https://doi.org/10.59793/ijcp.v34i4.597
Abstract
The abdomen, often referred to as the "Pandora's Box", presents diagnostic challenges, especially in cases of chronic pain. This report details a perplexing case of chronic, vague abdominal pain accompanied by nonspecific laboratory findings, ultimately leading to the diagnosis of atypical celiac disease through duodenal biopsy and serological investigations. The presented case highlights the significance of a thorough and methodical work-up in managing chronic conditions, emphasizing the importance of considering celiac disease in cases of ambiguous abdominal pain.
Keywords: Gluten sensitivity, celiac disease, malabsorption, abdominal pain, biopsy
Celiac disease (CD) is an immune-mediated intestinal condition triggered by the consumption of gluten, which is found in wheat, rye and barley. It is recognized as one of the most prevalent lifelong food-related disorders worldwide. Formerly referred to as celiac sprue, the term “sprue” derived from Dutch and was used to describe a condition resembling tropical sprue, characterized by symptoms like diarrhea, emaciation, aphthous stomatitis and malabsorption.1,2 While the symptoms and indications of CD have been acknowledged for over a century, it was during the 1940s that Dicke, a Dutch pediatrician, established a connection between the exposure to gluten, a protein component found in wheat and CD.3 It is triggered in genetically predisposed individuals upon consuming gluten, the primary storage protein found in wheat and similar grains. Initially considered a rare malabsorption syndrome affecting children, it is now understood to be a common condition that can be diagnosed at any age and affects multiple organ systems. Diagnosis involves a combination of CD serology and histological examination of small intestinal tissue obtained during a gluten-containing diet. Currently, the only effective treatment for CD is a lifelong strict gluten-free diet.2
Case Details
A 17-year-old male of North-East Indian ethnicity presented to our outpatient clinic with a 1-month history of decreased appetite. The onset of decreased appetite coincided with the initiation of medication for a skin rash, which he had been taking for a month. The patient also reported experiencing generalized abdominal pain for the past 2 years. He denied any significant medical history, including previous abdominal surgeries or gastrointestinal disorders. There was no family history of similar symptoms or significant medical conditions.
On examination, the patient’s height was 177 cm, weight was 71 kg and body mass index (BMI) was 22.66 kg/m2, indicating a healthy weight range. He appeared well-nourished and showed no signs of pallor or edema. His cardiovascular examination revealed a regular rhythm with a heart rate of 56 beats per minute. Blood pressure in the supine position was measured at 110/70 mmHg. Respiratory examination demonstrated normal vesicular breath sounds. Abdominal examination did not reveal any organomegaly or tenderness. His skin examination did not reveal any itchy, bilaterally symmetrical rash, suggestive of dermatitis herpetiformis.
Initial laboratory investigations revealed elevated transaminases and serum globulin levels. However, there was no evidence of anemia. Further details of the investigations have been listed in Table 1. Considering the patient’s symptoms and laboratory findings, further investigations were pursued. The ultrasound scan of the abdomen was normal. An esophagogastroduodenoscopy was performed to further evaluate the nonspecific abdominal symptoms. It revealed scalloping of the duodenal mucosa in the D2 segment, and a biopsy was taken for histopathological examination.
