Abstract
Introduction: Talar tuberculosis is very rare presentation of osteoarticular
tuberculosis. Affection of foot is less than 10% in osteoarticular
tuberculosis. Presentation of this disease in peripheral bones is highly
unusual posing diagnostic dilemma and missed diagnosis. Delayed diagnosis may
lead to complications. Case report: A 19-month-old male child presented
with painful swelling over left foot and inability to bear weight. Hemogram was
inconclusive. Radiograph showed lytic lesion in talus with cortical breach in
the superior cortex. Aspiration biopsy was inconclusive and open curettage was
done under anesthesia. Culture reports were negative but histopathological
examination proved tuberculosis. Patient was given antitubercular therapy for 9
months and improved. Conclusion: Tuberculosis can affect any part of
skeleton and high level of suspicion is essential. Culture negative lesion
should be investigated and histopathological examination is essential for any
lytic or infective lesion.
Keywords: Tuberculosis, talus, osteomyelitis, osteolytic
Tubercular talar osteomyelitis is an
uncommon entity in children. Although tuberculosis of foot is well reported but
talar tuberculosis is rare1,2. Involvement of talus due to subacute
hematogenous osteomyelitis in children has been reported by several authors3-8.
But, tubercular involvement of talus in children less than 2 years is rare1,2,9-17.Moreover, the mimicking radiological features with aneurysmal bone cyst,
giant cell tumor and other infections poses diagnostic dilemma18-20.
Here, we report a rare case of tubercular talar osteomyelitis in a 19-month-old
male child who presented with pain and inability to bear weight on left lower
limb.
A 19-month-old baby presented to our OPD with
pain and intermittent low-grade fever for 5 months. Repeated consultation was
done for fever with poor response. Limp was not noticed earlier, since child
started walking at 14 months and repeated falls were taken as normal. The child
had no history of cough, night sweating, loss of weight and appetite. Birth history
and perinatal history were insignificant. Immunization schedule was followed as
per guidelines including BCG vaccination at birth.
On examination, the general condition was fine.
Vitals were stable and child was afebrile. On examination of left foot, it was
swollen, tender and warm below the medial malleolus (Fig. 1). There was a boggy
swelling over the talonavicular area medially. Ankle range of motion was
normal. Foot pronation and supination was restricted. Distal neurovascular
assessment was unremarkable. On investigation, hemoglobin (Hb) - 9.90 g/dL,
total leukocyte count (TLC) - 14,680/mm3, differential leukocyte
count (DLC) showed neutrophils - 30%, lymphocytes = 56%, blood urea - 7.3
mg/dL, serum creatinine - 0.6%, erythrocyte sedimentation rate (ESR) - 55 mm
and C-reactive protein (CRP) titer was raised. Test for viral markers was
unreactive. X-ray showed osteolytic lesion in talus with breach in dorsal
cortex (Fig. 2). On needle aspiration from talus, we found sanguineous fluid.
It was sent for culture sensitivity, which was found sterile. On
histopathological examination, only blood cell was found. The patient was kept
on below knee pop slab, antibiotics and analgesics with no improvement.
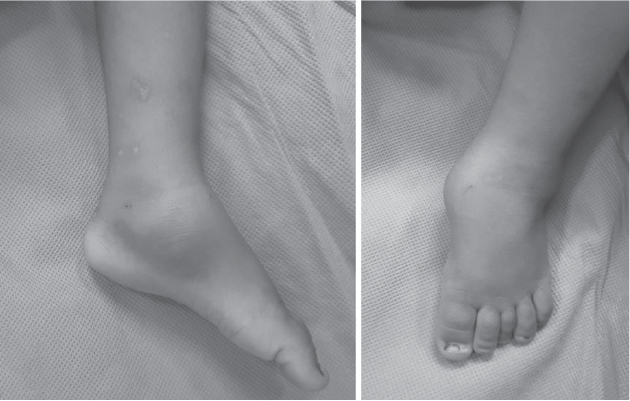
Figure 1. Clinical picture of the patient showing
swelling and signs of inflammation.
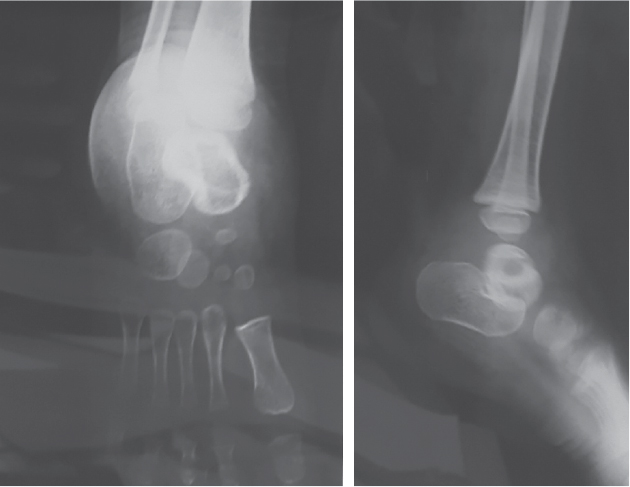
Figure 2. X-ray anteroposterior and lateral view showing
a lytic lesion involving the talus.
