Abstract
Background:
Glimepiride/metformin is commonly used in India for its cost-effectiveness and efficacy, but one-third of patients may not achieve adequate glycemic control. Dapagliflozin/linagliptin fixed-dose combination (FDC) could be an effective alternative.
Methods: A retrospective study of type 2 diabetes patients aged >18 years, not meeting glycated hemoglobin (HbA1c) goal <7% on glimepiride and metformin. Patients received dapagliflozin (10 mg)/linagliptin (5 mg) FDC. Changes in
HbA1c, fasting plasma glucose (FPG)/postprandial plasma glucose (PPG), blood pressure, and body weight were analyzed over 3 months. Results: The study included patient (n = 114) with a mean age of 55.15 years and mean duration of 8.5 years.
Adjunct to dietary/lifestyle modifications, up-titration of metformin-glimepiride along with FDC of dapagliflozin/linagliptin demonstrated significant reduction in HbA1c (from mean 8.8 ± 1.12 to 6.92 ± 0.23; p < 0.001), FPG (157.8 ± 28.86 mg/dL
to 113.6 ± 9.24 mg/dL; p < 0.001), and PPG (237.21 ± 40.58 mg/dL to 161.39 ± 12.76 mg/dL; p < 0.001) indicating improved glycemic control. Additionally, there was a marginal decrease in body weight (73.19 to 72.12 kg), systolic blood pressure
(137.6 to 129.11 mmHg), and diastolic blood pressure (84.26 to 78.68 mmHg) largely attributed to dapagliflozin. Treatment was well-tolerated. Conclusion: FDC of dapagliflozin/linagliptin is an attractive therapeutic option for patients with
type 2 diabetes mellitus not controlled with ongoing glimepiride and metformin.
Keywords: Type 2 diabetes mellitus, dapagliflozin, linagliptin, LEAD INDIA, glycemic control
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder characterized by hyperglycemia stemming from insulin resistance and relative insulin deficiency. The management of T2DM typically involves a stepwise approach starting with lifestyle modifications
and oral antidiabetic agents such as metformin, followed by the addition of other agents to achieve glycemic targets. Glimepiride, a sulfonylurea, and metformin, a biguanide, are commonly prescribed together due to their complementary mechanisms of
action. Despite their effectiveness, a substantial proportion of patients do not attain optimal glycemic control with this combination, demanding alternative treatment strategies1-4.
Moreover, the shortcomings of monotherapy in achieving glycemic targets can lead to increased risks of both macrovascular and microvascular complications, as well as hypoglycemic events. Therefore, combination therapies offer a more aggressive and proactive
approach to managing T2DM, potentially reducing the incidence of adverse effects. Moreover, combining different drugs can reduce the pill burden on patients, which experts suggest may improve treatment adherence1-4.
Combining sodium-glucose co-transporter 2 (SGLT2) inhibitors with dipeptidyl peptidase-4 (DPP-4) inhibitors in treating T2DM presents a synergistic strategy targeting multiple pathways involved in glucose regulation. SGLT2 inhibitors like dapagliflozin block
renal glucose reabsorption, increasing urinary glucose excretion, and lowering plasma glucose levels independently of insulin. Meanwhile, DPP-4 inhibitors such as linagliptin enhance glucose-dependent insulin secretion and suppress glucagon release,
further reducing hyperglycemia without raising the risk of hypoglycemia. This dual action addresses insulin resistance and pancreatic dysfunction, central to T2DM’s pathophysiology. Clinical trials confirm that combining SGLT2 and DPP-4 inhibitors
results in synergistic or additive effects on glycemic control, significantly reducing glycated hemoglobin (HbA1c), fasting plasma glucose (FPG), and postprandial plasma glucose (PPG) levels compared to using either medication alone. Moreover, this
combination therapy offers additional benefits such as weight loss, improved blood pressure, and potential cardiovascular advantages, positioning it as a promising option for comprehensive management of T2DM5,6.