|
Table 1. Investigation Details of the Patient
|
|
Test parameters
|
Patient’s data
|
Normal range
|
|
Complete blood count
|
|
Hemoglobin
Total WBC count
Neutrophils
Lymphocytes
Monocytes
Platelet count
ESR
|
14.3 (g/dL)
7.42 × 103/mm3
56%
36%
7%
150 × 103/mm3
8
|
13.0-16.5 (g/dL)
4-11 × 103/mm3
40-80%
20-40%
02-10%
150-450 × 103/mm3
0-20 mm/hour
|
|
Liver function tests
|
|
Total bilirubin
Direct bilirubin
Indirect bilirubin
SGOT/ AST
SGPT/ALT
ALP
GGTP
Serum albumin
Serum globulin
|
0.7 mg/dL
0.4 mg/dL
0.3 mg/dL
61 U/L
85 U/L
146 U/L
63 U/L
4.4 g/dL
3.6 g/dL
|
0.0-1.3 mg/dL
0.0-0.5 mg/dL
0.0-1.2 mg/dL
<31 U/L
<34 U/L
<98
<140 U/L
3.2-4.6 g/dL
2.0-3.5 g/dL
|
|
Renal function tests
|
|
Urea
Creatinine
|
9 mg/dL
0.9 mg/dL
|
13-43 mg/dL
0.6-1.1 mg/dL
|
|
Special tests
|
|
Anti-tTG antibody (IgG and Ig A)
Random blood sugar
ANA screening
|
IgG: 3.07
IgA: 61.19
96 mg/dL
Negative
|
Negative: <20
Weak positive: 20-30
Moderate to strong positive: >30 U/mL
<99 mg/dL
Negative
|
WBC = White blood cell; ESR = Erythrocyte sedimentation rate; SGOT = Serum glutamic-oxaloacetic transaminase; AST = Aspartate aminotransferase; SGPT = Serum glutamic-pyruvic transaminase; ALT = Alanine aminotransferase; ALP = Alkaline phosphatase; GGTP = Gamma-glutamyl transpeptidase; tTG = Tissue transglutaminase; ANA = Antinuclear antibody.
Microscopic examination of the duodenal biopsy demonstrated fragments of duodenal mucosa exhibiting focal villous blunting. Focal activity was noted, along with a mild increase in intraepithelial lymphocytes. The lamina propria showed expansion with a lymphoplasmacytic infiltrate, occasionally forming aggregates. Evidence of cryptitis was present, but no crypt abscess was observed. These findings were consistent with chronic duodenitis with mild villous blunting and focal increase in intraepithelial lymphocytes, with a modified Marsh Classification type 3a,4 suggestive of CD (Fig. 1). Tissue transglutaminase (tTG) antibodies were positive, indicating the presence of CD.
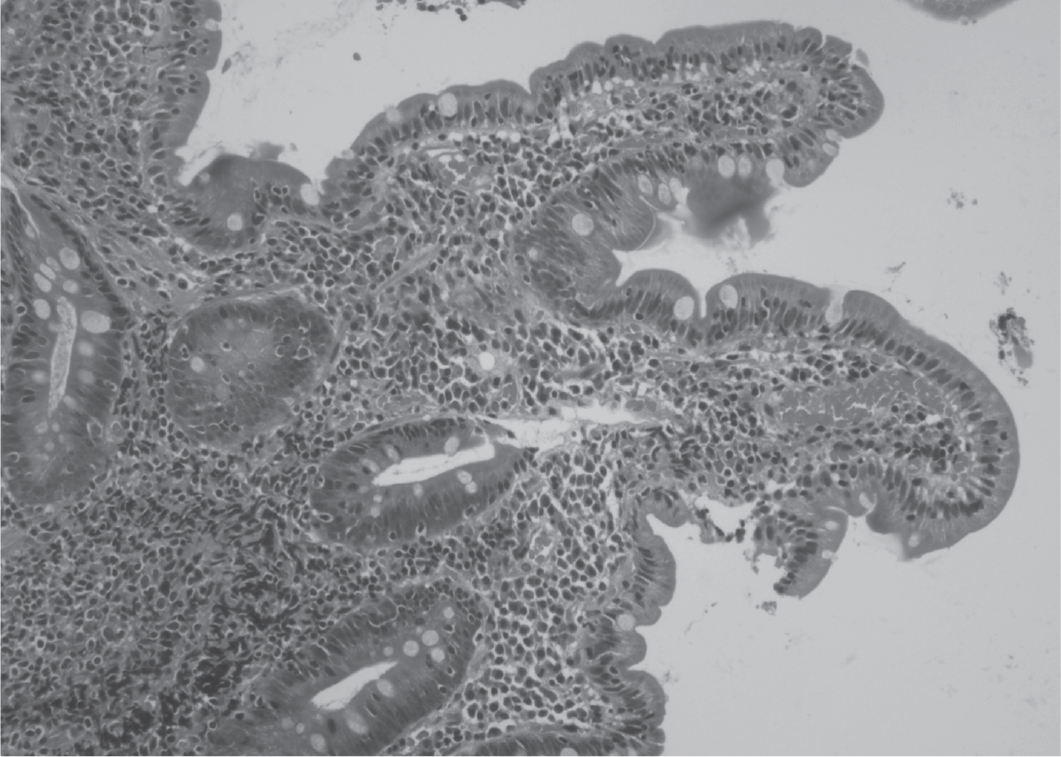
Figure 1. Photomicrograph of 100x magnification showing chronic duodenitis with mild villous blunting and focal increase in intraepithelial lymphocytes.
