Abstract
Self-monitoring of blood glucose (SMBG)
using blood glucose monitoring (BGM) devices is recommended for people with
diabetes to improve glycemic control and to detect and prevent episodes of
hypoglycemia in these patients. The International Organization for
Standardization (ISO) and World Health Organization (WHO) have defined specific
criteria for accuracy, precision, user evaluation, and interfering agents for
the quality of these devices. In this targeted literature review, Accu-Chek®
devices (Instant®, Guide®, Active®) were found
to have stable results with appropriate accuracy and precision and did not
respond to interfering agents. The devices were also found to be cost-effective
and ranked high on patient preference.
Keywords: SMBG, Accu-Chek, Diabetes
*Treasurer, International Society of
Endocrinology (ISE); Vice President, South Asian Obesity Forum (SOF); Bharti
Hospital, Karnal, Haryana, India
Diabetes
mellitus (DM) is one of the major public health
concerns and one of the top noncommunicable diseases. According to
the 2021 International Diabetes Federation (IDF) Atlas, approximately 537
million adults are living with diabetes worldwide, with a rising prevalence in
low-middle-income countries compared to high-income countries1.
According to the Indian Council of Medical Research-India study published in
2023, the overall weighted prevalence of DM in India was reported to be 11.4%,
while the prevalence of prediabetes was reported to be 15.3%2. The
Indian Council of Medical Research (ICMR) guidelines for diabetes management
recommend self-monitoring of blood glucose (SMBG) to improve glycemic
control3.
SMBG using blood glucose monitoring (BGM)
devices is widely utilized worldwide to improve outcomes in DM. SMBG helps
optimize treatment in both insulin-dependent and non-insulin-dependent
patients. SMBG can also be used to identify hypoglycemic episodes and can help
personalize therapy4. Further, studies have shown that frequent
self-monitoring is associated with increased quality-adjusted life expectancy
due to improvement in glycated hemoglobin (HbA1c) levels compared with no SMBG5,6.
According to the World Health Organization
(WHO) HEARTS D study, SMBG can be used to diagnose diabetes, albeit with a
higher cut-off of 220 for post-load glucose7.
SMBG is usually conducted with a capillary blood
sample collected from a fingertip prick. However, for BGM, samples from
alternate sites such as the earlobe, heel, forearm, and palm can also be
utilized. BGM can also be done using venous blood, plasma, and serum. As
glucose equilibrates in the aqueous portion of the sample, samples such as
plasma are preferable as they have lower concentration of other blood
components such as cells8.
Glucose meters comprise of two components
including a dehydrated enzyme on the test strip and a detector. Glucose from
the blood sample rehydrates the enzyme and carries out a reaction, which can be
detected by the detector. Current glucometers use one of the three principle
enzymatic reactions namely glucose oxidase, glucose dehydrogenase, and
hexokinase8.
Fortwaengler et al
showed that inaccurate BGM is associated with additional costs when
International Organization for Standardization (ISO)
standards are not met9. More recently, continuous glucose monitoring
(CGM) has been employed to help attain better glycemic control. CGM devices may
improve diabetes outcomes when used in adjunct to SMBG10.
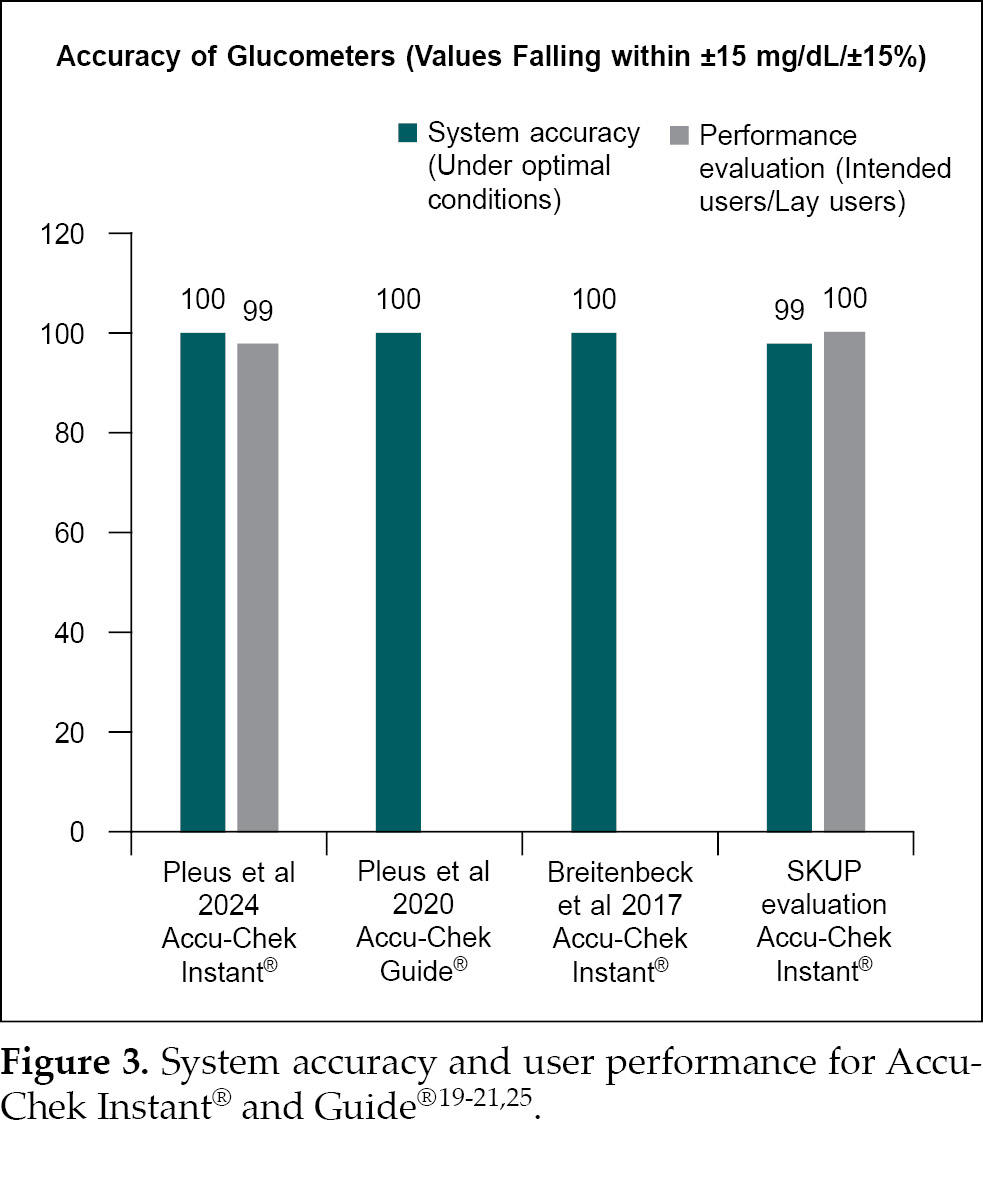
For an SMBG device
to be considered of appropriate standards, compliance with ISO 15197:2013 is
the minimum requirement by the regulatory authorities. According to these
recommendations, to establish system accuracy of SMBG device, =95% of the
individual glucose measured values shall fall within ±15 mg/dL of the reference