This retrospective study aimed to assess the effectiveness of dapagliflozin/linagliptin fixed-dose combination (FDC) in patients with T2DM who had failed to achieve their HbA1c goal (<7%) on a stable regimen of glimepiride and metformin. The study
evaluated changes in FPG, PPG, blood pressure, body weight, and lipid profile after initiating dapagliflozin/linagliptin therapy along with safety and tolerability profile.
Study Design and Participants
This retrospective study included adult patients with T2DM, aged >18 years, who were receiving treatment with glimepiride and metformin but had not achieved an HbA1c goal of <7%. Patients were identified from electronic medical records at a tertiary
care center in India between January 2024 and April 2024. The decision to put patients to dapagliflozin/linagliptin FDC was made by treating physicians based on clinical judgment and the inability to achieve glycemic targets with the existing regimen.
Out of the 190 records screened, 114 patients satisfied inclusion criteria thus were included in the study. Other all medications for respective conditions were continued as per physician’s discretion.
Data Collection and Outcomes
Baseline demographic and clinical characteristics, including age, gender, duration of diabetes, and comorbid conditions, were extracted from medical records. Laboratory parameters at baseline and after 3 months of dapagliflozin/linagliptin therapy were
recorded, including HbA1c, FPG, PPG, lipid profile. Blood pressure and body weight were also monitored at each visit.
Statistical Analysis
Continuous variables were expressed as mean ± SD (standard deviation) or median with interquartile range (IQR) depending on data distribution. Paired t-tests or Wilcoxon signed-rank tests were used to compare changes in continuous variables from
