Abstract
Splenic rupture, although rare in dialysis patients, can be life-threatening. In this case, a 45-year-old man with kidney failure on hemodialysis experienced sudden and severe abdominal pain in the left flank, without any history of trauma. He displayed
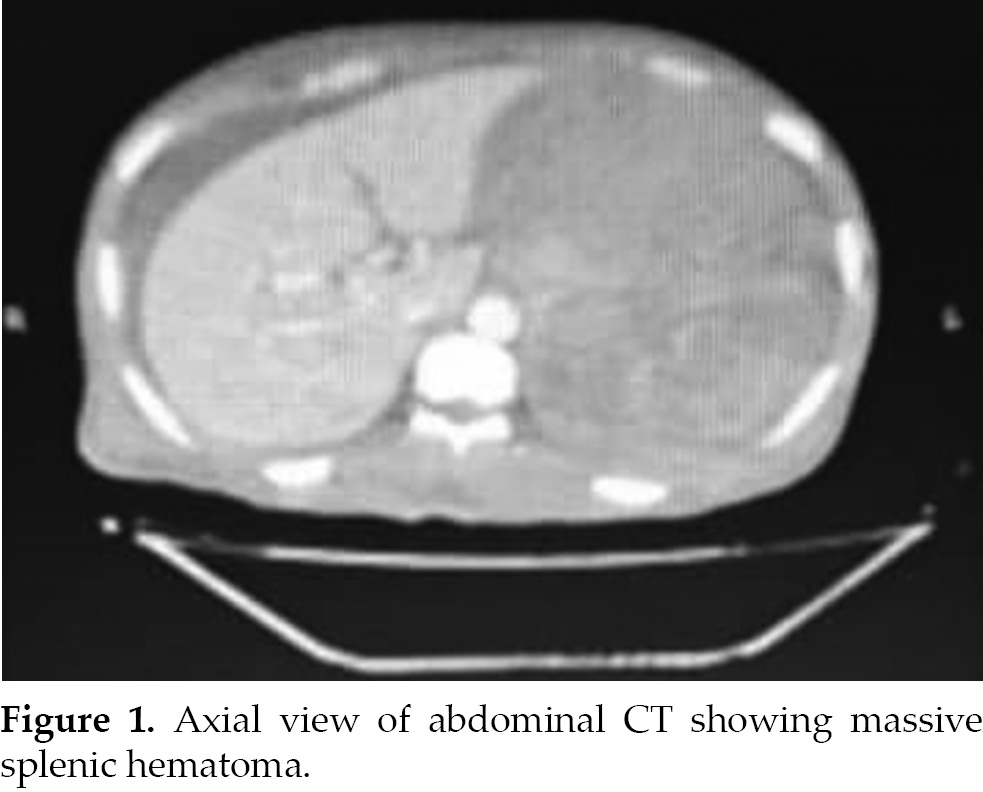
symptoms of hypovolemic shock, characterized by pallor, hypotension, and tachycardia. Additionally, he had abdominal distension and tenderness. An abdominal CT scan revealed a splenic hematoma and intra-abdominal hemorrhage. The patient required a
splenectomy to address the ongoing bleeding, but unfortunately, he succumbed to post-splenectomy sepsis 2 weeks later.
Keywords: Atraumatic, splenic rupture, hemodialysis, platelet dysfunction, kidney failure
Atraumatic splenic rupture is less common than trauma-related cases1. However, in dialysis patients, platelet dysfunction significantly increases the risk of bleeding from internal organs like the spleen1. Nontraumatic splenic rupture
can also occur due to factors such as heparin use, infections, splenic infarction, amyloidosis, and portal hypertension1-4.
Spontaneous splenic rupture is a potentially life-threatening condition and it is important to maintain a high level of suspicion in kidney failure patients experiencing abdominal pain and bleeding. In cases of hemodynamic instability, splenectomy may
be necessary. However, it carries a risk of post-splenectomy sepsis. In this case, a patient with kidney failure, on hemodialysis, experienced spontaneous splenic rupture, but unfortunately developed post-splenectomy sepsis and passed away.
Medical History
A 45-year-old man with kidney failure due to chronic glomerulonephritis began hemodialysis in January 2024. He visited the emergency department with severe abdominal pain that had been ongoing for 8 hours. The pain started suddenly, was sharp, and rated
as 10 on a scale of 1 to 10.