results at glucose concentrations <100 mg/dL or
within ±15% at glucose concentrations =100 mg/dL for both the technician and
the patient, and =99% of individual glucose measured values shall fall within
Zones A and B of the Consensus Error Grid (CEG) for diabetes. Precision is
defined as standard deviation (SD), which requires to be =3 mg/dL at glucose
concentrations <100 mg/dL and the coefficient of variation (CV) shall be
=3.0% at glucose concentrations =100 mg/dL. Impact of hematocrit is defined as
mean bias (to reference glucose) that does not exceed ±10 mg/dL to the nominal
hematocrit sample (42%) mean bias (to reference glucose) at glucose
concentrations <100 mg/dL. Mean bias (to reference glucose) that does not
exceed ±10% to the nominal hematocrit sample (42%) mean bias (to reference
glucose) at glucose concentrations =100 mg/dL11.
The WHO recommendations for intermediate
precision state the criteria for repeatability (within-run variability) % CV shall be <5.0%. Usually in many countries,
the % CV acceptance criterion is =7.1%. However, systems with higher precision (lower values of % CV) depict a product with robust quality. WHO recommends the trueness of measure to be <15% (better 10%), which means the percentage
of inaccuracies obtained should be <10%12.
Glucose dehydrogenase-glucose oxidase (GDH-GOD)
based glucometers are prone to oxygen interference because oxygen is a
physiological electron acceptor and is naturally affected by both low and high
oxygen levels. In contrast, GDH is not affected by oxygen levels because oxygen
is not involved in its electrochemical reaction13. WHO further
recommends that all strips should have at least 12 months validity from the
date of production12. Strips providing higher stability than 18
months are considered to be an added advantage for health care setups for
cost-effective management of patients with DM. There are certain interfering
agents that may confound the reading by the device, such as high hematocrit,
elevated triglyceride levels, and certain drugs and environmental factors. The
presence of interfering agents results in inaccurate reading by the device14,15.
WHO has also defined criteria for the time to
result. The guidelines recommend that in
the case of self-monitoring/single-patient device, results should be
available in less than 30 seconds (preferably <10 seconds). The devices
that provide instant results in less than 5 seconds are of higher clinical
significance in the decision-making process, especially in emergency cases12.
Considering the importance of SMBG in management of DM and the standards of BGM devices, we conducted this literature review to understand how the effectiveness of a BGM device is measured in terms of achieving target glucose levels, accuracy,
precision, economic analysis, and performance from published literature, especially from the point of view of those Accu-Chek® devices which are available in India (Accu-Chek® Instant®, Instant S®, Guide®,
and Active®). We also sought to understand the patient and provider preferences for using these BGM devices.
METHODS
A comprehensive
literature search was conducted on the PubMed databases utilizing different
SMBG glucometer specific search terms. Additional searches were conducted in
Google Scholar and from other review article reference lists through
cross-referenced articles. The search was not limited by time, and all applicable literature was screened. Only studies conducted in
human populations and published in English language were considered.
All the retrieved articles were screened for
population, objectives, and use of SMBG devices. Studies reporting accuracy,
precision, patient/provider preference, and economic analysis of Accu-Chek®
devices (Instant®, Guide®, Active®) and only
those articles with full text available in English were included.
