Modulation of Kupffer cell activity by Tinospora cordifolia in liver damage. J Postgrad Med. 1994;40(2):65-7.
- Sharma V, Pandey D. Protective role of Tinospora cordifolia against lead-induced hepatotoxicity. Toxicol Int. 2010;17(1):12-7.
- Shetty SN, Mengi S, Vaidya R, Vaidya AD. A study of standardized extracts of Picrorhiza kurroa Royle ex Benth in experimental nonalcoholic fatty liver disease. J Ayurveda Integr Med. 2010;1(3):203-10.
- Vaidya AB, Antarkar DS, Doshi JC, Bhat AD, Ramesh V, Vora PV, et al. Picrorhiza kurroa(Kutaki) Royle ex Benth as a hepatoprotective agent - experimental & clinical studies. J Postgrad Med. 1996;42(4):105-8.
- Masood M, Arshad A, Qureshi R, Sabir S, Amjad MS, Qureshi H, et al. Picrorhiza kurroa: an ethnopharmacologically important plant species of Himalayan region. Pure Appl Biol. 2015;4(3):407-17.
- Chattopadhyay RR, Sarkar SK, Ganguly S, Basu TK, Mukherjee A. Hepatoprotective activity of Azadirachta indica leaves on paracetamol induced hepatic damage in rats. Indian J Exp Biol. 1992;30(8):738-40.
- Chattopadhyay RR, Bandyopadhyay M. Possible mechanism of hepatoprotective activity of Azadirachta indica leaf extract against paracetamol-induced hepatic damage in rats: part III. Indian J Pharmacol. 2005;37:184-5.
- Alzohairy MA. Therapeutics role of Azadirachta indica (Neem) and their active constituents in diseases prevention and treatment. Evid Based Complement Alternat Med. 2016;2016:7382506.
- Kale BP, Kothekar MA, Tayade HP, Jaju JB, Mateenuddin M. Effect of aqueous extract of Azadirachta indicaleaves on hepatotoxicity induced by antitubercular drugs in rats. Indian J Pharmacol. 2003;35:177-80.
- Nayak P, Thirunavoukkarasu M. A review of the plant Boerhaavia diffusa: its chemistry, pharmacology and therapeutical potential. J Phytopharmacol. 2016;5(2):83-92.
- Chandan BK, Sharma AK, Anand KK. Boerhaavia diffusa: A study of its hepatoprotective activity. J Ethnopharmacol. 1991;31(3):299-307.
- Kumar R, Gautam S, Singh KD, Kumar P, Haque N, Yadav V, et al. Pharmacological properties of Boerhavia diffusa: a review. IJCS. 2018;6(4):72-80.
- Kumar KP, Bhowmik D, Chiranjib, Biswajit. Aloe vera: a potential herb and its medicinal importance. J Chem Pharm Res. 2010;2(1):21-9.
- Hoogenboom TC, Patel N, Cook NA, Williams R, Taylor-Robinson SD, Lim AKP. The effect of Aloe vera juice on liver enzymes and hepatic structure in a healthy population. Integr Med (Encinitas). 2020;19(3):30-4.
- Abubakar AM, Dibal NI, Attah MO, Chiroma SM. Exploring the antioxidant effects of Aloe vera: potential role in controlling liver function and lipid profile in high fat and fructose diet (HFFD) fed mice. Pharmacol Res - Modern Chinese Med. 2022;4:100150.
- Bagalkotkar G, Sagineedu SR, Saad MS, Stanslas J. Phytochemicals from Phyllanthus niruri and their pharmacological properties: a review. J Pharm Pharmacol. 2006;58(12):1559-70.
- Narendra K, Swathi J, Sowjanya KM, Satya AK. Phyllanthus niruri: A review on its ethno botanical, phytochemical and pharmacological profile. J Pharm Res. 2012;5(9):4681-91.
- Sowjanya K, Girish C, Bammigatti C, Prasanna Lakshmi NC. Efficacy of Phyllanthus niruri on improving liver functions in patients with alcoholic hepatitis: a double-blind randomized controlled trial. Indian J Pharmacol. 2021;53(6):448-45.
- Jahan R, Al-Nahain A, Majumder S, Rahmatullah M. Ethnopharmacological significance of Eclipta alba (L.) Hassk. (Asteraceae). Int Sch Res Notices. 2014;2014:385969.
- Manvar D, Mishra M, Kumar S, Pandey VN. Identification and evaluation of anti hepatitis C virus phytochemicals from Eclipta alba. J Ethnopharmacol. 2012;144(3):545-54.
- Naik KS, Gurushanthaiah M, Kavimani M, Prabhu K, Lokanadham S. Hepatoprotective role of Eclipta alba against high fatty diet treated experimental models - a histopathological study. Maedica (Bucur). 2018;13(3):217-22.
- Mishra SK, Sangwan NS, Sangwan RS. Andrographis paniculata (Kalmegh): a review. Pharmacogn Rev. 2007;1(2):283-98.
- Chturvedi GN, Tomar GS, Tiwari SK, Singh KP. Clinical studies on Kalmegh (Andrographis paniculata Nees) in infective hepatitis. Anc Sci Life. 1983;2(4):208-15.
- Singh H, Sharma A. A clinical evaluation of hepatoprotective effects of kalmegh (Andrographis paniculata wall. ex Nees) in patients of acute viral hepatitis. J Res Educ Indian Med. 2013;19(1-2):9-16.