The patient was started on gluten-free diet (GFD) and showed marked improvement in his clinical features on first follow-up.
DISCUSSION
Abdominal pain is a frequently encountered issue in primary care, with various possible causes that can present challenges for diagnosis and treatment. The nonspecific nature of the symptoms and the lack of clear correlation to specific organ pathology often make it difficult to reach accurate diagnoses. While many cases of abdominal pain are harmless, a considerable portion of patients may have severe conditions that necessitate a careful and thorough management approach to avoid complications and potential fatalities. 5
Celiac disease is a known contributor to persistent abdominal pain, with a worldwide pooled prevalence of biopsy-confirmed cases at 0.7%.1 The age-adjusted prevalence of celiac autoantibodies in different regions of India was found to be 1.23% in the northern region, 0.87% in the northeastern region and 0.10% in the southern region.6
The specific elements associated with CD are human leukocyte antigen (HLA)-DQ2 and HLA-DQ8, an auto-
antigen called tTG and an environmental trigger known as gluten. The pathogenesis involves several steps: partially digested gliadin fragments interact with chemokine receptor 3 on the epithelium’s apical side, leading to the release of zonulin, which then interacts with the intestinal epithelium, causing increased intestinal permeability. The compromised gut barrier allows gliadin peptides to move from the lumen to the lamina propria. Gliadin peptides stimulate the release of interleukin-15, which results in the upregulation of maturation markers and the release of proinflammatory cytokines. Innate immune-mediated apoptosis of intestinal cells leads to the release of intracellular tTG, resulting in the partial deamidation of gliadin peptides. DQ2/8+ antigen presenting cells recognize deamidated gliadin and present it to T helper cells. T helper cells then activate and mature B cells, which produce antibodies (IgM, IgG and IgA) against tTG. These mechanisms collectively initiate enteropathy. Damaged enterocytes enhance the trafficking of gluten from the gut lumen to the lamina propria, ultimately causing crypt hyperplasia and villous blunting due to intestinal epithelial cell death induced by intraepithelial lymphocytes.2,7
In 2011, the Oslo classification of CD established the following clinical presentations: classic, nonclassic, subclinical potential and refractory.8 Additionally, a more practical classification was proposed by Caio et al, categorizing it as intestinal or extraintestinal.7
In classical CD, which accounts for 27% of cases, individuals experience symptoms such as diarrhea, weight loss and malabsorption. Nonclassical disease, making up 52% of cases, is characterized by symptoms like constipation, anemia, hypertransaminasemia, neurologic disorders and dermatitis herpetiformis. Asymptomatic celiac disease, observed in 21% of cases, does not present any noticeable symptoms.7 This case clearly fulfilled the classification of nonclassical CD.
