Abstract
Pheochromocytomas are rare endocrine tumors which secrete catecholamines leading to severe cardiovascular complications. Dilated cardiomyopathy (DCM) is known to exist in patients with pheochromocytomas. The presence of these two conditions makes the
anesthetic management of such cases even more complex. In this article, we present the case of a young male who had both pheochromocytoma and DCM, and who underwent robot-assisted laparoscopic surgery. The challenges in managing this case are discussed.
Keywords: Adrenal pheochromocytoma, retroperitoneal mass, catecholamines, catecholamine-induced cardiomyopathy
Dilated cardiomyopathy (DCM) is an idiopathic clinical condition characterized by left ventricular or biventricular dilation and impaired contractions. DCM is a potentially lethal disease and its 5-year mortality rate is above 50%1. The anesthetic
management of patients with DCM is challenging for anesthesiologists because of poor left systolic function, risk of arrhythmias, chamber enlargement and risk of sudden cardiac death2. Pheochromocytomas are rare tumors of the adrenal medulla.
The catecholamines secreted by the tumor can lead to serious cardiovascular complications including paroxysmal hypertension and cardiac failure. In individuals with pheochromocytomas, activities such as movement, endotracheal intubation, pneumoperitoneum,
and compression of the tumor during surgery can trigger the release of significant amounts of catecholamines3. The substantial fluctuations in blood pressure that occur during surgery are the primary reason why pheochromocytoma resection is
deemed a high-risk procedure. A rapid decline in catecholamine levels following tumor removal may result in difficult-to-manage hypotension, particularly during the induction of anesthesia or the surgical excision of pheochromocytomas. However, if
the pheochromocytoma is removed swiftly, the prognosis is generally very favorable4.
Additionally, patients with these tumors may experience severe complications, such as DCM, which poses considerable challenges and requires more extensive preoperative evaluation and anesthesia management
5. In this report, we discuss a rare case of DCM in an adult patient who underwent a highly risky pheochromocytoma resection.
CASE REPORT
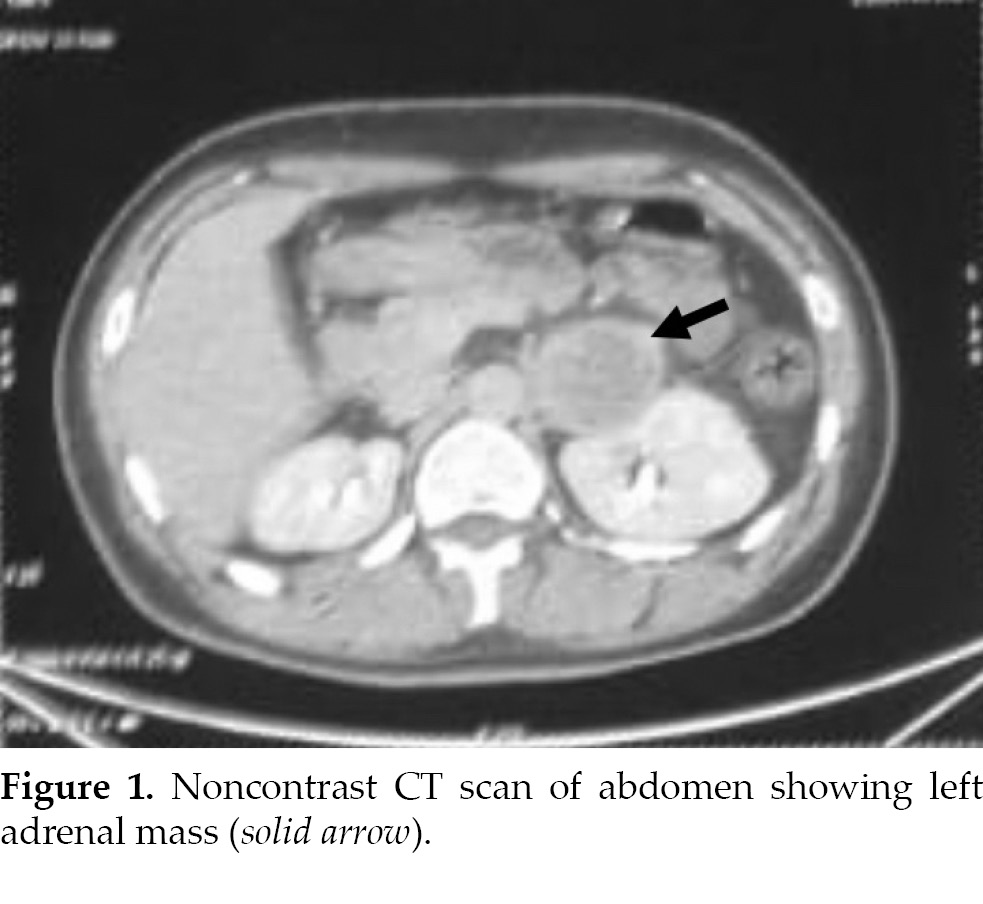
A 23-year-old male was admitted due to a left retroperitoneal mass identified via computed tomography (CT) scan while investigating left-sided abdominal pain (Fig. 1). The lesion measured 5.6 cm in diameter, and the patient exhibited no additional symptoms
such as palpitations, fatigue, central obesity, or purple striae. Laboratory tests indicated elevated catecholamine levels and their metabolites, suggesting a diagnosis of pheochromocytoma. The patient had a history of DCM for 2 years and reported
recent shortness of breath during stair climbing. There was no other significant medical history.