baseline to 3 months post-initiation of dapagliflozin/linagliptin therapy. Categorical variables were presented as frequencies and percentages. Statistical significance was set at p < 0.05. All analyses were performed using statistical software
(e.g., SPSS, version 20).
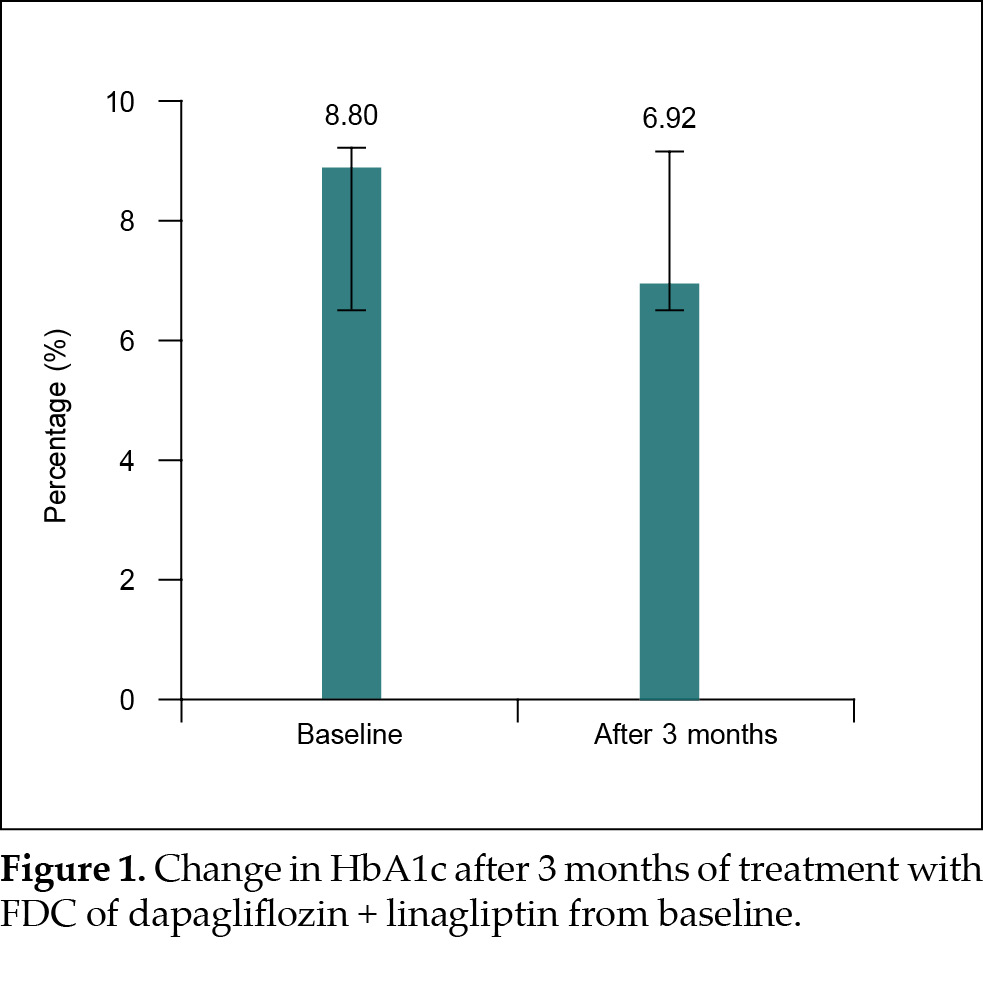
A total of 114 patients with T2DM were included in the study. The mean age of the cohort was 55.15 years, and the mean duration of diabetes was 8.5 years. The majority of patients were male (58%), mean body weight of the cohort was 73.19 kg. Significant
number of patients we having comorbidities of hypertension (69.2%), dyslipidemia (54.3%), or both (36.8%) (Table 1).
At baseline, patients had suboptimal glycemic control with a mean HbA1c of 8.8% ± 1.12% even after glimepiride and metformin therapy. After 3 months of treatment with dapagliflozin/linagliptin FDC, there was a significant reduction in HbA1c to 6.92%
± 0.23% (p < 0.001) (Fig. 1). This corresponded to a mean reduction in HbA1c of 1.88%. Similarly, significant improvements were observed in FPG (from 157.8 ± 28.86 mg/dL to 113.6 ± 9.24 mg/dL; p < 0.001) and PPG (from
237.21 ± 40.58 mg/dL to 161.39 ± 12.76 mg/dL; p < 0.001) (Table 2).
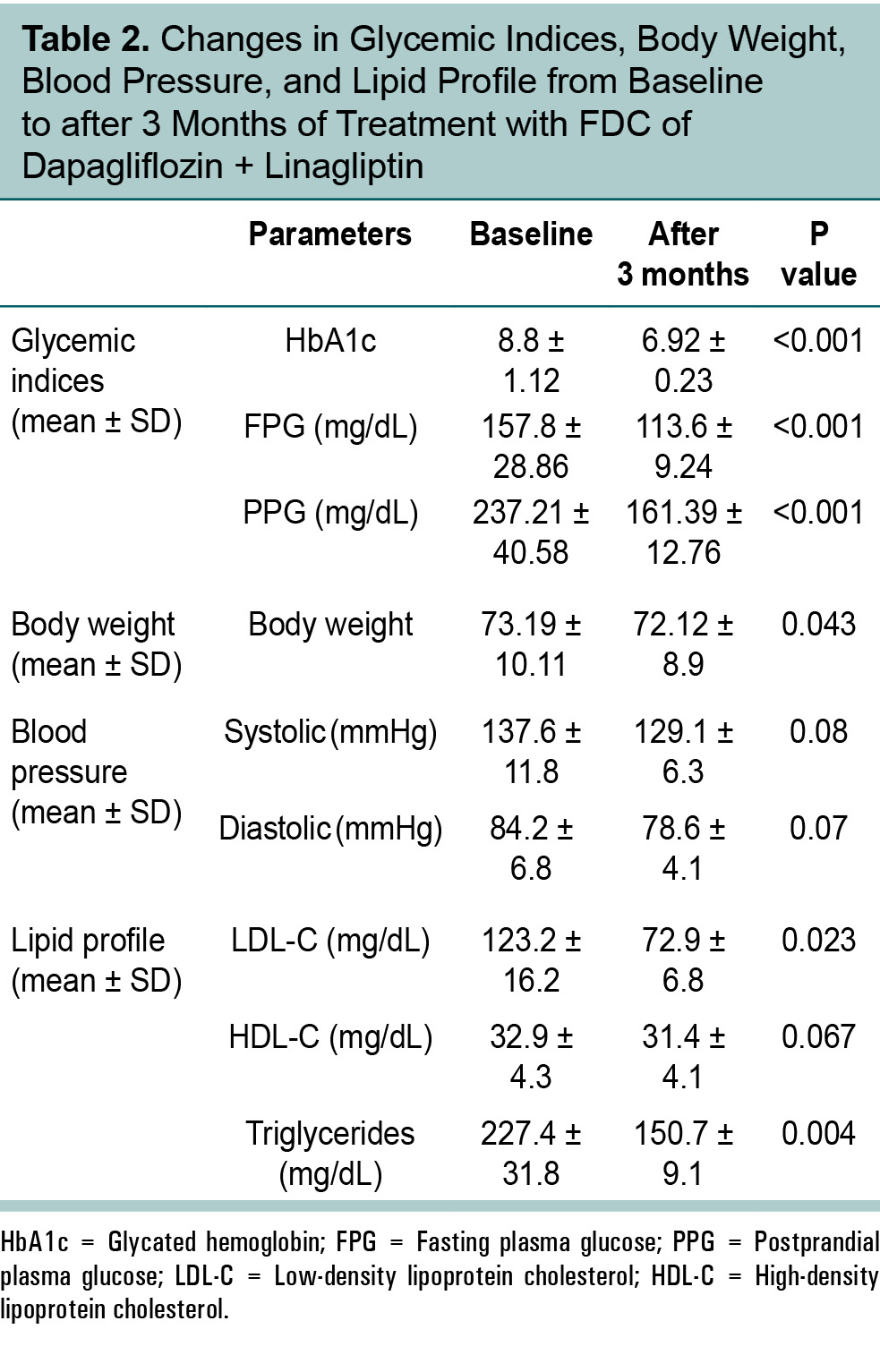
In addition to glycemic improvements, patients experienced modest reductions in body weight (from 73.19 to 72.12 kg), systolic blood pressure (from 137.6 to 129.11 mmHg), and diastolic blood pressure (from 84.26 to 78.68 mmHg) after
3 months of dapagliflozin/linagliptin therapy. These changes were largely attributed to the SGLT2 inhibitor dapagliflozin’s mechanism of action. Moreover, improvement in lipid profile was also observed, demonstrating significant reduction in low-density
