Introduction: Negative
pressure wound therapy (NPWT) is a procedure in which vacuum is used to enhance
wound healing. Vacuum-assisted closure (VAC) refers to wound dressing that uses
pressure below normal continuously or intermittently to the surface of a wound.
The negative pressure is maintained by an apparatus; this promotes healing in
various kinds of wounds and also helps in wound debridement. Aims: This
study was carried out with an aim to find out the rate of wound contraction,
compare infection clearance, granulation tissue formation and to study
postoperative pain after using NPWT. Material and methods: All types of
infected wounds with slough were selected. Patients irrespective of sex between
18 and 70 years of age were included. The wounds included were traumatic, diabetic
foot, varicose ulcer, infected wounds, carbuncle, etc. The procedure included
surgical debridement as a preliminary procedure, followed by application of
NPWT. The wound criteria: 1) size, 2) shape, 3) wound margin, and floor,
4) edge and contraction were studied. Results and Discussion: Infected
wounds can be treated by specific modalities like daily wound dressing,
surgical debridement, hyperbaric oxygen therapy and NPWT. NPWT seems more
efficient than standard wound care for infected wounds. In our study, the mean
size of ulcer in diabetic patients before NPWT was found to be 6.33 × 4.52 cm;
after application of NPWT, it was 4.7 × 2.95 cm. The mean size of ulcer in
traumatic patients before NPWT was found to be 7.1 × 5.1 cm, while after
application of NPWT, it was 5 × 3.63 cm. The mean size of ulcer in
vascular patients was found to be 5.71 × 3.85 cm before NPWT, and after
application of NPWT, it was 4 × 2.42 cm. NPWT dressings have been proven
to be beneficial as a variant method of dressing, mainly by negative pressure
which sucks out serous fluid and helps in the formation of granulation tissue.
Used in various wounds, continuous suction over period of time and later
intermittent suction depending on wound status enhance wound healing process and
lead to faster recovery compared to conventional methods of dressing. Conclusion:
The wound healing period for large traumatic wounds and chronic diabetic wounds
is 123 days as per published data. In our study, where NPWT was used, the
average wound healing period was 35 days, ranging between 10 and 62, which is
statistically significant (p < 0.005). NPWT is cost-effective, reduces
hospital stay of patient with minimal chances of limb amputation with better
results than standard wound care.
Keywords: Vacuum-assisted closure, hyperbaric oxygen
therapy, negative pressure wound therapy
Negative pressure wound therapy (NPWT) is a
relatively novel method used for managing wounds, both acute and chronic1.
Vacuum-assisted closure (VAC), or NPWT, uses vacuum to improve wound healing.
VAC involves wound dressing that applies pressure below normal, continuously or
intermittently, to the surface of a wound2-4. The negative pressure
promotes healing in different types of wounds5-7. It also assists
with wound debridement. Wound healing is best at negative pressure of 85-125
mmHg. Application of negative pressure removes fluid, decreases edema and
enhances blood flow, and decreases bacterial counts. It is less costly than
traditional management of infected wounds8-13.
A negative pressure of 50-125
mmHg lowers the interstitial pressure, and
fluid and debris from the wound gets sucked into a collection chamber14-18.
In the beginning, the vacuum is continuous, but as the drainage is
reduced, the vacuum is applied intermittently. The vacuum dressing is usually
changed at approximately 2- to 6-day interval.19-25
This study was carried out with an aim to find out
rate of wound contraction, compare infection clearance, granulation tissue
formation and to study postoperative pain after using NPWT. The study also
aimed to determine the length of hospital stay and to evaluate the
cost-effectiveness of the procedure and effect on amputation prevention.
Source of Data
·
Patients of Grant Government Medical College and JJ
Hospital, Mumbai, Maharashtra.
·
A total of 50 cases clinically
presenting as ulcer between
June 2017 and December 2019 were included
in the study.
Inclusion Criteria
·
Both male and female.