Upon admission, the physical examination revealed the following: height 172 cm, weight 89 kg, pulse rate 78 bpm, and blood pressure 112/78 mmHg. The patient was alert with a regular heart rhythm, clear lung sounds, and no lower limb edema. Other
medical tests showed no significant abnormalities. Blood analysis indicated normal platelet counts, white and red blood cell counts, coagulation parameters, and liver and kidney functions. Urinary tests showed a 24-hour normetanephrine level of 1,005
µg/g creatinine (normal range: 70.00- 335.00 µg/g), and metanephrine level of 178 µg/g creatinine (normal range: 20.0-150.0 µg/g). Plasma normetanephrine was at 526 ng/L (normal range: 20.1-135.40 ng/L), while plasma metanephrine was significantly
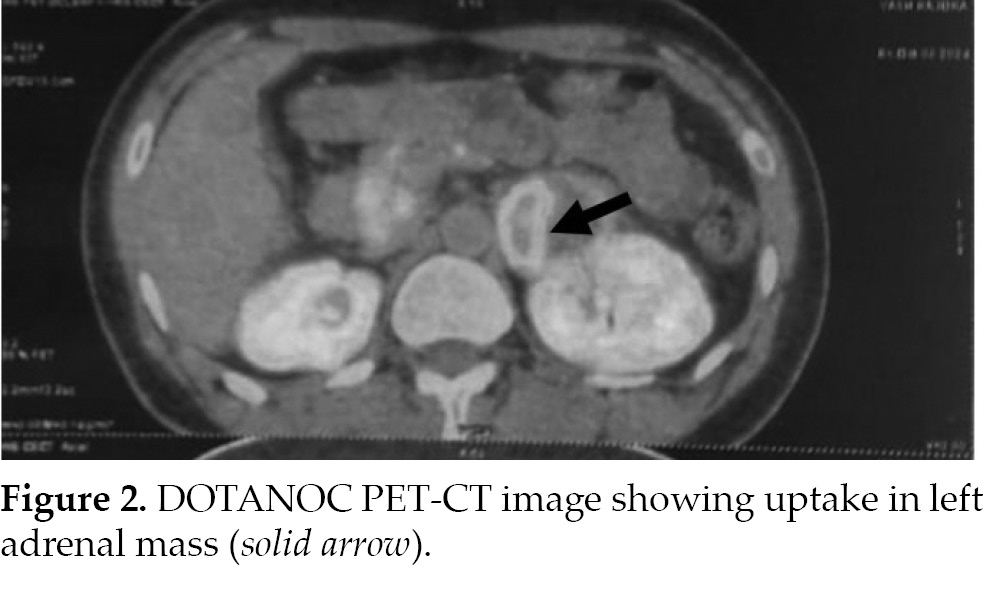
elevated at 3,910 ng/L (normal range: 7.90-88.7 ng/L). A DOTANOC-PET/CT scan indicated increased uptake in the left adrenal mass with no abnormal uptake elsewhere in the body (Fig. 2). A two-dimensional echocardiogram revealed an ejection fraction
of 20%, along with dilation of the left atrium and ventricle and global hypokinesia.