It was present throughout his abdomen, particularly severe on the left side, and worsened with movement. He had no prior history of left chest wall or shoulder pain, no previous instances of similar pain, and no abdominal injuries. He mentioned passing
loose, watery stool before coming to the hospital but had not experienced vomiting, increased abdominal swelling, or a fever. About 1 month ago, he was treated for an anterior chest wall abscess, which had developed at the point of previous insertion
of a subclavian venous catheter.
He had an incision and drainage and was treated with antibiotics in the outpatient clinic but did not return for a follow-up. He had been consistently receiving hemodialysis twice a week along with intradialytic doses of unfractionated heparin (5,000
IU).
For the past 2 months, he had been producing limited urine and developed ascites, requiring occasional abdominal paracentesis guided by ultrasound. Four days before his hospital visit, his packed cell volume was measured at 28% before his routine dialysis
session. He did not have diabetes and had not recently undergone surgery.
His regular medications included nifedipine, torsemide, hydralazine, metoprolol, iron sucrose, vitamins, and erythropoietin.
On examination, he was sweaty, pale, and in shock with pulse rate and blood pressure of 110 bpm and 97/54 mmHg, respectively. His pulse was weak but equal on both sides. The abdomen was distended and generalized abdominal tenderness, maximal in the left
hypochondriac region, was present. There were no significant findings in other systems.
Initial differential diagnosis:
· Suspected abdominal aortic dissection
· Splenic rupture
· Spontaneous bacterial peritonitis.
Treatment
He was admitted to the high-dependency unit and started on intravenous noradrenaline. A blood transfusion of 4 pints of blood and 1 unit of fresh frozen plasma was urgently requested. He was also administered intravenous antibiotics (meropenem and metronidazole)
and the general surgical team was promptly consulted for further assessment.
Oxygen therapy was initiated and he was advised nil per os. He underwent laparotomy, on Day 1 of admission which revealed a ruptured spleen with a subcapsular hematoma of approximately 2 liters and 2.5 liters of blood in his abdomen (hemoperitoneum) necessitating
a splenectomy. He received an additional 5 units of blood and 2 units of fresh frozen plasma due to significant blood loss during surgery.
While on inotropic support, his pulse rate was 102 bpm and his blood pressure was 130/60 mmHg. He was not given heparin for his subsequent dialysis. He had pneumococcal and meningococcal vaccination on the 7th day of surgery as vital signs were stable
and results were normal.
Outcome and Follow-Up
On the 13th day of surgery, he developed a massive upper gastrointestinal bleed and blood parameters were consistent with a disseminated intravascular coagulopathy from sepsis (see Table 1).

Thereafter, he received 4 units of fresh frozen plasma with blood and but bleeding did not stop. He succumbed to the illness on 15th day of surgery.
DISCUSSION
Atraumatic splenic rupture is a less common but potentially fatal cause of massive intra-abdominal hemorrhage. Splenic rupture can occur in a healthy spleen5, although splenic injury from trauma is more prevalent. A systematic review of over
800 published cases identified the main causes of nontraumatic splenic rupture as malignancies, infections, and inflammatory diseases (30%, 27%, and 20%, respectively)6,7.
Other causes included drugs (9%), mechanical causes such as pregnancy (7%), and unknown causes (7%)6,7. The mortality rate was 12%, with most deaths occurring in patients with splenomegaly, those over the age of 40, and those with malignancies6,7.
Several mechanisms of splenic rupture have been postulated8,9.
However, the most widely accepted explanations include raised intra-abdominal pressure due to contracting abdominal muscles during physical activities, intrasplenic cellular congestion, and increased fragility of splenic blood vessels resulting from
infarctions and thromboses8,9.
Spontaneous splenic rupture in dialysis patients is most frequently caused by uremic coagulopathy8,10. This condition is characterized mainly by platelet dysfunction, specifically a decrease in active integrin alpha IIb/beta 3 (formerly
known as glycoprotein IIb/IIa), due to the accumulation of blood uremic toxins11-13.
However, some other factors also contribute to this condition, for example, the use of antiplatelet medications, administration of heparin during dialysis, and anemia13. Splenic infarcts, thrombocytopenia, portal hypertension, and amyloidosis
are also potential risk factors14.