·
Patients between 18 years and 70 years.
·
Patients who signed informed written valid consent
to be included in the study.
·
Patients having acute or chronic wounds, including
traumatic wounds, varicose ulcer, bed sore, diabetic wounds.
Exclusion Criteria
·
Age less than 18 or above 70 years.
·
Patients on chemotherapy or suffering from malignancy.
·
Suspected poor compliance.
·
If the patient did not sign the consent form.
·
Peripheral vascular disease
wound with acute or chronic osteomyelitis.
Procedure
Preparation of the wound
After cleaning the wound, foam
dressing was
cut to shape and kept into the wound cavity. The wound was then sealed with an
adhesive dressing ensuring that the drapes covered the foam and tubing and 3 cm
of healthy skin.
Negative pressure application
Negative pressure was applied to the wound using vacuum pump (Fig. 1), which delivered
continuous orintermittent pressures, ranging from 50 to 125 mmHg. The foam dressing squeezed to the negative pressure. The pressure
was applied continuously for the first 48 hours and then changed.


Figure 1. VAC instrument.
The study was done on 50 patients in Dept. of
Surgery, JJ Hospital, Mumbai, Maharashtra.
In our study, as shown in Table 1, the mean size of
ulcer in diabetic patients before VAC was found to be 6.33 × 4.52 cm,
while after the application of VAC, it appeared to be 4.75 × 2.9 cm; the mean
size of ulcer in traumatic patients before VAC was found to be
7.1 × 5.1 cm, and after application of VAC, it appeared to be 5
× 3.63 cm; the mean size of ulcer in vascular patients was found to be 5.71 ×