RESULTS OF LITERATURE SEARCH
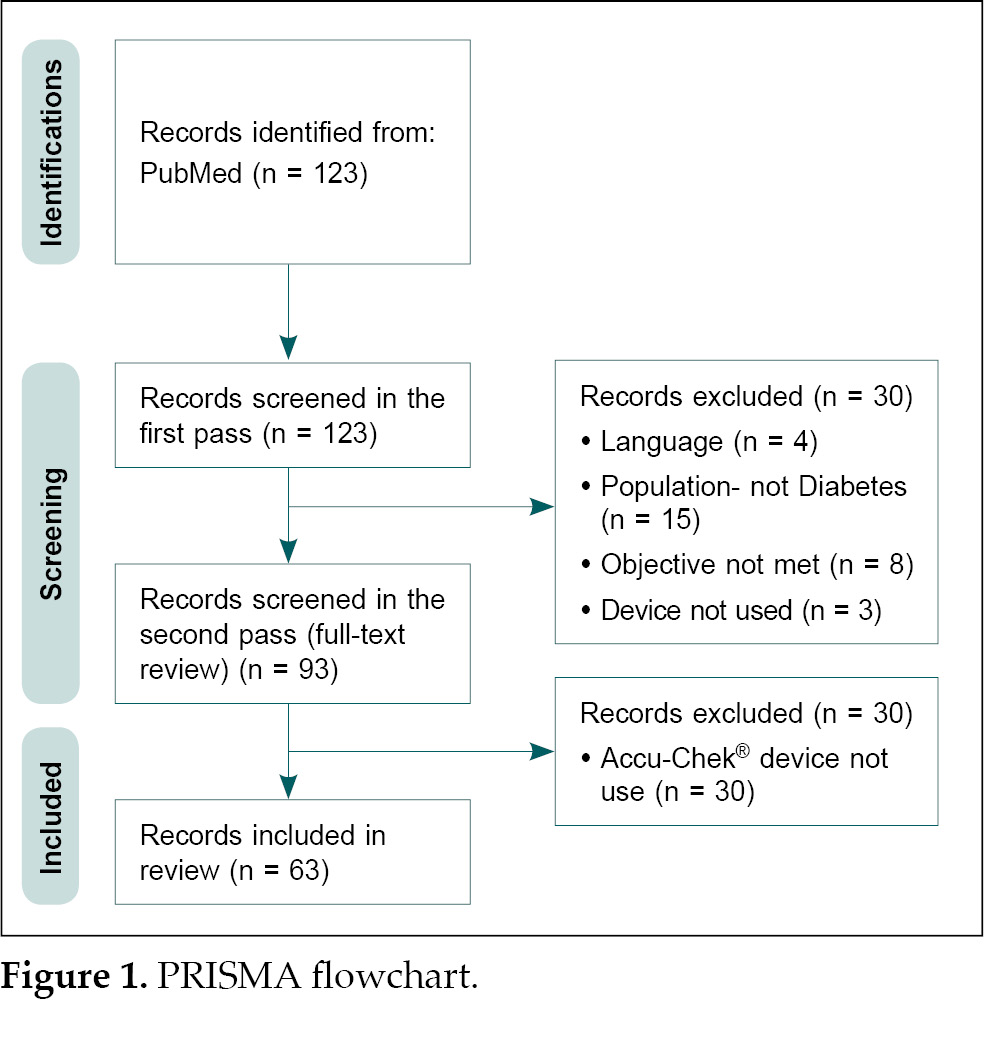
A total of 123 studies were identified. After
screening for language and objectives, 93 studies were included in the
full-text screening and were further screened and assessed for the core
objectives of the study: accuracy, precision, patient/provider preference, and
economic analysis of the devices considered. A total of 63 studies were
included in the review (Fig. 1).
ACCURACY AND PRECISION
According to the ISO standard 15197:2013, system
accuracy assessment defines accuracy requirements for BGMs (ISO 15197, clause
6.3), which is calculated by performance under laboratory conditions, and user
performance evaluation (ISO 15197, clause 8), which is the performance of the
device under real-world scenarios11. According to the standards, 95%
of the individual glucose results shall fall within ±15 mg/dL of the
manufacturer’s measurement procedure at glucose concentrations <100 mg/dL
and within ±15% of glucose concentrations =100 mg/dL. For clinical accuracy,
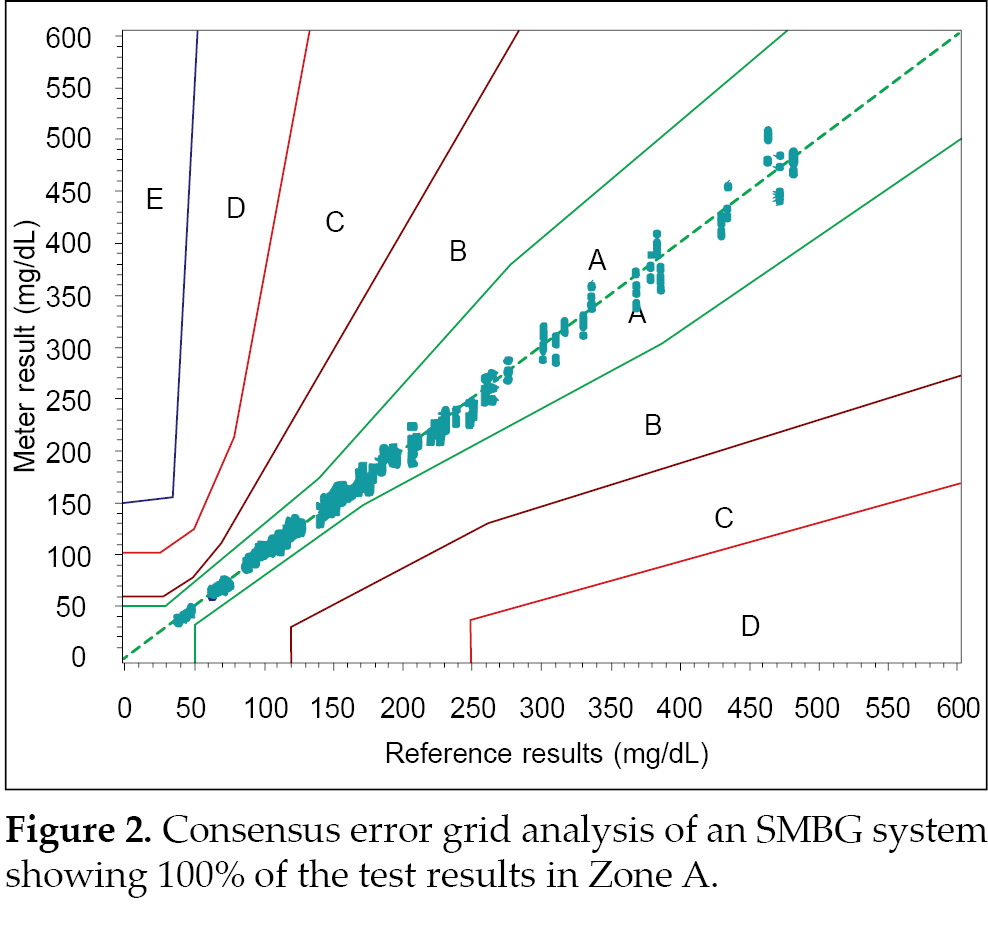
99% of results should fall within Zone A + B of the CEG for type 1 diabetes.