lipoprotein cholesterol (LDL-C) and triglycerides from baseline to after 3 months of treatment with FDC of dapagliflozin + linagliptin (Table 2).
Treatment with dapagliflozin/linagliptin FDC was generally well-tolerated, with no reports of severe hypoglycemia or significant adverse events leading to treatment discontinuation. Common side effects included mild genital mycotic infections (3%) and
urinary tract infections (2%), which were managed conservatively.
The present retrospective study contributes to the growing body of evidence supporting the efficacy and safety of combining dapagliflozin and linagliptin in patients with T2DM who have inadequate glycemic control on glimepiride and metformin. The rationale
behind combining SGLT2 inhibitors and DPP-4 inhibitors lies in their complementary mechanisms of action, targeting both insulin resistance, and pancreatic dysfunction.
Mechanistic Synergies
SGLT2 inhibitors, exemplified by dapagliflozin, lower blood glucose levels by inhibiting renal glucose reabsorption, leading to increased urinary glucose excretion. This mechanism operates independently of insulin secretion or action, making it effective
even in patients with varying degrees of beta-cell function impairment. Conversely, DPP-4 inhibitors like linagliptin enhance endogenous insulin secretion in response to elevated blood glucose levels while suppressing glucagon secretion, thereby mitigating
postprandial hyperglycemia without increasing the risk of hypoglycemia. By simultaneously targeting different pathways in glucose metabolism, the combination of dapagliflozin and linagliptin offers a comprehensive approach to improving glycemic control
in T2DM
5,6.