3.85 cm before VAC, and after application of VAC, it appeared to be 4 × 2.42
cm. The p value was <0.05 and it was statistically significant. Figure 2
shows diabetic foot infection and Figure 3 depicts necrotizing fasciitis before
and after treatment.
|
Table 1. Mean Ulcer Size (cm) Before and After VAC
Therapy in Wounds of Different Etiology
|
|
Etiology
|
Before VAC
|
After VAC
|
|
Length (cm)
|
Breadth (cm)
|
Length (cm)
|
Breadth (cm)
|
|
Diabetic
|
6.33
|
4.52
|
4.75
|
2.95
|
|
Traumatic
|
7.1
|
5.1
|
5
|
3.63
|
|
Vascular
|
5.71
|
3.85
|
4
|
2.42
|
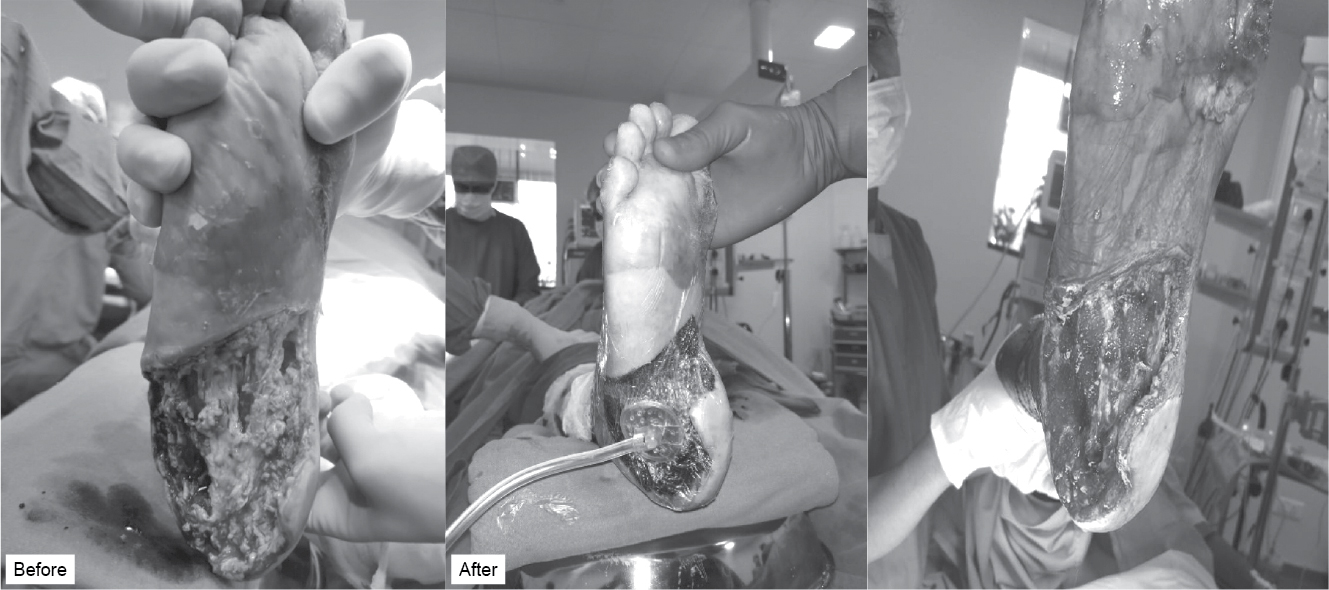
Figure 2. Diabetic foot infection (biofilm).
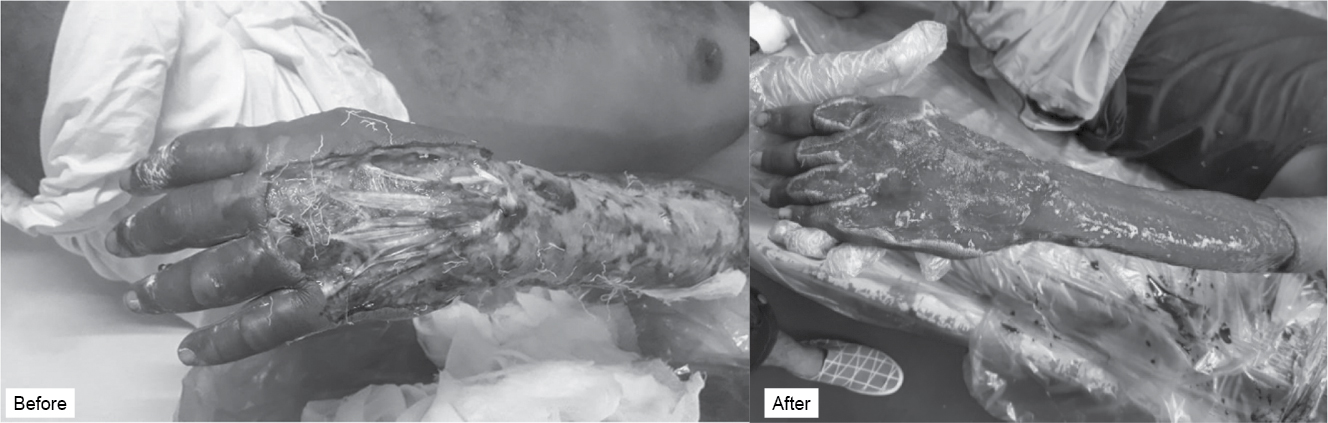
Figure 3. Necrotizing fasciitis.
VAC therapy is an alternative to routine
wound management.
In our study, average age of wounds was 35 days. In
a study by Caniano et al26, average age of wounds was 37 days and in
that by Ulusal et al27, it was 32 days, as compared to 59 days
with standard dressing. In our study, the mean duration of wound healing
was found to be 35.2 days with standard deviation (SD) of 12.03 days. In a
study by Zimny et al28, the mean duration of wound healing was found
to be 123.4 days with SD of 10.5 days. On statistical analysis, the p value was
calculated to be <0.00001, which is statistically significant with 95%
confidence interval (CI).
The wound healing period for large traumatic wounds
and chronic diabetic wounds is 123 days as per published data28. In
our study, where NPWT was used, the average wound healing period was 35 days.