Figure 2 depicts a CEG with 100% of the test results falling within
Zone A. The standard defines precision as % CV <5.0% (Table 1)11.
In the system evaluation report for Accu-Chek® Active®,
Guide®, and Instant®, which operate by the GHD-GOD
mechanism, all 3 meters met all ISO requirements for accuracy and precision for
multiple tested lots.
The results show that the systems had 99%-100% of the data within the bias
requirements, and 99%-100% of the results fell within Zone A of the CEG,
clearly exceeding the acceptance criteria. The three SMBG devices were found to
meet accuracy requirements in neonates and pregnant women16-18.
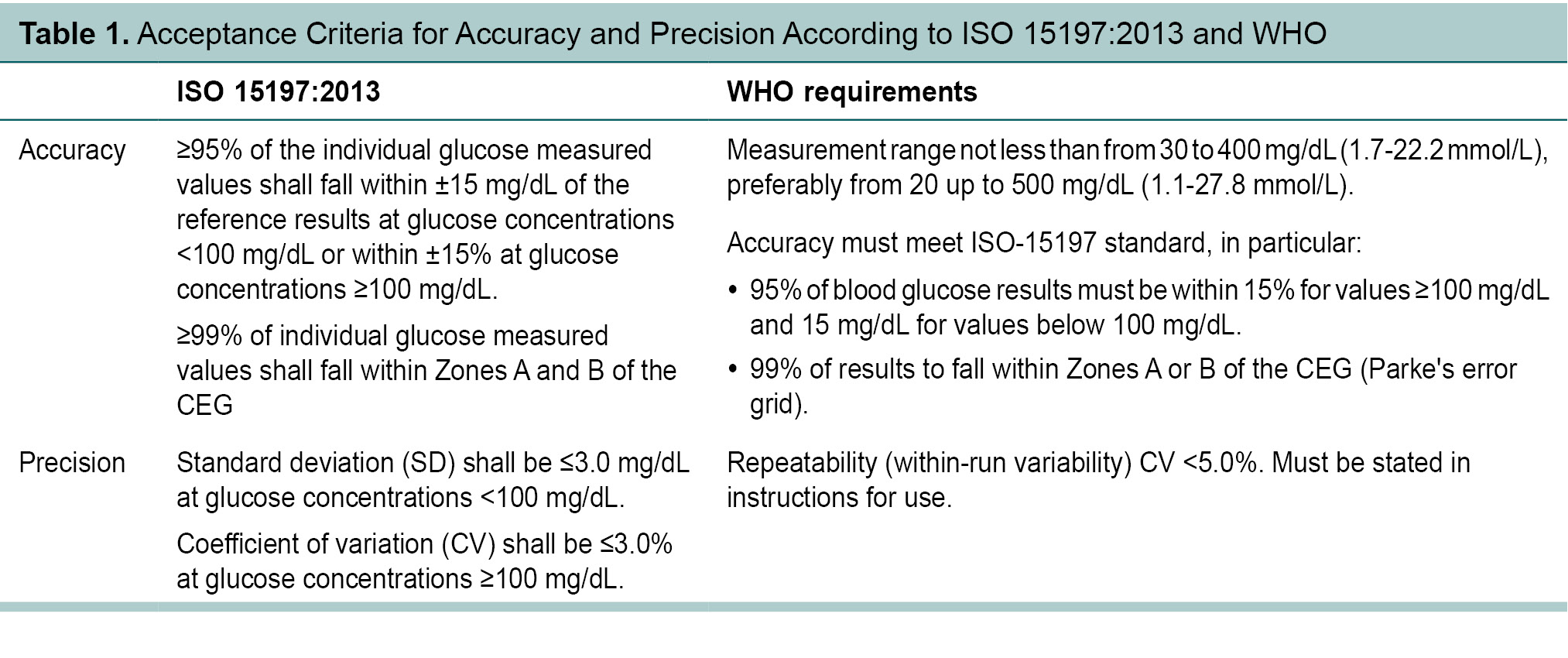
In a comparative study conducted by Pleus et al,
performance evaluation and system accuracy of Accu-Chek Instant®
(99% and 100%) achieved =95% of results within ±15 mg/dL or ±15%19.