Diagnosing CD can pose difficulties, particularly in nonclassical and subclinical presentations. However, the presence of anemia, unexplained elevation of transaminases and steatorrhea observed in stool examination should raise suspicion and prompt the clinician to consider CD as a potential diagnosis. Individuals suspected of having CD need to follow a gluten-containing diet before undergoing serological and histological testing. For serological testing, the initial step is to perform IgA tTG testing, which has a high sensitivity and specificity (average of 94% and 97%, respectively) for diagnosing CD. If the IgA-tTG test is weakly positive, IgA endomysial antibody (EMA) testing can also be used. If an individual has positive celiac serology, the diagnosis of CD is confirmed through histological examination of duodenal biopsies taken from the duodenal bulb and distal duodenum. Taking at least four biopsies improves the diagnostic rate. Histological features of CD include an increase in intraepithelial lymphocytes, crypt hyperplasia and villous atrophy.9 Additional genetic studies to look for HLA-DQ2/HLA-DQ8 positivity increase the probability of diagnosing the celiac disease.7
After confirming a diagnosis of CD, physicians should strongly advise individuals to follow a strict GFD and ensure they understand the reasons behind it. The GFD involves the complete exclusion of wheat, rye and barley from the diet, preferably with the guidance of a specialized dietitian.9,10 In preliminary studies, ZED1227, an inhibitor of transglutaminase 2, showed promising results in reducing gluten-induced damage to the duodenal mucosa in patients with CD. This suggests that it could potentially be a promising treatment option for CD in the future.11 According to the current guidelines from the National Institute for Health and Care Excellence (NICE), individuals with CD should have annual follow-up appointments.9
Approximately 30% of patients may not respond well to therapy, and persistent or recurring symptoms are often attributed to noncompliance with the dietary regimen. If an individual has been managing the GFD for more than 12 months, and other possible diagnoses have been ruled out, a diagnosis of refractory CD may be considered. In cases where concerning symptoms such as abdominal pain, diarrhea and weight loss continue despite strict adherence to a GFD, it is crucial to investigate and exclude complications associated with CD, such as small intestinal adenocarcinoma, refractory sprue and enteropathy-associated T-cell lymphoma.2,9
CONCLUSION
This case report highlights the challenges involved in diagnosing nonclassical CD, particularly in adolescents presenting with vague abdominal pain. Early recognition and appropriate management of CD are crucial to prevent complications and improve the patient’s quality of life. This case underscores the importance of maintaining a high index of suspicion for CD in patients with atypical presentations, emphasizing the need for further research and awareness to facilitate early diagnosis and intervention.
Declaration by Authors
Ethical Approval: Not applicable.
Consent: Telephonic consent has been obtained from the patient, on the condition of anonymity.
Source of Funding: None.
Conflict of Interest: The authors declare no conflict of interest.
REFERENCES
- Lindfors K, Ciacci C, Kurppa K, Lundin KEA, Makharia GK, Mearin ML, et al. Coeliac disease. Nat Rev Dis Primers. 2019;5(1):3.
- Green PH, Cellier C. Celiac disease. N Engl J Med. 2007;357(17):1731-43.
- van Berge-Henegouwen GP, Mulder CJ. Pioneer in the gluten free diet: Willem-Karel Dicke 1905-1962, over 50 years of gluten free diet. Gut. 1993;34(11):1473-5.
- Yantiss R, Johncilla M. Malabsorption disorders. In: Wang HL, Chen ZE (Eds.). Practical Gastrointestinal Pathology. Springer, Cham; 202. pp. 89-108. Available from: https://link.com/chapter/10.1007/978-3-030-51268-2_5. Accessed July 5, 2023.
- Govender I, Rangiah S, Bongongo T, Mahuma P. A primary care approach to abdominal pain in adults. S Afr Fam Pract (2004). 2021;63(1):e1-e5.
- Ramakrishna BS, Makharia GK, Chetri K, Dutta S, Mathur P, Ahuja V, et al. Prevalence of adult celiac disease in India: regional variations and associations. Am J Gastroenterol. 2016;111(1):115-23.
- Caio G, Volta U, Sapone A, Leffler DA, De Giorgio R, Catassi C, et al. Celiac disease: a comprehensive current review. BMC Med. 2019;17(1):142.
- Ludvigsson JF, Leffler DA, Bai JC, Biagi F, Fasano A, Green PH, et al. The Oslo definitions for coeliac disease and related terms. Gut. 2013;62(1):43-52.
- Rej A, Sanders DS. An update on coeliac disease from the NHS England National Centre for Refractory Coeliac Disease. Clin Med (Lond). 2021;21(2):127-30.
- Husby S, Koletzko S, Korponay-Szabó IR, Mearin ML, Phillips A, Shamir R, et al; ESPGHAN Working Group on Coeliac Disease Diagnosis; ESPGHAN Gastroenterology Committee; European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr. 2012;54(1):136-60.
- Schuppan D, Mäki M, Lundin KEA, Isola J, Friesing-Sosnik T, Taavela J, et al; CEC-3 Trial Group. A randomized trial of a transglutaminase 2 inhibitor for celiac disease. N Engl J Med. 2021;385(1):35-45.