Many mechanisms are suggested. VAC works by
increasing the local blood flow and diminishes the edema fluid and colonization
rates. The procedure promotes wound closure as it accelerates the formation of
granulation tissue and also via mechanical effects on the wound29.
It provides a clean moist wound and removes excess wound fluid, thus giving way
to an ideal wound healing environment.
In our study, out of 50 patients, 36 patients had
wound over foot region, 12 patients had wound over back region.
NPWT provides a moist wound environment, favoring
granulation of edge of ulcer. A moist wound bed promotes re-epithelialization,
action of growth factors, angiogenesis.
A moist wound environment also limits local pain,
protecting the nerve endings and enhancing quality of life. Decrease in edema
limits interstitial pressure and has a positive impact on microvascular
occlusion and lymphatic drainage, thus enhancing the availability of nutrients,
oxygen and antibiotics in the wound area30.
From our study, it can be concluded that NPWT is
useful in wound healing in various types of wounds; therefore, NPWT should be
the modality of choice in management of infected wounds. Vacuum-assisted
dressing is more effective than traditional wound dressing. NPWT, in
combination with surgical debridement and antibiotic therapy, is effective in managing
infected wounds.
The wound healing period for large
traumatic wounds and chronic diabetic wounds is 123 days as per published
data. In our study, where NPWT was used, the average wound healing period was
35 days, ranging between 10 and 62 days, which was statistically significant
(p < 0.005).
NPWT is cost-effective, reduces hospital stay of
patient with minimal chances of limb amputation with better results than
standard wound care.
1.
Eginton MT, Brown KR, Seabrook
GR, Towne JB, Cambria RA. A prospective randomized evaluation of
negative-pressure wound dressings for diabetic foot wounds. Ann Vasc Surg.
2003;17(6):645-9.
2.
Banwell PE, Teot L. Topical
negative pressure (TNP): the evolution of a novel wound therapy. J Wound Care.
2003;12(1):22-8.
3.
Morykwas MJ, Argenta LC,
Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice.
First South-East Asia Edition, Philadelphia, PA: Elsevier Saunders; 2012. p.
152.
4.
Sabiston DC, Townsend CM.
Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice.
First South-East Asia Edition, Philadelphia, PA: Elsevier Saunders; 2012. p.
152.
5.
Schwartz SI, Brunicardi FC.
Schwartz's Principles of Surgery: Self-assessment and Board Review. 10th
Edition, New York, London; 2009. p. 266.
6.
Greenfield LJ, Mulholland MW.
Surgery: Scientific Principles and Practice. 6th Edition, Philadelphia:
Lippincott Williams & Wilkins; 2001. p. 3211.
7.
Armstrong DG, Lavery LA;
Diabetic Foot Study Consortium. Negative pressure wound therapy after partial
diabetic foot amputation: a multicentre, randomised controlled trial. Lancet.
2005;366(9498):1704-10.
8.
Lazarus GS, Cooper DM,
Knighton DR, Percoraro RE, Rodeheaver G, Robson MC. Definitions and guidelines
for assessment of wounds and evaluation of healing. Wound Repair Regen.
1994;2(3):165-70.
9.
Joseph E. A prospective
randomized trial of vacuum-assisted closure versus standard therapy of chronic
nonhealing wounds. Wounds. 2000;12:60-7.
10.
Lionelli GT, Lawrence WT.
Wound dressings. Surg Clin North Am. 2003;83(3):617-38.
11.
Bowler PG. Wound
pathophysiology, infection and therapeutic options. Ann Med. 2002;34(6):419-27.
12.
Williams NS, Bullstrode CJK,
O'Connell PR. Bailey & Love's Short Practice of Surgery, 25th Edition, Ann
R Coll Surg Engl. 2010;92(2):178.
13.
Webb LX, Schmidt U. Wound
management with vacuum therapy. Unfallchirurg. 2001;104(10):918-26.
14.