In another study
comparing 18 BGM devices conducted by Pleus et al in 126 participants,
Accu-Check Guide® was found to meet the ISO 15197:2015 guidelines
with 100% accuracy20.
Further, in another study conducted by
Breitenbeck et al, Accu-Chek Instant® was found to meet and
exceed the ISO 15197:2013 and EN ISO 15197:2015 requirements with 100% accuracy
and with all the tested lots of the BGM falling within Zone A of the CEG (Fig.
3)21.
In a cross-sectional
study, Choukem et al showed that none of the assessed
glucometers met the criteria for the required level of technical accuracy of
99%; however, Accu-Chek Active® met the ISO 15197:2013
recommendations for clinical accuracy based on Parke's CEG analysis, with 99%
of values falling within Zones A and B. In this study, Accu-Chek Active®
did not meet all the criteria for precision; however, it was found to be
precise in the high-standard concentrations22. In a study conducted
by Dhatt et al, Accu-Chek Active® met the criteria for the required
level of accuracy of 99%, with regards to the lowest and highest proportion in
the range of glycemia =75 mg/L (88% of results within ± 5% and 99.9% of the
results within ±20%, respectively)23. In another study conducted by
Freckmann et al comparing 4 BGM devices, including Accu-Chek Active®
and Accu-Chek Performa®, showed that Accu-Chek Active® met the ISO 15197:2013 criteria for the required level of accuracy (results
within ±15 mg/dL or ±15%) of 99.5% against both hexokinase and glucose oxidase
as reference methods in the hands of trained study personnel. The mean absolute
relative difference (MARD) values (%) varied between four glucometers.
Accu-Chek Active® met the ISO 15197:2003 criteria, with 100% of its
values falling under Zones A and B where errors are clinically acceptable in
the hands of lay users and with 100% of its values falling within ±15 mg/dL or
±20% in the hands of trained study personnel24.
Scandinavian evaluation of laboratory equipment
for primary health care (SKUP) evaluated the accuracy of Accu-Chek Instant®.
The evaluation found that under optimal conditions, 100% of the results for
Accu-Chek Instant® were within the allowable deviation limits for
accuracy, and when handled by intended users, 99% of the results were within
the limits (within ±15 mg/dL or 15%) (Fig. 3). This evaluation further
indicated that Accu-Chek Instant® precision was fulfilled both under
optimal conditions and by intended users compared to the glucose hexokinase
method25.
Interfering Agents
While SMBG using BGM is a fairly accurate and
precise way of monitoring a patient’s glucose levels, there are certain
confounders or interfering agents, which can affect the outcomes of the blood
test14,15. Table 2 summarizes the possible factors, which may
interfere with test results. Interference is defined as “a cause of medically
significant difference in the measured test result due to the effect of another
component or property of the sample”26. According to ISO 15197:2013,
hematocrit and interfering substances in the blood can affect the analytical
performance of an SMBG system. A list showing examples of interfering
substances which could be present in the blood samples is given in the annex of
the ISO standard document. Certain examples of commonly known interfering
substances include ascorbic acid, paracetamol/acetaminophen, maltose, etc.
Further, for patients being treated in the intensive care unit, drugs such as
cardiac inotropes and vasoconstrictors may act as interfering agents. Venous
blood is the preferred sample for the evaluation of influence quantities.
Hematocrit influences are required to be investigated for a minimum of five
different hematocrit levels at three defined glucose concentrations.
Interfering substances are required to be investigated for a minimum of two
defined glucose concentrations. ISO 15197:2013 defines that influence
quantities >10 mg/dL and >10% difference between the test sample and the
respective control sample for glucose concentrations =100 mg/dL and >100
mg/dL, respectively, are required to be reported in the instructions for use
along with the respective hematocrit levels or interfering substance
concentrations11.

Human factors such as incorrect use of blood
glucose meters, incorrect performance of coding, inappropriate storage and
usage of test strips, inappropriate education of patients and the diabetes
team, manufacturing factors such as lot-to-lot variances, vial-to-vial
variances, and strip-to-strip variances, and environmental factors such as
temperature, humidity, altitude, and electromagnetic radiation act as external
interfering agents. In addition to external factors, low hematocrit, high
triglycerides, abnormal levels of bilirubin, and uric acid act as internal
interfering agents14,15. Considering the wide variety of interfering
agents, it is pertinent for a device to be unaffected by these agents and
provide appropriate readings in the presence of such confounders.
According to the evaluation report of the
Accu-Chek Active® system, the system had no interference from 31
tested interfering agents, except ascorbic acid, galactose, xylose, and
ceftriaxone16. The Accu-Chek Guide® system was evaluated
for interference with 202 potential interfering agents and was only found to be
affected by high levels of ascorbic acid, triglycerides, and xylose15,18.
Similar results were observed for the Accu-Chek Instant® system17.
In various studies, topical agents such as
hydroquinone-containing creams and other topical lotions and creams have been
shown to be associated with significant false increase in capillary glycemia,
irrespective of the enzymatic system of the glucometer used, which can lead to potentially wrong clinical decisions. Authors of these studies advocate for hand hygiene to achieve optimal responses22,27. Further, patients with comorbid conditions
such as chronic kidney disease and hyperlipidemia have impaired blood parameters, which can potentially interfere with the accuracy of blood glucose results
14,28.
