Abstract
Vonoprazan helps in management and treatment of heartburn, peptic ulcers and gastroesophageal reflux disease. The structure, mechanism of action, and clinical applications have been discussed in this article. A brief review of literature is carried out.
Keywords: Acid peptic disorders, gastroesophageal reflux disease, peptic ulcer
Acid peptic disorders stem from unique yet interrelated pathogenic mechanisms, characterized by either heightened acid secretion or compromised mucosal defense. The diagnosis of gastroesophageal reflux disease (GERD), peptic ulcer disease and
acid peptic disorders is further complicated by frequent overlap with conditions such as functional dyspepsia and irritable bowel syndrome posing challenges in clinical identification
1. GERD is expected to remain a common cause for primary care consultations2.
The incidence of GERD is 15.6 (range 11.046-20.714). The risk factors are age, body mass index (BMI), nonvegetarian diet, tea and coffee, tobacco, and alcohol consumption3. Chavan and Bhaktavatsalam found the prevalence of acid peptic disease
to be very high, i.e., 38.1% in Indians4. Clinically, acid peptic disorders present as heartburn and regurgitation in GERD patients. They also experience extraesophageal symptoms with reflux cough syndrome, which is the most common symptom.
Other symptoms include lower abdominal pain and constipation1.
The impact of acid peptic disorders affects quality of life and causes work absenteeism and if left untreated, it may lead to complications like peptic stricture, Barrett’s esophagus, and esophageal adenocarcinoma. However, the frequency, severity,
and complications of GERD depend on geographic and ethnic factors. Dietary factors are responsible to lower the frequency and severity of GERD and Barrett’s esophagus3.
POTASSIUM-COMPETITIVE ACID BLOCKERS
A novel treatment in acid-related diseases are potassium-competitive acid blockers, which include fexuprazan, revaprazan, tegoprazan, and vonoprazan. Among these, vonoprazan has undergone extensive study and was initially approved in Japan in 2015
for various acid-related diseases including gastric ulcer, duodenal ulcer, reflux esophagitis, and prevention of recurrence of ulcers as well as adjunct therapy for Helicobacter pylori eradication5. The US Food and Drug Administration
(FDA) approved two vonoprazan-containing regimens for H.
pylori treatment and also accepted a new drug application for vonoprazan for the treatment of erosive esophagitis5. Potassium-competitive acid blockers act at the final step in the acid secretory pathway by inhibiting the hydrogen potassium
ATPase (H+/K+-ATPase) transporter on the luminal membrane of gastric parietal cells, the same proton pump targeted by proton pump inhibitors (PPIs). After systemic absorption, potassium-competitive acid blockers concentrate
in parietal cell canaliculi and ionically bind to H+/K+-ATPase transporters to prevent acidifying proton secretion. Once bound, the potassium-competitive acid blockers block K+ ion access to the proton pump. Unlike
PPIs, potassium-competitive acid blockers are acid stable and thus do not require enteric coating or optimal dosing administration 30 minutes prior to meals. Additionally, they are not prodrugs and act immediately at the proton pump. These mechanistic
differences facilitate more rapid attainment of peak plasma levels and onset of action5.
Patients in whom lifestyle modifications are ineffective in controlling GERD symptoms can benefit from pharmacologic therapy. Acid suppression forms the cornerstone of pharmacological management for GERD. Therapeutic agents include PPIs, histamine
2 receptor antagonists, and antacids. Additionally, medications affecting gastrointestinal motility such as prokinetics are also utilized in treatment of GERD6.
The alternative formulations of PPIs include immediate-release omeprazole and modified-release dexlansoprazole in certain regions. Dexlansoprazole MR achieves a higher percentage of time with intragastric pH >4 compared to lansoprazole. However, these
formulations offer only modest advantages in acid secretion control compared to conventional PPIs7.
There is growing interest in potassium-competitive acid blockers as promising alternatives, poised to overcome some limitations associated with PPIs5,7. When protons are transported by H+,K+-ATPase from cytoplasm to the
canalicular space across the apical membrane of parietal cells, an equal amount of K+ ions are counter transported into cytoplasm of parietal cells, so that total process is electrically balanced.
As a result, the activity of H+,K+-ATPase in the parietal cells is regulated by the availability of extracellular K+. Studies on molecular mechanisms of H+,K+-ATPase, therefore, led to discovery of
potassium-competitive acid blockers, which is a new class of antisecretory agents and a novel treatment in acid-related diseases. This class of drugs exhibits their antisecretory effects by competitively blocking availability of K+ for
H+,K+-ATPase8.