Clinical Efficacy
Consistent with previous clinical trials and observational studies, our findings demonstrate significant improvements in glycemic parameters following initiation of dapagliflozin/linagliptin therapy. The observed reduction in HbA1c from a mean baseline
of 8.8% to 6.92% after 3 months underscores the effectiveness of this combination in lowering overall glucose levels. Furthermore, reductions in FPG and PPG levels highlight the complementary roles of SGLT2 and DPP-4 inhibition in managing both fasting
and meal-induced hyperglycemia. Jain et al in phase 3 study demonstrated significant reduction in HbA1c, FPG, PPG with FDC of dapagliflozin + linagliptin in patients not controlled with metformin, findings similar to our study7. These results
align with the concept that multifactorial interventions are pivotal in achieving and maintaining optimal glycemic targets in T2DM, particularly in patients resistant to conventional monotherapy approaches.
Additional Benefits
Beyond glycemic control, the combination of dapagliflozin and linagliptin demonstrated favorable effects on body weight and blood pressure. Modest reductions in body weight and improvements in systolic and diastolic blood pressure observed in our
study are consistent with the known metabolic and cardiovascular benefits associated with SGLT2 inhibitors. These findings are of particular significance in the context of managing T2DM, where cardiovascular risk reduction remains a critical therapeutic
goal8,9.
In addition to modest reduction of body weight and blood pressure, our study has also shown improvement in lipid profile evident by reduction in LDL and triglycerides. Literature has shown promising effect on dapagliflozin on lipid metabolism, which could
further contribute to its broader cardiovascular benefits in patients with T2DM. The mechanism of action of dapagliflozin involves inhibiting renal glucose reabsorption, leading to increased urinary glucose excretion and subsequent caloric loss, which
in turn promotes weight reduction and improvements in insulin sensitivity. These metabolic changes are accompanied by favorable alterations in lipid profiles, as evidenced by several clinical studies8.
Clinical trials such as the DECLARE-TIMI 58 and EMPA-REG OUTCOME trials have consistently demonstrated that treatment with SGLT2 inhibitors, including dapagliflozin, leads to reductions in circulating levels of triglycerides and LDL-C, while increasing
levels of high-density lipoprotein cholesterol (HDL-C). For instance, in the DECLARE-TIMI 58 trial, dapagliflozin was associated with a significant decrease in triglyceride levels compared to placebo, with a mean change from baseline of –1.0 mmol/L
(p < 0.001). Similarly, LDL-C levels were also reduced by –0.16 mmol/L (p < 0.001), and HDL-C levels increased by 0.03 mmol/L (p < 0.001)9.
These lipid-modifying effects are thought to be mediated through several mechanisms. The caloric loss induced by dapagliflozin leads to increased lipolysis in adipose tissue, thereby reducing circulating triglyceride levels. Moreover, improvements in
insulin sensitivity and reduction in visceral adiposity contribute to decreased hepatic synthesis of triglycerides and LDL-C. The net result is a shift towards a more favorable lipid profile, characterized by lower triglycerides, LDL-C, and higher
HDL-C levels
8-10.
Safety Profile
Importantly, the safety profile of dapagliflozin/linagliptin combination therapy was favorable in our study population. Literature have shown lesser incidence of genital mycotic infection with FDC of SGLT2 inhibitor and DPP-4 inhibitors
11. With adequate guidance on genital hygiene, our study has reported very few cases (3%) of any genital infections in our cohort. This reinforces the tolerability of combining SGLT2 and DPP-4 inhibitors, supporting their use in clinical
practice for patients requiring intensified glycemic management.
Limitations and Considerations
Several limitations of our study warrant consideration. Being retrospective in nature, our study design inherently carries risks of selection bias and confounding variables that may influence outcomes. The relatively short duration of follow-up (3 months)
limits our ability to assess long-term efficacy and safety outcomes associated with dapagliflozin/linagliptin therapy. Future prospective studies with larger sample sizes and longer follow-up periods are needed to further elucidate the durability
of glycemic control and cardiovascular benefits associated with this combination therapy.