In a study conducted by Hattemer et al in
patients with type 1 DM, type 2 DM, and nondiabetic population, Accu-Chek
Instant® was not affected by varying hematocrit levels in the
patient population29.
Certain recent therapeutic approaches for
diabetes management, such as the use of sodium-glucose co-transporter 2 (SGLT2)
inhibitors, may interfere with SMBG. However, in a study conducted by Mills
et al to evaluate the effect of various SGLT2 inhibitors (Canagliflozin, Dapagliflozin, Empagliflozin, and Ertugliflozin) on various Accu-Chek® devices (Accu-Chek Active®, Accu-Chek Aviva®, Accu-Chek
Guide
®, Accu-Chek Instant®, and Accu-Chek Performa®). It was concluded that canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin do not interfere with the Accu-Chek
® systems at the measured concentrations30.
SKUP evaluation
concluded that glucose measurements on Accu-Chek Instant® were not
affected by hematocrit within the range tested (29%-50%)25.
User Performance Evaluation and Preference
ISO 15197:2013 requires user performance
evaluation as part of accuracy assessment and is concerned with assessing
whether intended users are able to obtain accurate blood glucose measurement
results. According to this standard, 95% of measurements obtained within ±15
mg/dL of the reference measurement results at glucose concentrations <100
mg/dL and within ±15% at glucose concentrations =100 mg/dL in at least 100
people with diabetes11. Previously conducted research indicates that
proper usage of the BGM device may positively affect patients’ engagement and
adherence to the treatment and may lead to an improvement in their quality of
life31,32.
In a study
conducted by Pinelli et al, which investigatedthe patient and provider
preference from 3 rounds of interviews with patients using the Accu-Chek
Instant® glucometer against findings from the literature review, 89%
of the participants mentioned that they would recommend the device. In this
study, the majority of participants (>75%) mentioned that the backlit
display made reading results easier, it was easy to apply blood on the dosing
area and to eject the strip, and they could learn to operate the device without
training from their health care provider33. In another study
comparing four glucometers, including Accu-Chek Instant®, most
participants agreed or completely agreed that manuals provided with the system
were clear and appropriate; this comprised instructions for use (88% agreement
rate), quick reference guide (94%), and reagent system package insert (84%),
with only small differences between the systems. Accu-Chek Instant®
was rated to be easy to use by 99% of the participants of the study19.
In a study conducted with 197 participants,
Harvey et al found that Accu-Check Guide® meters had superior
usability compared to other meters. The majority of the study participants
found all aspects of the BGM system, including the test strips, strip vials,
and data analysis on the BGM and the mobile app, to be acceptable for their
lifestyle and to provide a better testing experience34.
In the SKUP
evaluation, user-friendliness was assessed by 88 persons with diabetes. A total
of 47 participants had one or more positive comments regarding the operation
facilities of Accu-Chek Instant®, and 46 participants had one
or more negative comments. The meter is easy to use, has a short measuring
time, needs a small amount of blood, has a convenient small size, is
lightweight, easy to read the result, and has clear, large, and illuminated
numbers. The large numbers were among few of the positive comments noted by the
participants. The size of the strips and the convenience of handling them were
noted in the negative comments25.
Other Factors
In addition to accuracy, precision, user
preference, and interfering agents, WHO and ISO standard recommendations,
additional stability parameters such as storage temperature, operating
temperature, altitude, humidity, etc. are
also included. According to the data from system evaluation reports, Accu-Chek® Instant®,
Instant S®, Guide®, and Active® significantly
surpass these requirements16-18.
Apart from the requirements of standard
organizations and regulatory agencies, certain factors, such as economic
analysis of the device, may be beneficial in assessing the preferability of one
device over another. Economic evaluation has shown that an SMBG based on
technology with software to analyze its results, accompanied by medical
support, brings both health and economic benefits that can be translated into a
reduced cost associated with DM32.
When compared with newer technologies such as
CGM, SMBG is an established technology and can be considered a significantly
cost-effective measure, especially in markets with a predominant out-of-pocket
payment by patients, such as India. The short lifetime of the CGM sensor also
adds to the cost for the patient. The daily costs associated with using CGM can
be as high as US$5-10, amounting to approximately US$3000 of additional costs,
which is unaffordable for most of the patient population in developing countries.
Also, as the technology is rapidly evolving, there can be further increases in
costs related to the upgradation of the device used10. Additionally,
though CGM may appear to be cost-effective in intensively managed patients,
BGMs are considered more cost-effective in nonintensively managed patients35,36.
Another factor contributing to the cost of CGM
devices is the limited number of manufacturers developing CGM devices37.
Though the total cost of CGM is trending downwards through the years,
affordability is still an issue in developing countries. Though significant
data regarding the cost-effectiveness of the two systems is still lacking, it
is reasonable to conclude that SMBG and CGM can be used in a complementary
manner to form an effective strategy for optimal diabetes management.
STRENGTHS AND LIMITATIONS
There are several strengths of this study. We
are a group of researchers from India and have primarily focused on the
Accu-Chek® devices, which are available in India. Though we found
limited studies conducted in India, these data are relevant in Indian context
due to the availability of the devices.
Additionally, we have not restricted ourselves
to only particular type of studies. We have considered a wide range of
literature including clinical studies, systematic reviews, evaluation reports
released by the manufacturer and also the evaluations conducted by regulatory
bodies. The inclusion of these sources makes the data robust.