VONOPRAZAN
Vonoprazan is a novel, orally active potassium-competitive acid blocker that has demonstrated rapid, potent and long-lasting gastric acid suppression. It has been approved in Japan and 14 other countries in Asia and South America for a number of years
for the treatment of a variety of acid-related diseases7.
Vonoprazan Structure
Vonoprazan is a potassium-competitive acid blocker with the chemical structure C17H16FN3O2S. It is a pyrrole derivative that binds to the potassium ion binding site on the gastric proton pump, preventing the
pump from activating and blocking gastric acid secretion.
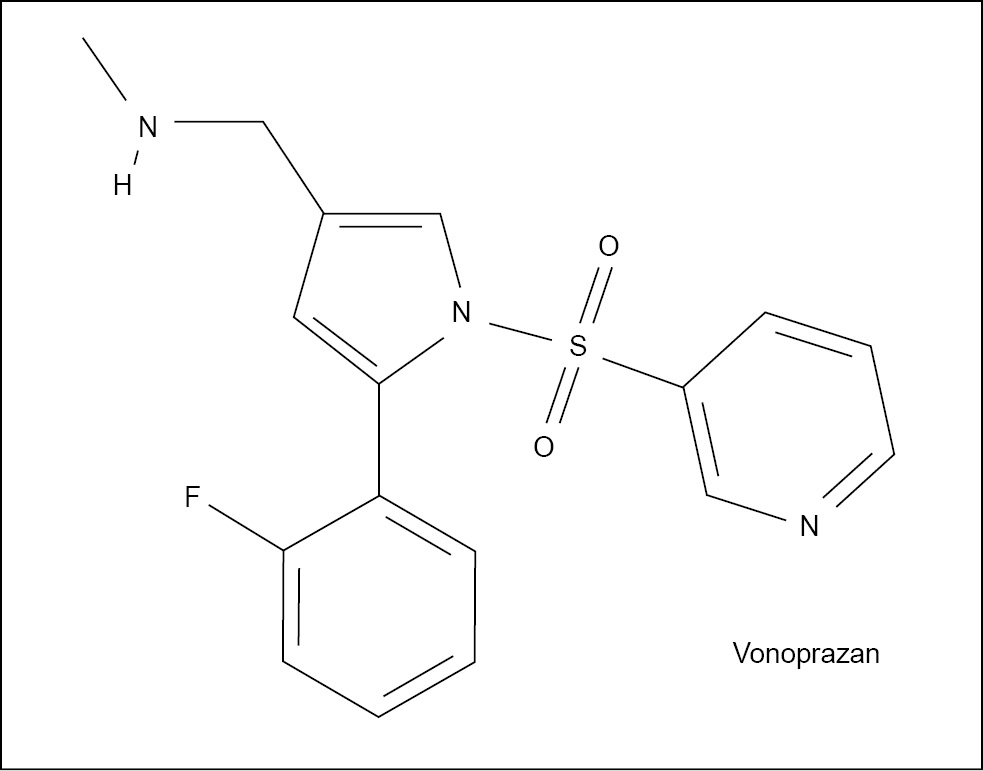
Vonoprazan Uses
The principal use of vonoprazan is in management of GERD. It binds and inhibits H+,K+-ATPase at the final step in the acid secretory pathway in gastric parietal cells and it has different mechanisms of action than conventional PPIs.
It can inhibit the proton pump even in neutral environments with an inhibitory constant (Ki) of 10 nM at pH 7 and 3 nM at pH 6.5. It has stronger potential to inhibit the gastric proton pump than other potassium-competitive acid blocker and lansoprazole7.
The treatment of acid peptic disease is based on providing symptomatic relief and enhancement of ulcer healing in affected area, i.e., esophagus, stomach and duodenum and to heal and maintain remission of erosive esophagitis. The aim of treatment is also
to prevent complications and recurrence and to improve the quality of life6,9. The medical management of GERD comprises of lifestyle modifications and pharmacologic agents10. Lifestyle modifications to treat GERD should remain
the first line of therapy and it includes weight loss, avoiding late night meals, elevating head of bed and steering clear of habits or foods that may worsen symptoms11.