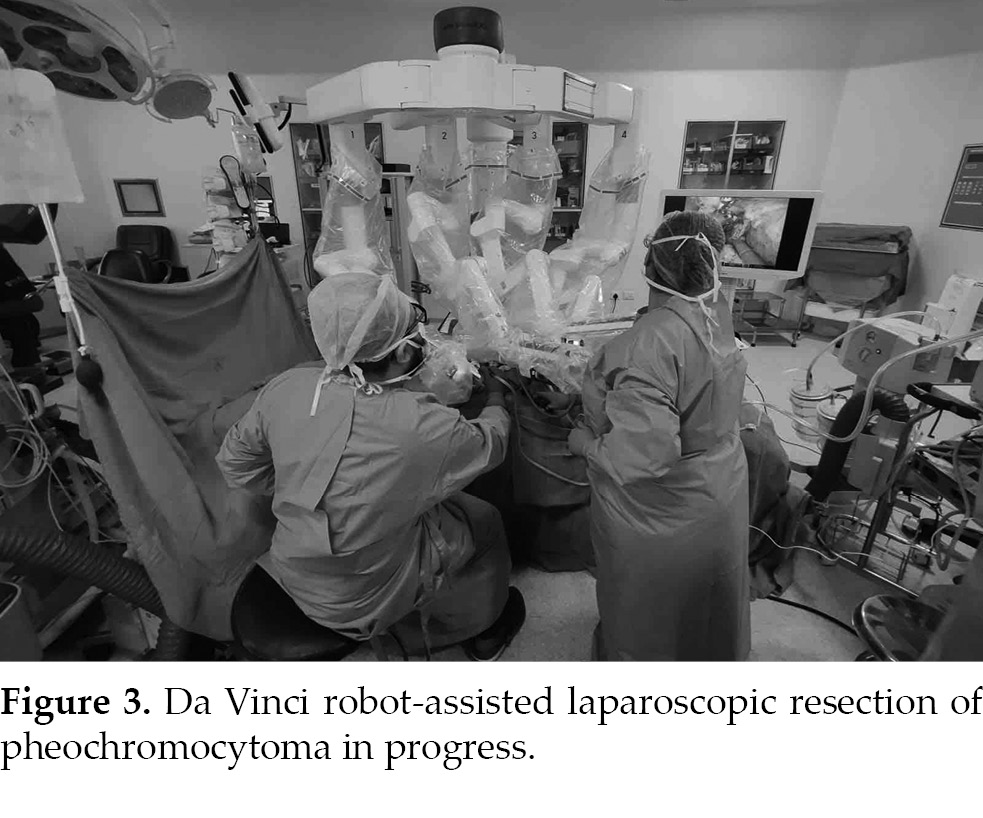
The preoperative diagnosis was established as left pheochromocytoma with DCM. The patient underwent a Da Vinci robot-assisted laparoscopic resection of the left adrenal pheochromocytoma (Fig. 3). General anesthesia induction was planned during
endotracheal intubation, along with invasive arterial pressure monitoring, central venous pressure monitoring, intraoperative transesophageal echocardiography (TEE), and bispectral index monitoring. Preoperative multidisciplinary consultation
included administering prazosin (20 mg/day for 2 weeks) and metoprolol (25 mg/day for 1 week), along with echocardiography and N-terminal pro-B-type natriuretic peptide (NT-proBNP) assessments every 2 days to manage blood pressure and provide volume
expansion therapy for 1 week, while continuously monitoring cardiac function.
Anesthesia induction involved intravenous administration of midazolam (1 mg), etomidate (20 mg), cisatracurium (16 mg), dexmedetomidine (0.5 µg/kg/h for 30 minutes), and fentanyl (100 µg). Maintenance included propofol (3-5 mg/kg/h), fentanyl (0.5 µg/kg/h),
cisatracurium (6 mg/h), and sevoflurane (1%) inhalation. Throughout the procedure, intraoperative cardiac function and left heart volume status were monitored using TEE following general anesthesia induction with endotracheal intubation. Invasive
arterial blood pressure, central venous pressure, depth of anesthesia, and temperature were also monitored during surgery.
To prevent stress ulcers and potential esophageal damage from the TEE probe, intravenous omeprazole was administered, along with methylprednisolone to enhance stress response capability during the operation; human serum albumin was also given. Intraoperative
TEE showed a left ventricular ejection fraction ranging from 27% to 31% and a normal inferior vena cava diameter (16-18 mm). The total operative time was 2 hours and 15 minutes, with uneventful procedures performed throughout.