However, this review is not without limitations.
We have primarily focused on the devices from a single manufacturer, which
precludes any comparative data. As we are a group from India, our goal is to
review the existing data for various devices available in India. Comparison of
variables between various devices can be an interesting topic of future
research.
Further, we tried to understand the
cost-effectiveness of these devices in comparison to newer technologies such as
CGM, however, data was scarce and was not available for Indian scenario. This
topic of understanding comparative cost-effectiveness of various devices and
technologies should be assessed under comprehensive future research.
CONCLUSION
According to the widely available data, the
Accu-Chek® devices (Instant®, Guide®, and
Active®) are compliant with the prescribed requirements as per
15197:2013. Additionally, the devices appear to be cost-effective and are
acceptable to patients based on their usability.
Acknowledgments
Ethical Declaration
As this is a
noninterventional, nonhuman, and nonanimal review study, ethical approval was
not required.
Data Availability
All relevant data is
included in this article.
Author Contributions
SK, NK, RM, CP, JR, VG,
AA had full access to the whole data set and contributed to conceptualization,
data curation, formal analysis, methodology, supervision, validation,
visualization, writing, review, and editing the manuscript.
BS,
RS, SS, SC, SNS, AGU contributed towards methodology, validation,
visualization, writing, review, and editing the manuscript.
Funding
Roche funded the
literature review, medical writing, and editorial support for this study.
Conflict-of-Interest
Disclosure
SK
has received speaker fees from Abbott, Novo Nordisk, Roche, and Sanofi.
NK
has received research grants from the National Health and Medical Research
Council of Australia, Indian Council of Medical Research, and the Global
Alliance for Chronic Diseases. He has also been a principal investigator for
several industry-sponsored clinical trials for Novo Nordisk, Novartis, Eli
Lilly, and Amgen. He is also an elected executive committee member of the
governing council of the Endocrine Society of India (2022-25), All India
Association for Advancing Research in Obesity (2022-25), and the Indian Society
of Bone and Mineral Research (2023-2025).
BS, RS, AGU, SS, SC, and
SNS declare no conflicts of interest.
RM,
JR, CP, VG, and AA are employees of Roche Diabetes Care.
REFERENCES
1.
IDF Diabetes Atlas 2021.
Available from: https://diabetesatlas.org/atlas/tenth-edition/. Accessed May
10, 2024.
2.
Anjana RM, Unnikrishnan R,
Deepa M, Pradeepa R, Tandon N, Das AK, et al; ICMR-INDIAB Collaborative Study
Group. Metabolic non-communicable disease health report of India: the
ICMR-INDIAB national cross-sectional study (ICMR-INDIAB-17). Lancet Diabetes Endocrinol.
2023;11(7):474-89.
3.
ICMR
Guidelines for Management of Type 2 Diabetes -2018.
ICMR New Delhi. Available from:
https://main.icmr.nic.in/sites/default/files/guidelines/ICMR_Guidelines
Type2diabetes2018_0.pdf. Accessed May 12, 2024.
4.
Kirk JK, Stegner J.
Self-monitoring of blood glucose: practical aspects. J Diabetes Sci Technol.
2010;4(2):435-9.
5.
Pollock RF, Valentine WJ,
Goodall G, Brändle M. Evaluating the cost-effectiveness of self-monitoring of
blood glucose in type 2 diabetes patients on oral anti-diabetic agents. Swiss
Med Wkly. 2010;140:w13103.
6.
Tunis SL, Minshall ME.
Self-monitoring of blood glucose in type 2 diabetes: cost-effectiveness in the
United States. Am J Manag Care. 2008;14(3):131-40.
7.
HEARTS D: diagnosis and
management of type 2 diabetes. World Health Organization. 22nd April, 2020.
Available from:
https://www.who.int/publications-detail-redirect/who-ucn-ncd-20.1. Accessed May
10, 2024.
8.
Tonyushkina K, Nichols JH.
Glucose meters: a review of technical challenges to obtaining accurate results.
J Diabetes Sci Technol. 2009;3(4):971-80.
9.
Fortwaengler K, Campos-Náñez E,
Parkin CG, Breton MD. The financial impact of inaccurate blood glucose
monitoring systems. J Diabetes Sci Technol. 2018;12(2):318-24.
10.
Tripathi S, Jindal S, Chawla M,
Gupta A, Jha S, Phadke U, et al. The implication of time-in-range for the
management of diabetes in India: a narrative review. Clin Diabetol.
2022;11(3):192-9.
11.
In vitro diagnostic test
systems—requirements for blood-glucose monitoring systems for self-testing in
managing diabetes mellitus (ISO 15197:2013). International Organization for
Standardization 2015. Available from: https://www.iso.org/standard/54976.html
12.
WHO Technical specifications
for blood glucose meter, handheld. WHO. Available from:
https://cdn.who.int/media/docs/default-source/medical-devices/priority-medical-devices/medevis_tech_specs_blood_glucose_meter_01_2023.xlsx?sfvrsn=ff246958_1&download=true
13.
Ferri
S, Kojima K, Sode K. Review of glucose oxidases and glucose dehydrogenases: a
bird’s eye view of glucose sensing enzymes. J Diabetes Sci Technol.
2011;5(5):1068-76.
14.
Erbach
M, Freckmann G, Hinzmann R, Kulzer B, Ziegler R, Heinemann L, et al. Interferences and limitations in blood glucose
self-testing: an overview of the current knowledge. J Diabetes Sci Technol.