Conversely, the serum levels of pro-hemostatic clotting factors are generally normal or increased in chronic kidney disease, which may be prothrombotic13.
Additionally, the reduced levels of naturally occurring anticoagulants such as protein C and S may also result in thrombosis in patients having nephrotic syndrome13.
Therefore, strategies to prevent uremic bleeding should include optimization of hemodialysis, treatment of anemia, judicious use of medications such as anticoagulants and antiplatelets, and lastly administration of desmopressin before high-risk procedures13,15.
Our patient had risk factors for uremic coagulopathy such as exposure to heparin, azotemia, and anemia. He had no other risk factor for splenic rupture. Due to the massive intraperitoneal hemorrhage and hemodynamic instability, he underwent splenectomy
but developed post-splenectomy sepsis afterwards.
Splenic rupture should be considered in the differential diagnosis of abdominal pain with shock in a hemodialysis patient. Urgent investigation is needed for prompt surgical intervention.
Patients on hemodialysis who have had splenectomy are at risk of sepsis and have to be aggressively treated to prevent mortality.
1. Kim HJ, Lee GW, Park DJ, Lee JD, Chang SH. Spontaneous splenic rupture in a hemodialysis patient. Yonsei Med J. 2005;46(3):435-8.
2. Lund L, Nielsen FB, Sørensen K. Spontaneous rupture of the spleen in a patient with uremia. Clin Nephrol. 1988;29(2):107-8.
3. Lloveras JJ, Prevost F, Goudable C, Durand D, That HT, Suc JM. Rupture of the spleen in hemodialyzed patients. Clin Nephrol. 1986;26(3):160.
4. Gascón A, Iglesias E, Bélvis JJ, Berisa F. The elderly haemodialysis patient with abdominal symptoms and hypovolemic shock-splenic rupture secondary to splenic infarction in a patient with severe atherosclerosis. Nephrol Dial Transplant. 1999;14(4):1044-5.
5. Badenoch DF, Maurice HD, Gilmore OJ. Spontaneous rupture of a normal spleen. J R Coll Surg Edinb. 1985;30(5):326-7.
6. Renzulli P, Hostettler A, Schoepfer AM, Gloor B, Candinas D.Systematic review of atraumatic splenic rupture. Br J Surg. 2009;96(10):1114-21.
7. Bona R. Splenomegaly and other splenic disorders in adults. In: Means RT, Tirnauer JS, ed. Up-to-Date [database on the internet] Waltham MA: UpToDate; 2024 [cited 6 September]. Available from: http://www.uptodate.com
8. Gazel E, Açikgöz G, Kasap Y, Yigman M, Günes ZE. Spontaneous splenic rupture due to uremic coagulopathy and mortal sepsis after splenectomy. Int J Crit Illn Inj Sci. 2015;5(2):119-22.
9. Yagmur Y, Kara IH, Aldemir M, Büyükbayram H, Tacyildiz IH, Keles C. Spontaneous rupture of malarial spleen: two case reports and review of literature. Crit Care. 2000;4(5):309-13.
10. Hou YC, Chen YL, Lu KC. Spontaneous arterial calcification: a possible etiology for spontaneous splenic rupture in a patient on maintenance hemodialysis. Blood Purif. 2014;38(2):96-9.
11. Escolar G, Cases A, Bastida E, Garrido M, López J, Revert L, et al. Uremic platelets have a functional defect affecting the interaction of von Willebrand factor with glycoprotein IIb-IIIa. Blood. 1990;76(7):1336-40.
12. Gawaz MP, Dobos G, Späth M, Schollmeyer P, Gurland HJ, Mujais SK. Impaired function of platelet membrane glycoprotein IIb-IIIa in end-stage renal disease. J Am Soc Nephrol. 1994;5(1):36-46.
13. Berns JS. Uremic platelet dysfunction. In: Golper TA, Taylor EN, ed. UpToDate; 2024 [cited 6 September]. Available from: http://www.uptodate.com
14.
AlMuhsin AM, Privitera A, Balhareth A, Sabr K. Spontaneous splenic rupture following colorectal surgery
and hemodialysis. Case Rep Surg. 2019;2019:8278419.
15. Galbusera M, Remuzzi G, Boccardo P. Treatment of bleeding in dialysis patients. Semin Dial. 2009;22(3):279-86.