Intraoperative anesthesia remained stable with parameters including invasive arterial blood pressure between 110 to 130 mmHg/66 to 78 mmHg; central venous pressure between 6 to 10 cmH2O; intraoperative blood loss at 250 mL; urine output at
400 mL and infusion volume at 1,500 mL. The adrenal mass was successfully excised, with postoperative histopathology confirming pheochromocytoma (Fig. 4). Postoperative analgesia was initiated under close observation, leading to the patient’s transfer
to the intensive care unit for further monitoring before returning to the general ward 3 days post-operation. He was discharged 4 days later in good condition and expressed satisfaction with his treatment and recovery.
DISCUSSION
This report outlines the successful execution of a rare, high-risk surgical procedure assisted by robotic technology, along with the optimization of anesthesia management through comprehensive preoperative evaluation and preparation. Intraoperative
monitoring of cardiac function and hemodynamics was crucial for accurately guiding fluid therapy and the administration of vasoactive medications. An effective multidisciplinary team, comprising hospital physicians, anesthetists, urologists, and
cardiovascular specialists, is essential in managing patients with DCM and pheochromocytoma6. Optimal anesthetic management for patients with DCM necessitates comprehensive preoperative assessment, vigilant perioperative monitoring, appropriate
anesthesia techniques, refined fluid management, and maintenance of stable hemodynamic conditions.
Pheochromocytoma releases catecholamines that can lead to catecholamine-induced cardiomyopathy, including DCM; however, this complication is rarely reported7. Chronic use of adrenaline inhalers may also contribute to DCM development8.
The mechanisms behind cardiomyopathy in pheochromocytoma patients include catecholamine-induced vasoconstriction of small arterioles, direct toxic effects from catecholamines and their metabolites, as well as receptor-mediated pathways9.
Pheochromocytoma may result in myocardial fibrosis along with both systolic and diastolic dysfunction. While impaired myocardial function can often be improved through medical treatment or surgery, complete restoration may not be achievable in all
patients
10.
Early surgical intervention is critical for managing catecholamine-induced cardiomyopathy associated with pheochromocytoma, with laparoscopic tumor removal being the preferred approach. Robot-assisted laparoscopy can significantly enhance perioperative
conditions by reducing stress and energy expenditure while minimizing complications11. This precision surgical technique lessens stimulation from pheochromocytomas, thereby improving safety during the procedure. Consequently, we successfully
performed a Da Vinci robot-assisted laparoscopic resection of the adrenal pheochromocytoma. There are documented cases indicating that surgical treatment can reverse myocardial dysfunction12.
Preoperative preparation for pheochromocytoma resection must meet stringent standards, including maintaining blood pressure below 120/80 mmHg, a heart rate of 60 to 80 bpm, and ensuring electrocardiographic recovery from ventricular premature contractions.
Additionally, there should be a decrease in hematocrit of more than 5% following volume expansion therapy, alongside improvements in hypermetabolic syndrome and correction of impaired glucose metabolism13. Elevated catecholamine levels
can cause sustained vasoconstriction and reduced effective circulating blood volume, leading to hypertension, tachycardia, hypovolemia, and hemoconcentration in patients. In this case, the patient had DCM with a low ejection fraction of 20%,
necessitating careful monitoring and gradual volume expansion therapy to avoid exacerbating heart failure. Preoperative blockade with alpha-blockers is recommended for all patients with hormonally active paragangliomas, and combining these with calcium
antagonists may further stabilize intraoperative blood pressure. Proper preoperative anesthesia preparation is essential for maintaining stable circulation during the surgical procedure14.
The perioperative mortality rate for pheochromocytomas is notably high, with risks including hypertensive crises and heart failure. However, hypotension and hypoglycemic shock can occur following tumor resection. Additionally, the patient presented with
DCM. To optimize anesthesia induction and management, we closely monitored the depth of anesthesia, cardiac function, and hemodynamics, maintaining systolic blood pressure between 90 and 140 mmHg and diastolic blood pressure between 60 and 90 mmHg.
The patient was susceptible to hypotension and tachycardia after anesthesia induction, prompting the administration of norepinephrine via a micropump to stabilize his condition. These measures ensured that the surgery proceeded safely and smoothly.
Anesthesia induction was conducted smoothly, ensuring adequate sedation, analgesia, and muscle relaxation. We utilized intravenous anesthesia while continuously monitoring the depth of anesthesia to maintain stability throughout the procedure.