PHARMACODYNAMICS AND PHARMACOKINETIC
The half-life of vonoprazan dissociation by potassium chloride is 12.5 hours in isolated proton pumps. Vonoprazan has high affinity and slow clearance from gastric parietal cells, accumulating in both resting and stimulated conditions. The acid dissociation
constant of vonoprazan is 9.37, which is higher than that of conventional PPIs and other potassium-competitive acid blockers. When vonoprazan is exposed to acidic conditions, it is instantly protonated and remains stable. It can accumulate, function,
and bind the proton pump of gastric parietal cells in strongly acidic secretory canaliculi. The dissociation rate of vonoprazan from H+,K+-ATPase is slow and acid does not decompose it. Nonionic type of vonoprazan is decreased
in a strong acidic secretory canaliculi and passive transport from the acidic secretory canaliculus to the cytoplasm is inhibited. It is therefore retained for a long time inside the parietal cells and can inhibit H+,K+-ATPase
that is activated by further stimulation of acid secretion. The concentration of vonoprazan is up to 108-fold higher in secretory canaliculus of parietal cell than in plasma. It can stay in the protonated form and binds to H+,K+-ATPase
subunit to compete with potassium binding and inhibits function of pump. The binding of vonoprazan to the proton pump is ionic, and its effects are reversible and dose-dependent. Vonoprazan can be taken regardless of meal ingestion and the rate of
absorption is not affected by meals. It is rapidly absorbed and the time taken to reach maximum concentration in plasma is less than 2 hours after oral administration. After absorption, the half-life in plasma is approximately 2 hours for conventional
PPIs, but up to 9 hours for vonoprazan. Thus, vonoprazan stays in blood longer and can block acid secretion continuously
7.
Vonoprazan demonstrated noninferiority and superiority compared to PPI lansoprazole in both healing and maintenance of erosive esophagitis. This advantage was particularly notable in cases of more severe erosive esophagitis12. Vonoprazan has
noninferior efficacy compared to lansoprazole in terms of erosive esophagitis healing rate13. Transitioning from a PPI to vonoprazan is significantly linked to symptom improvement in GERD patients resistant to PPI therapy. Hence, vonoprazan
presents a promising treatment option for PPI-resistant GERD14. It significantly improved heartburn and patient satisfaction, among patients with PPI-resistant GERD15. Vonoprazan 20 mg shows a similar tolerability profile to
lansoprazole 30 mg and is noninferior in terms of gastric ulcer healing. It also demonstrates similar efficacy for duodenal ulcer healing, despite
not meeting the noninferiority criteria16. Vonoprazan 10-20 mg demonstrated efficacy
comparable to lansoprazole 15 mg in preventing peptic ulcer recurrence during low-dose aspirin therapy, exhibited a similar long-term safety profile and was well-tolerated17.
A vonoprazan-based regimen is more effective than a PPI-based regimen as a first-line H. pylori eradication therapy18. Vonoprazan triple therapy and vonoprazan quadruple therapy regimens demonstrated increased H. pylori eradication
rates compared to traditional quadruple therapy. Vonoprazan triple therapy exhibited fewer side effects, suggesting its potential applicability for
H. pylori eradication in clinical practice19.
Vonoprazan Dosage and Administration
Vonoprazan film-coated tablets of 10 and 20 mg are available. They can be taken without regard to food or timing of food and should be swallowed and not chewed. The recommended dosage of vonoprazan is 20 mg once a day for 4 to 8 weeks in reflux esophagitis,
10 mg once a day for maintenance of healing esophagitis, 20 mg once a day for 8 weeks and 6 weeks for gastric and duodenal ulcers, respectively, 10 mg once a day for prevention of nonsteroidal anti-inflammatory drug (NSAID)-induced ulcer and 20 mg
twice daily with triple drug regimen for 1 week for H. pylori eradication.
Vonoprazan Contraindications
Vonoprazan is contraindicated in patients with known hypersensitivity to vonoprazan or any component of vonoprazan, rilpivirine containing products. As a precaution, vonoprazan should not be given during pregnancy and lactation.
Vonoprazan Side Effects
The side effects of vonoprazan include nausea, headache, dysgeusia, dyspepsia, gastritis, pain abdomen, abdominal distension, diarrhea, nasopharyngitis, vulvovaginal candidiasis, urinary tract infection, and hypertension.
CONCLUSION
Vonoprazan plays a significant role in management of acid peptic diseases, especially in the treatment of GERD, peptic ulcer disease and H. pylori infection. It causes potent acid suppression, rapid symptom relief and has better outcome as compared
to traditional PPI therapies. Further, it can be given empty stomach as well as after meals.