The findings from this retrospective study support the use of dapagliflozin/linagliptin FDC therapy as an effective and well-tolerated option for patients with T2DM inadequately controlled on glimepiride and metformin. The synergistic effects of SGLT2
and DPP-4 inhibition on glycemic control, combined with their favorable safety profile and potential cardiovascular benefits, highlight the therapeutic potential of this combination in managing T2DM comprehensively. These results underscore the importance
of individualized treatment approaches tailored to address the multifaceted pathophysiology of T2DM and optimize patient outcomes.
Acknowledgments: None.
Declaration of
Patient Consent: The informed consent requirement was waived by the ethical committee due to the retrospective design of the study.
Funding/Grant Support: This research received no specific grant from any funding agency in the public, commercial, or any profit sectors.
Declaration of
Conflicting Interests: All authors report no conflicts of interest relevant to this research.
1. Davies MJ, Aroda VR, Collins BS, Gabbay RA, Green J, Maruthur NM, et al. Management of Hyperglycemia in Type 2 Diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes
(EASD). Diabetes Care. 2022;45(11):2753-86.
2. Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, et al. Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the
European Association for the Study of Diabetes. Diabetes Care. 2015;38(1):140-9.
3. Kalra S, Das AK, Priya G, Ghosh S, Mehrotra RN, Das S, et al. Fixed-dose combination in management of type 2 diabetes mellitus: expert opinion from an international panel. J Family Med Primary Care. 2020;9(11):5450-7.
4. Xie X, Wu C, Hao Y, Wang T, Yang Y, Cai P, et al. Benefits and risks of drug combination therapy for diabetes mellitus and its complications: a comprehensive review. Front Endocrinol (Lausanne). 2023;14:1301093.
5. Chadha M, Das AK, Deb P, Gangopadhyay KK, Joshi S, Kesavadev J, et al. Expert opinion: Optimum clinical approach to combination-use of SGLT2i + DPP4i in the Indian diabetes setting. Diabetes Ther. 2022;13(5):1097-114.
6. DeFronzo RA, Lewin A, Patel S, Liu D, Kaste R, Woerle HJ, et al. Combination of empagliflozin and linagliptin as second-line therapy in subjects with type 2 diabetes inadequately controlled on metformin. Diabetes Care. 2015;12(38):384-93.
7. Jain A, Vispute A, Dange A, Naskar A, Mondal A, Vivekanand B, et al. A randomized, double-blind, parallel-group phase III trial investigating the glycemic efficacy and safety profile of fixed-dose combination dapagliflozin and linagliptin over linagliptin
monotherapy in patients with inadequately controlled type 2 diabetes with metformin. Diabetes Ther. 2024;15(1):215-27.
8. Saleem F. Dapagliflozin: Cardiovascular safety and benefits in type 2 diabetes mellitus. Cureus. 2017;9(10):e1751.
9. Fatima A, Rasool S, Devi S, Talha M, Waqar F, Nasir M, et al. Exploring the cardiovascular benefits of sodium-glucose cotransporter-2 (SGLT2) inhibitors: expanding horizons beyond diabetes management. Cureus. 2023;15(9):e46243.
10. Hayashi T, Fukui T, Nakanishi N, Yamamoto S, Tomoyasu M, Osamura A, et al. Dapagliflozin decreases small dense low-density lipoprotein-cholesterol and increases high-density lipoprotein 2-cholesterol in patients with type 2 diabetes: comparison with sitagliptin. Cardiovasc Diabetol. 2017;16(1):8.
11. Rodriguez-Gutierrez R, Montori VM. Review: Adding a DPP-4 inhibitor to an SGLT-2 inhibitor reduces genital, but not genitourinary, tract infections. Ann Intern Med. 2018;168(6):JC33.