Perioperative hemodynamic instability is a significant challenge during pheochromocytoma surgery, especially in patients with compromised cardiac function15. To manage this, an intraoperative TEE probe was used for real-time monitoring of left
heart function and volume, which is crucial for guiding fluid management. TEE revealed a stable left ventricular ejection fraction of 28% to 32%, and careful monitoring of invasive arterial and central venous pressures allowed for timely adjustments
in vasoactive drug administration. Goal-directed fluid therapy was implemented to replenish intravascular volume while avoiding fluid overload, which could lead to complications such as heart failure. Continuous monitoring of blood glucose levels
post-surgery was also essential to prevent hypoglycemia, particularly in patients with epinephrine-predominant tumors16.
CONCLUSION
Anesthetic management for adrenal pheochromocytoma resection in adult patients with DCM presents significant risks but is achievable with careful planning. Our multidisciplinary team conducted thorough preoperative assessments and maintained vigilant
perioperative monitoring; anesthesia and fluid management were optimized to achieve stable hemodynamics throughout the procedure. The use of robot-assisted laparoscopy further enhanced surgical safety. The challenges inherent in perioperative anesthetic
management can be mitigated through invasive monitoring techniques and minimally invasive surgical approaches. This case underscores the importance of personalized management strategies and multidisciplinary collaboration for achieving successful
outcomes.
REFERENCES
-
Schultheiss HP, Fairweather D, Caforio ALP, Escher F, Hershberger RE, Lipshultz SE, et al. Dilated cardiomyopathy. Nat Rev Dis Primers. 2019;5(1):32.
-
Chen CQ, Wang X, Zhang J, Zhu SM. Anesthetic management of patients with dilated cardiomyopathy for noncardiac surgery. Eur Rev Med Pharmacol Sci. 2017;21(3):627-34.
-
Naranjo J, Dodd S, Martin YN. Perioperative management of pheochromocytoma. J Cardiothorac Vasc Anesth. 2017;31(4):1427-39.
-
Azadeh N, Ramakrishna H, Bhatia NL, Charles JC, Mookadam F. Therapeutic goals in patients with pheochromocytoma: a guide to perioperative management. Ir J Med Sci. 2016;185(1):43-9.
-
Kassim TA, Clarke DD, Mai VQ, Clyde PW, Mohamed Shakir KM. Catecholamine-induced cardiomyopathy. Endocr Pract. 2008;14(9):1137-49.
-
Ramachandran R, Rewari V. Current perioperative management of pheochromocytomas. Indian J Urol. 2017;33(1):19-25.
-
Sethi P, Peiris CD. A review of catecholamine associated cardiomyopathies and channelopathies. Cureus. 2020;12(2):e6957.
- Stewart MJ, Fraser DM, Boon N. Dilated cardiomyopathy associated with chronic overuse of an adrenaline inhaler. Br Heart J. 1992;68(2):221-2.
-
Vladimirova-Kitova L, Kitov S, Uchikov P, Pavlov G. Catecholamine-induced cardiomyopathy - a clinical case. Folia Med (Plovdiv). 2022;64(1):156-61.
- Kalra Y, Agarwal HS, Smith AH. Perioperative management of pheochromocytoma and catecholamine-induced dilated cardiomyopathy in a pediatric patient. Pediatr Cardiol. 2013;34(8):2013-6.
- Bihain F, Klein M, Nomine-Criqui C, Brunaud L. Robotic adrenalectomy in patients with pheochromocytoma: a systematic review. Gland Surg. 2020;9(3):844-8.
- Sadowski D, Cujec B, McMeekin JD, Wilson TW. Reversibility of catecholamine-induced cardiomyopathy in a woman with pheochromocytoma. CMAJ. 1989;141(9):923-4.
- Fang F, Ding L, He Q, Liu M. Preoperative management of pheochromocytoma and paraganglioma. Front Endocrinol (Lausanne). 2020;11:586795.
- Lenders JW, Duh QY, Eisenhofer G, Gimenez-Roqueplo AP, Grebe SK, Murad MH, et al. Pheochromocytoma and paraganglioma: an Endocrine Society Clinical Practice guideline. J Clin Endocrinol Metab. 2014;99(6):1915-42.
- Pisarska M, Pedziwiatr M, Budzynski A. Perioperative hemodynamic instability in patients undergoing
laparoscopic adrenalectomy for pheochromocytoma. Gland Surg. 2016;5(5):506-11.
- Chen Y, Hodin RA, Pandolfi C, Ruan DT, McKenzie TJ. Hypoglycemia after resection of pheochromocytoma. Surgery. 2014;156(6):1404-8; discussion 1408-9.