REFERENCES
1. Rai RR, Gangadhar A, Mayabhate MM. Clinical profiling of patients with acid peptic disorders (APD) in India: a cross sectional survey of clinicians. Int J Basic Clin Pharmacol. 2017;6:194-202.
2. Zhang D, Liu S, Li Z Wang R. Global, regional and national burden of gastroesophageal reflux disease, 1990-2019: update from the GBD 2019 study. Ann Med. 2022;54(1):1372-84.
3. Rai S, Kulkarni A, Ghosal UC. Prevalence and risk factors for gastroesophageal reflux disease in the Indian population: a meta-analysis and meta-regression study. Indian J Gastroenterol. 2021;40(2):209-19.
4. Chavan MS, Bhaktavatsalam M. Prevalence and risk factors of acid peptic ulcer disease at a tertiary care hospital. Int J Adv Med. 2018;5(4):988-92.
5. Wong N, Reddy A, Patel A. Potassium-competitive acid blockers: present and potential utility in the armamentarium for acid peptic disorders. Gastroenterol Hepatol (N Y). 2022;18(12):693-700.
6. Rawla P, Sunkara T, Ofosu A, Gaduputi V. Potassium-competitive acid blockers-are they the next generation of proton pump inhibitors? World J Gastrointest Pharmacol Ther. 2018;9(7):63-8.
7. Oshima T, Miwa H. Potent potassium-competitive acid blockers: a new era for the treatment of acid-related diseases. J Neurogastroenterol Motil. 2018;24(3):334-44.
8. Echizen H. The first-in-class potassium-competitive acid blocker, vonoprazan fumarate: pharmacokinetic and pharmacodynamic considerations. Clin Pharmacokinet. 2016;55(4):409-18.
9. Mejia A, Kraft WK. Acid peptic diseases: pharmacological approach to treatment. Expert Rev Clin Pharmacol. 2009;
2(3):295-314.
10. Kröner PT, Cortés P, Lukens FJ. The medical management of gastroesophageal reflux disease: a narrative review. J Prim Care Community Health. 2021;12:21501327211046736.
11. Katz PO, Dunbar KB, Schnoll-Sussman FH, Greer KB, Yadlapati R, Spechler SJ. ACG clinical guideline for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol. 2022;117(1):27-56.
12. Laine L, DeVault K, Katz P, Mitev S, Lowe J, Hunt B, et al. Vonoprazan versus lansoprazole for healing and maintenance of healing of erosive esophagitis: a randomized trial. Gastroenterology. 2023;164(1):61-71.
13. Xiao Y, Zhang S, Dai N, Fei G, Goh KL, Chun HJ, et al. Phase III, randomised, double-blind, multicentre study to evaluate the efficacy and safety of vonoprazan compared with lansoprazole in Asian patients with erosive oesophagitis. Gut. 2020;69(2):224-30.
14. Niikura R, Yamada A, Hirata Y, Hayakawa Y, Takahashi A, Shinozaki T, et al. Efficacy of vonoprazan for gastro-esophageal reflux symptoms in patients with proton pump inhibitor-resistant non-erosive reflux disease. Intern Med. 2018;57(17):2443-50.
15. Gotoh Y, Ishibashi E, Honda S, Nakaya T, Noguchi C, Kagawa K, et al. Efficacy of vonoprazan for initial and maintenance therapy in reflux esophagitis, nonerosive esophagitis, and proton pump inhibitor-resistant gastroesophageal reflux disease.
Medicine (Baltimore). 2020;99(11):e19520.
16. Miwa H, Uedo N, Watari J, Mori Y, Sakurai Y, Takanami Y, et al. Randomised clinical trial: efficacy and safety of vonoprazan vs. lansoprazole in patients with gastric or duodenal ulcers - results from two phase 3, non-inferiority randomised controlled
trials. Aliment Pharmacol Ther. 2017;45(2):240-52.
17. Kawai T, Oda K, Funao N, Nishimura A, Matsumoto Y, Mizokami Y, et al. Vonoprazan prevents low-dose aspirin-associated ulcer recurrence: randomised phase 3 study. Gut. 2018;67(6):1033-41.
18. Maruyama M, Tanaka N, Kubota D, Miyajima M, Kimura T, Tokutake K, et al. Vonoprazan-based regimen
is more useful than PPI-based one as a first-line Helicobacter pylori eradication: a randomized controlled trial. Can J Gastroenterol Hepatol. 2017;2017:4385161.
19. Huang J, Lin Y. Vonoprazan on the eradication of Helicobacter pylori infection. Turk J Gastroenterol. 2023;34(3):221-6.