After a week, the patient was operated with
differential diagnosis of chronic osteomyelitis of talus and aneurysmal bone
cyst. Through anteromedial incision along tibialis anterior, the talus was
approached. Talonavicular joint was exposed to confirm the talus. The talus was
drilled over the neck proximal to talonavicular joint and it was thoroughly
curetted. Material obtained was sent for Gram staining and Ziehl-Neelsen (ZN) staining, aerobic culture and sensitivity and histopathological examination. Once the normal endosteum was found then the wound was thoroughly lavaged and closure
done. Postoperatively, the patient was kept on intravenous antibiotics and below knee plaster-of-Paris (POP) cast in equinus. ZN staining was positive for acid-fast bacilli. The culture and sensitivity report showed no growth, after 10 days; histopathological
report confirmed tuberculosis. At 12th day, the stitches were removed and antitubercular therapy was started. After consultation with pediatric department. The patient was discharged on below knee POP cast in equinus. Total 9 months of antitubercular
therapy was given with 3 months each for intensive, continuation and maintenance phase. At 1-year follow-up, the patient is doing well with complete healing of the lesion.
Tuberculous involvement of skeleton is 1%-3% of
extrapulmonary tuberculosis1. Involvement of foot among
osteoarticular tuberculosis is less than 10%1. Among all bones of
foot, osteomyelitis of talus is rare1-8. Our search in electronic
and print media revealed cases mostly about subacute hematogenous osteomyelitis
of talus and foot bones3-8. Dhillon1 and Mittal2 have
reported exclusively on tuberculosis of foot bones. Only one case of talar
osteomyelitis out of 24 cases was reported by Dhillon et al1 and none
out of 44 cases in a series by Mittal et al2. Isolated case reports
on talus have been reported in recent years and are given chronologically in
Table 19-17.Our patient is the youngest case to be reported.
|
Table 1. Isolated Cases of TB Talus as Reported in Chronological Order
|
|
Author
|
Year of reporting
|
Age of patient (years)
|
|
Anand et al9
|
2002
|
8
|
|
Teklali et al10
|
2003
|
20 months
|
|
Ebrahimzadeh et al11
|
2006
|
7
|
|
Mardanpour et al12
|
2010
|
52
|
|
Arora et al13
|
2014
|
45
|
|
Dahuja et al14
|
2014
|
14
|
|
Mohammad et al15
|
2015
|
42
|
|
Sekhon et al16
|
2015
|
14
|
|
Khan et al17
|
1999
|
5
|
The diagnosis is usually difficult since
presentation is vague and nonspecific4,7. Flexion at hip and knee
with limb in external rotation may be present4. Swelling and redness
in the foot may be delayed feature4. Location of swelling is
variable. Verbeek4 reported swelling on the lateral aspect while
Ganaisan5 reported swelling over the ankle area. We noticed swelling
on the medial aspect of mid-foot. Constitutional symptoms are usually nil1,2,6,7.
In our case, low-grade fever and inability to bear weight for 5 months was the
only complaint.
Delayed diagnosis is usually due to lack of
constitutional symptoms, poor localizing signs, low level of suspicion and
simulating radiological features1-8,18,19. Symptoms to admission was
5 months average (Dhillon1), 1 month (Ganaisan5), 2-12
weeks (Ezra6), 5 days to 4 weeks (Grattan-Smith7) and 1-5
months (Skevis8). In our case it was 5 months.
Hemogram shows signs of infection with raised ESR1,4-8,but CRP is rarely raised4,6. In our case, the ESR and CRP were
raised along with lymphocytosis.
Conventional radiography is the primary tool for
diagnosis. Phemister's triad of periarticular osteoporosis, marginal erosions
and narrowing of joint space is usually seen in osteoarticular tuberculosis.
But, this feature is not evident in foot bones always2.Mittal
et al2 observed five patterns of foot bone lesions in tuberculosis:
cystic, rheumatoid, subperiosteal, kissing, and spina ventosa. In our case, it
was cystic type of lesion, which has central osteolytic lesion with no
sequestrum and no periosteal reaction.
The lesion in foot usually mimics other
conditions as well18-20. Aneurysmal bone cyst, giant cell tumors,
and infections of foot bones mimics cystic type of tuberculosis. Shirazi et al20
noted aneurysmal bone cyst was as common as giant cell tumor of small
bones of hand and feet. Infections and inflammatory simple cysts were equally
prevalent in less than 10 years old children20. We found blood on
aspiration from talus in our case. Hence, our differential diagnosis was
aneurysmal bone cyst and chronic osteomyelitis.
Computed tomography (CT) scan, magnetic
resonance scan and bone scan of foot is usually required to localize the lesion
and to see the soft tissue condition1,2,4-7,18. Magnetic resonance
imaging (MRI) shows changes consistent with chronic osteomyelitis5.