Lee HJ, Kim JW, Oh CW, Min WK,
Shon OJ, Oh JK, et al. Negative pressure wound therapy for soft tissue injuries
around the foot and ankle. J Orthop Surg Res. 2009;4:14.
15.
Gammal S, Popp R, Schäfer M,
el Gammal C, Altmeyer P. A color CD-ROM image analysis system to quantify
débridement and healing of ulcers. In: Abatangelo G, Donati L, Vanscheidt W
(Eds.). Proteolysis in Wound Repair. Berlin, Heidelberg: Springer Berlin Heidelberg;
1996. pp. 71-86.
16.
Cohen IK. An overview of wound
healing biology. In: Ziegler TR, Pierce GF, Herndon DN (Eds.). Growth Factors
and Wound Healing: Basic Science and Potential Clinical Applications. New York,
NY: Springer; 1997. pp. 3-7.
17.
Jones SM, Banwell PE,
Shakespeare PG. Advances in wound healing: topical negative pressure therapy.
Postgrad Med J. 2005;81(956):353-7.
18.
Erba P, Ogawa R, Ackermann M,
Adini A, Miele LF, Dastouri P, et al. Angiogenesis in wounds treated by
microdeformational wound therapy. Ann Surg. 2011;253(2):402-9.
19.
Sato K, Yokota T, Ichioka S,
Shibata M, Takeda S. Vasodilation of intramuscular arterioles under shear
stress in dystrophin-deficient skeletal muscle is impaired through decreased
nNOS expression. Acta Myol. 2008;27(1):30-6.
20.
Roseborough IE, Grevious MA,
Lee RC. Prevention and treatment of excessive dermal scarring. J Natl Med
Assoc. 2004;96(1):108-16.
21.
Ryan TJ. Cellular responses to
tissue distortion. In: Bader DL (Ed.). Pressure Sores - Clinical Practice and
Scientific Approach. London: Macmillan Education UK; 1990. pp. 141-52.
22.
Hasegawa S, Sato S, Saito S,
Suzuki Y, Brunette DM. Mechanical stretching increases the number of cultured
bone cells synthesizing DNA and alters their pattern of protein synthesis.
Calcif Tissue Int. 1985;37(4):431-6.
23.
Fabian TS, Kaufman HJ, Lett
ED, Thomas JB, Rawl DK, Lewis PL, et al. The evaluation of subatmospheric
pressure and hyperbaric oxygen in ischemic full-thickness wound healing. Am
Surg. 2000;66(12):1136-43.
24.
Joseph E, Hamori CA, Sb B,
Roaf E, Swann N, Anastasi G. A prospective randomized trial of
vacuum-assisted closure versus standard therapy of chronic nonhealing wounds.
Wounds. 2000;12:60-7.
25.
Mullner T, Mrkonjic L, Kwasny
O, Vecsei V. The use of negative pressure to promote the healing of tissue
defects: a clinical trial using the vacuum sealing technique. Br J Plast Surg.
1997;50(3):194-9.
26.
Caniano DA, Ruth B, Teich S.
Wound management with vacuum-assisted closure: experience in 51 pediatric
patients. J Pediatr Surg. 2005;40(1):128-32.
27.
Ulusal AE, Sahin MS, Ulusal B,
Cakmak G, Tuncay C. Negative pressure wound therapy in patients with diabetic
foot. Acta Orthop Traumatol Turc. 2011;45(4):254-60.
28.
Zimny S, Schatz H, Pfohl M.
Determinants and estimation of healing times in diabetic foot ulcers. J Diabetes
Complications. 2002;16(5):327-32.
29.
Mendez-Eastman S. Guidelines
for using negative pressure wound therapy. Adv Skin Wound Care. 2001;14(6):314-22.
30.
Meloni M, Izzo V, Vainieri E,
Giurato L, Ruotolo V, Uccioli L. Management of negative pressure wound therapy
in the treatment of diabetic foot ulcers. World J Orthop. 2015;6(4):387-93.