2016;10(5):1161-8.
15.
Hauss O, Hinzmann R, Huffman B.
Drug interference in self-monitoring of blood glucose and the impact on patient
safety: we can only guard against what we are looking for. J Diabetes Sci
Technol. 2024;18(3):727-32.
16.
Accu-Chek® Active
System Evaluation Report
17.
Accu-Chek® Instant
System Evaluation Report
18.
Accu-Chek® Guide
System Evaluation Report
19.
Pleus S, Baumstark A, Schauer
S, Kölle J, Jendrike N, Mende J, et al. User performance evaluation and system
accuracy assessment of four blood glucose monitoring systems with color coding
of measurement results. J Diabetes Sci Technol. 2024;18(3):644-52.
20.
Pleus S, Baumstark A, Jendrike
N, Mende J, Link M, Zschornack E, et al. System accuracy evaluation of 18
CE-marked current-generation blood glucose monitoring systems based on EN ISO
15197:2015. BMJ Open Diabetes Res Care. 2020;8(1):e001067.
21.
Breitenbeck N, Brown A.
Accuracy assessment of a blood glucose monitoring system for self-testing with
three test strip lots following ISO 15197:2013/EN ISO 15197:2015. J Diabetes
Sci Technol. 2017;11(4):854-5.
22.
Choukem SP, Sih C, Nebongo D,
Tientcheu P, Kengne AP. Accuracy and precision of four main glucometers used in
a Sub-Saharan African country: a cross-sectional study. Pan Afr Med J.
2019;32:118.
23.
Dhatt GS, Agarwal MM, Othman Y,
Nair SC. Performance of the Roche Accu-Chek active glucose meter to screen for
gestational diabetes mellitus using fasting capillary blood. Diabetes Technol
Ther. 2011;13(12):1229-33.
24.
Freckmann G, Baumstark A,
Jendrike N, Rittmeyer D, Pleus S, Haug C. Accuracy evaluation of four blood
glucose monitoring systems in the hands of intended users and trained personnel
based on ISO 15197 requirements. Diabetes Technol Ther. 2017;19(4):246-54.
25.
SKUP Evaluation of Accu-Chek
Instant: A system for measurement of glucose manufactured by Roche Diabetes
Care GmbH. Scandinavian Evaluation of Laboratory Equipment for Primary Health
Care. 2017. Available from: https://www.skup.org/media/uy4hsx4u/2017-113-accu-chek-instant-glucose-rapport.pdf
26.
Interference Testing in
Clinical Chemistry (CLSI Guideline EP07). Clinical and Laboratory Standards
Institute 2018. Available from: Available from:
https://clsi.org/standards/products/method-evaluation/documents/ep07/
27.
Choukem
SP, Efie DT, Djiogue S, Kaze FF, Mboue-Djieka Y, Boudjeko
T, et al. Effects of hydroquinone-containing creams on capillary glycemia
before and after serial hand washings in Africans. PLoS One.
2018;13(8):e0202271.
28.
Bode B. The accuracy and
interferences in self-monitoring of blood glucose. US Endocr Rev. 2007;2:46-8.
29.
Hattemer
A, Wardat S. Evaluation of hematocrit influence on self-monitoring of blood
glucose based on ISO 15197: 2013: Comparison of a novel
system with five systems with different hematocrit ranges. J Diabetes Sci
Technol. 2018;12:333-40.
30.
Mills K, Roetschke J. Patients
with SGLT2 inhibitor therapy can reliably measure their blood glucose without
interference issues when up-to-date potentiometric and amperometric blood
glucose measurement systems are used. J Diabetes Sci Technol. 2022;16(1):261-3.
31.
Greenwood DA, Grady M.
Healthcare professional perceptions of blood glucose meter features that
support achievement of self-management goals recommended by clinical practice
guidelines. J Diabetes Sci Technol. 2021;15(5):1142-52.
32.
American Association of
Diabetes Educators. An effective model of diabetes care and education: Revising
the AADE7 Self-Care Behaviors®. Diabetes Educ. 2020;46(2):139-60.
33.
Pinelli
M, Lettieri E, Boaretto A, Casile C, Citro G, Zazzaro B, et al. Glucometer
usability for 65+ type 2 diabetes patients: insights on
physical and cognitive issues. Sensors (Basel). 2022;22(16):6202.
34.
Harvey C, Koubek R, Bégat V,
Jacob S. Usability evaluation of a blood glucose monitoring system with a
spill-resistant vial, easier strip handling, and connectivity to a mobile App.
J Diabetes Sci Technol. 2016;10(5):1136-41.
35.
Wan W, Skandari MR, Minc A,
Nathan AG, Winn A, Zarei P, et al. Cost-effectiveness of continuous
glucose monitoring for adults with type 1 diabetes compared with
self-monitoring of blood glucose: The DIAMOND randomized trial. Diabetes Care.
2018;41(6):1227-34.
36.
Kerr D, Duncan I, Repetto E,
Maroun R, Wu A, Perkins C, et al. Cost analysis of self-monitoring blood
glucose in nonintensively managed type 2 diabetes. Am J Manag Care.
2023;29(12):670-5.
37.
Diabetes self-monitoring
devices in low- and middle-income countries. October 2021. Available from:
https://haiweb.org/wp-content/uploads/2021/09/Market-Report_Self-monitoring-Devices-in-LMICs.pdf