Bone scan with gallium-67 (Ga-67) and technetium-99 (Tc-99) shows increased
tracer uptake in the tarsal bones6,7. On getting negative report on
aspiration cytology and culture sensitivity we directly opted for curettage
exploration of the lesion as suggested by Dhillon et al1. The same
has been done by other authors1,7,18,20. The culture report is
usually negative since osteoarticular tuberculosis is a paucibacillary
condition1,2,7. Open curettage and biopsy is usually required for
diagnosis1,2,7,8,18-20.
Minimal pus and granulation tissue is found with
evidence of necrotic bone and polymorphonuclear infiltrate7. As a
rule, we send sample for culture in every case of suspected tumor and we do
histopathological examination of every abscess.
Following the rule, we found granuloma in our
case. Culture negative and lack of conclusive diagnosis can be curtailed by
early biopsy. Delay in diagnosis may lead to complete destruction of bone and
joints. Hence, early diagnosis is the priority.
Age is no bar for
osteoarticular tuberculosis. Any osteolytic lesion in talus or foot bones
should be investigated and high level of suspicion is essential to rule out tuberculosis.
All the abscesses should be biopsied to prevent missed diagnosis. Early
diagnosis and complete treatment of tuberculosis should be the aim.
1.
Dhillon MS, Aggarwal S, Prabhakar S, Bachhal V. Tuberculosis of the foot: an osteolytic variety. Indian J Orthop. 2012;46(2):206-11.
2.
Mittal R, Gupta V, Rastogi S. Tuberculosis of the foot. J Bone Joint Surg Br. 1999;81(6):997-1000.
3.
Pabla R, Tibrewal S, Ramachandran M, Barry M. Primary subacute osteomyelitis of the talus in children: a case series and review. Acta Orthop Belg. 2011;77(3):294-8.
4.
Verbeek PA, de Roest JG, van Raaij TM. Osteomyelitis of the talus in a limping child. Ned Tijdschr Geneeskd. 2012;155(35):A5103.
5.
Ganaisan P, Singh A, Kumar C, Manikam R. A limping child: on a first look it is hip or knee in origin! No! It’s a Talus osteomyelitis. Int J Orthop Surg. 2013;20(1).
6.
Ezra E, Wientroub S. Primary subacute haematogenous osteomyelitis of the tarsal bones in children. J Bone Joint Surg Br. 1997;79(6):983-6.
7.
Grattan-Smith JD, Wagner ML, Barnes DA. Osteomyelitis of the talus: an unusual cause of limping in childhood. AJR Am J Roentgenol. 1991;156(4):785-9.
8.
Skevis XA. Primary subacute osteomyelitis of the talus. J Bone Joint Surg Br. 1984;66(1):101-3.
9.
Anand A, Sood LK. Isolated tuberculosis of talus without ankle and subtalar joint involvement. Med J Malaysia. 2002;57(3):371-3.
10.
Teklali Y, El Alami ZF, El Madhi T, Gourinda H, Miri A. Tuberculosis of the talus in the child. Eur J Orthop Surg Traumatol. 2003;13(1):52-4.
11.
Ebrahimzadeh MH, Sadri E. Isolated tuberculosis of the talus bone. Arch Iran Med. 2006;9(2):159-60.
12.
Mardanpour KK, Rahbar M. Isolated tuberculosis of the talus bone. Casp J Intern Med. 2010;1(2):75-7.
13.
Arora K, Choudhry P. Tuberculosis of talus bone in middle aged man - A case report. Int J Orthop Trauma Nursing. 2014;18(1):35-8.
14.
Dahuja A, Dahuja G, Kaur R, Bansal K. Isolated tuberculosis of talus: a case report. Malays Orthop J. 2014;8(1):61-2.
15.
Mohammad F, Singh S, Lal AK, Kumar S. Tuberculosis of talus: a case report. Indian J Orthop Surg. 2015;2(2):46-7.
16.
Sekhon G, Gill SS, Singh M, Gautam R. Tuberculosis of the talus bone. Int J Develop Res. 2015;11:5993-5.
17.
Khan FA, Khoshhal K, Saadeddin M. Tuberculosis of talus and cuboid - a report of 2 children. Acta Orthop Scand. 1999;70(6):627-39.
18.
Sharma S, Gupta P, Sharma S, Singh M, Singh D. Primary aneurysmal bone cyst of talus. J Res Med Sci. 2012;17(12):1192-4.
19.
Luna AR, Fahandez-Saddi H, Garcia AV, Reina Cde J, Martin JV. Aneurysmal bone cyst in children involving
infrequent locations. Report on two cases. Chir Organi Mov. 2004;89(4):347-52.
20.
Shirazi N, Gupta V, Kapoor I, Harsh M, Chauhan N, Ahmad S. Osteolytic lesions of hand and feet: a seven-year experience from a tertiary referral centre of North India. Malays J Pathol. 2014;36(2):115-24.